
Study Guide with Student Solutions Manual for Seager/Slabaugh/Hansen's Chemistry for Today: General, Organic, and Biochemistry, 9th Edition
9th Edition
ISBN: 9781305968608
Author: Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11.52E
Which of the following cycloalkanes could show geometric isomerism? For each that could, draw structural formulas, and name both the cis- and the trans- isomers.
a. 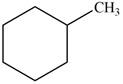 c.
c. 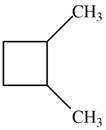
b. 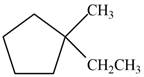 d.
d. 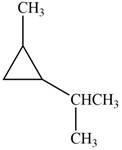
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Condensation polymers are produced when monomers containing two different functional groups link together with the loss of a small molecule such as H2O.
The difunctional monomer H2N(CH2)6COOH forms a condensation polymer. Draw the carbon-skeleton structure of the dimer that forms from this monomer.
What is the structure of the monomer?
→
BINDERIYA GANBO... BINDERIYA GANBO.
AP Biology Notes
Gamino acid chart - G...
36:22
司
10
☐ Mark for Review
Q
1
Hide
80
8
2
=HA O=A¯ = H₂O
Acid
HIO
HBrO
HCIO
Question 10 of 35 ^
Σ
DELL
□
3
%
Λ
&
6
7
* ∞
8
do 5
$ 4
# m
3
°
(
9
Highlights & Notes
AXC
Sign out
C
Chapter 11 Solutions
Study Guide with Student Solutions Manual for Seager/Slabaugh/Hansen's Chemistry for Today: General, Organic, and Biochemistry, 9th Edition
Ch. 11 - Prob. 11.1ECh. 11 - Prob. 11.2ECh. 11 - Prob. 11.3ECh. 11 - Prob. 11.4ECh. 11 - Prob. 11.5ECh. 11 - Prob. 11.6ECh. 11 - Prob. 11.7ECh. 11 - Prob. 11.8ECh. 11 - Prob. 11.9ECh. 11 - Prob. 11.10E
Ch. 11 - Prob. 11.11ECh. 11 - Prob. 11.12ECh. 11 - Prob. 11.13ECh. 11 - Prob. 11.14ECh. 11 - What molecular geometry exists when a central...Ch. 11 - Compare the shapes of unhybridized p and...Ch. 11 - Use Example 11.1 and Tables 11.2 and 11.6 to...Ch. 11 - Prob. 11.18ECh. 11 - Prob. 11.19ECh. 11 - Prob. 11.20ECh. 11 - Prob. 11.21ECh. 11 - Prob. 11.22ECh. 11 - Prob. 11.23ECh. 11 - Write a condensed structural formula for the...Ch. 11 - Write a condensed structural formula for the...Ch. 11 - Write an expanded structural formula for the...Ch. 11 - Prob. 11.27ECh. 11 - Classify each of the following compounds as a...Ch. 11 - Why are different conformations of an alkane not...Ch. 11 - Which of the following pairs represent structural...Ch. 11 - Prob. 11.31ECh. 11 - Prob. 11.32ECh. 11 - Identify the following alkyl groups: a. b....Ch. 11 - Prob. 11.34ECh. 11 - Prob. 11.35ECh. 11 - Draw a condensed structural formula for each of...Ch. 11 - Prob. 11.37ECh. 11 - Prob. 11.38ECh. 11 - Prob. 11.39ECh. 11 - Prob. 11.40ECh. 11 - The following names are incorrect, according to...Ch. 11 - The following names are incorrect, according to...Ch. 11 - Prob. 11.43ECh. 11 - Write the correct IUPAC name for each of the...Ch. 11 - Write the correct IUPAC name for each of the...Ch. 11 - Draw the structural formulas corresponding to each...Ch. 11 - Prob. 11.47ECh. 11 - Which of the following pairs of cycloalkanes...Ch. 11 - Prob. 11.49ECh. 11 - Prob. 11.50ECh. 11 - Prob. 11.51ECh. 11 - Which of the following cycloalkanes could show...Ch. 11 - Prob. 11.53ECh. 11 - Using the prefix cis- or trans-, name each of the...Ch. 11 - Prob. 11.55ECh. 11 - The compound decane is a straight-chain alkane....Ch. 11 - Explain why alkanes of low molecular weight have...Ch. 11 - Suppose you have a sample of 2-methylhexane and a...Ch. 11 - Identify circle the alkanelike portions of the...Ch. 11 - Prob. 11.60ECh. 11 - Prob. 11.61ECh. 11 - Write a balanced equation for the incomplete...Ch. 11 - Prob. 11.63ECh. 11 - Prob. 11.64ECh. 11 - Prob. 11.65ECh. 11 - Prob. 11.66ECh. 11 - Prob. 11.67ECh. 11 - Prob. 11.68ECh. 11 - Would you expect a molecule of urea produced in...Ch. 11 - Prob. 11.70ECh. 11 - Prob. 11.71ECh. 11 - Prob. 11.72ECh. 11 - Prob. 11.73ECh. 11 - Prob. 11.74ECh. 11 - Prob. 11.75ECh. 11 - A semi-truck loaded with cyclohexane overturns...Ch. 11 - Prob. 11.77ECh. 11 - Oil spills along coastal shores can be disastrous...Ch. 11 - Prob. 11.79ECh. 11 - Prob. 11.80ECh. 11 - Use the generic formula for alkanes (CnH2n+2) to...Ch. 11 - Prob. 11.82ECh. 11 - Which of the following is an example of an alkane?...Ch. 11 - Prob. 11.84ECh. 11 - Prob. 11.85ECh. 11 - Prob. 11.86ECh. 11 - The deadly property of carbon monoxide, if...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which representation(s) show polymer structures that are likely to result in rigid, hard materials and those that are likely to result in flexible, stretchable, soft materials?arrow_forward3. Enter the molecular weight of the product obtained from the Williamson Ether Synthesis? OH OH & OH excess CH3l Ag₂Oarrow_forwardPlease answer 1, 2 and 3 on the endarrow_forward
- In the box below, specify which of the given compounds are very soluble in polar aprotic solvents. You may select more than one compound. Choose one or more: NaCl NH4Cl CH3CH2CH2CH2CH2CN CH3CH2OH hexan-2-one NaOH CH3SCH3arrow_forwardOn the following structure, select all of the atoms that could ACCEPT a hydrogen bond. Ignore possible complications of aromaticity. When selecting be sure to click on the center of the atom.arrow_forwardRank the compounds below from lowest to highest melting point.arrow_forward
- 18 Question (1 point) Draw the line structure form of the given partially condensed structure in the box provided. :ÖH HC HC H2 ΙΩ Н2 CH2 CH3 CH3 partially condensed formarrow_forwardsomeone else has already submitted the same question on here and it was the incorrect answer.arrow_forwardThe reaction: 2NO2(g) ⇌ N2O4(g) is an exothermic reaction, ΔH=-58.0 kJ/molrxn at 0°C the KP is 58.If the initial partial pressures of both NO2(g) and N2O4(g) are 2.00 atm:A) Is the reaction at equilibrium? If not, what is the value of Q? B) Which direction will the reaction go to reach equilibrium? C) Use an ICE table to find the equilibrium pressures.arrow_forward
- The dissociation of the weak acid, nitrous acid, HNO2, takes place according to the reaction: HNO2 (aq) ⇌ H+(aq) + NO2–(aq) K=7.2 X 10-4 When 1.00 mole of HNO2 is added to 1.00 L of water, the H+ concentration at equilibrium is 0.0265 M.A) Calculate the value of Q if 1.00 L of water is added? B) How will reaction shift if 1.00 L of water is added?arrow_forwardSuppose a certain copolymer elastomeric material “styrene-butadiene rubber”) contains styrene ("S") monomers –(C8H8)– and butadiene ("B") monomers –(C4H6)– and that their numerical ratio S:B = 1:8. What is the mass ratio mS:mB of the two monomers in the material? What is the molecular mass M of a macromolecule of this copolymer with degree of polymerization n = 60,000? Data: AC = 12.01 u, AH = 1.008 u.arrow_forwardLab Questions from Lab: Gravimetric Determination of Calcium as CaC2O4•H2O What is the purpose of the methyl red indicator? Why does a color change to yellow tell you that the reaction is complete? Why is the precipitate rinsed with ice-cold water in step 4? Why not room temperature or hot water? Why is it important that the funnels be placed in a desiccator before weighing (steps 1 and 5)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License