
Interpretation: Given the decomposition of
Concept Introduction:
The integrated rate law equation explains how the concentrations of reactants change with time.
Consider a first order
The concentration of the reactant A at time t is given by the below equation
Where,
The integrated rate law for this first order reaction is obtained by taking the natural logarithm of both sides of
That is,
Using Dalton's law, the partial pressure of formic acid is given by
Where,
The order of the reaction can be determined from a plot of concentration against time.
If we plot concentration against time, and if the curve is linear, the reaction is a zero order reaction.
If we plot log of concentration against time and if the curve is linear, the reaction is a first order reaction.
If we plot concentration inverse against time and if the curve is linear, the reaction is a second order reaction.
Answer to Problem 11.50PAE
Solution: The rate constant is
Explanation of Solution
Given Information: The table containing the total pressures in the reaction vessel during the decomposition of
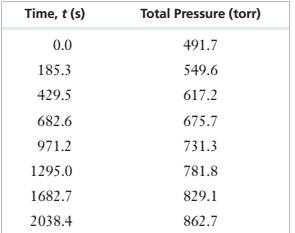
Decomposition of
The initial partial pressure of
Calculate the partial pressure of
Partial pressure of
Partial pressure of
Partial pressure of
Therefore, the total pressure at any given time t is given as
| Time(t) | Total pressure |
|
| 0 | 491.7 | 491.7 |
| 185.3 | 549.6 | 434.0 |
| 242.8 | 566.6 | 417.0 |
| 304.5 | 584.1 | 399.5 |
| 362.7 | 599.9 | 383.7 |
| 429.5 | 617.2 | 366.4 |
| 509.7 | 637.0 | 346.6 |
| 606.3 | 659.5 | 324.1 |
We need to plot these values of partial pressure with time to see if the reaction is zero order or not
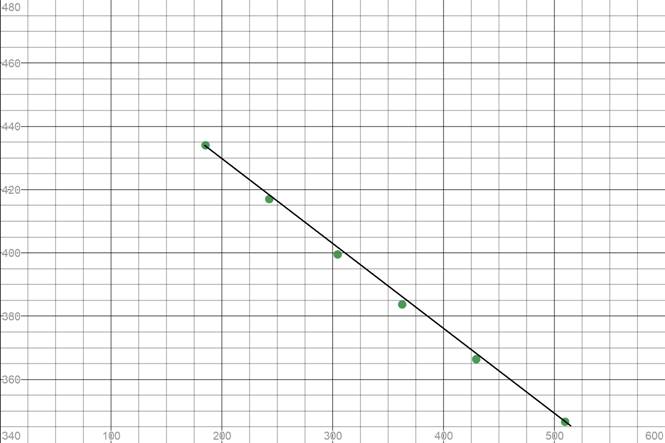
As some points do not lie on the straight line, the curve is not linear. Thus, it is not a zero order reaction.
| Time(t) | |
|
| 0 | 491.7 | 6.1978687744 |
| 185.3 | 434.0 | 6.0730445341 |
| 242.8 | 417.0 | 6.0330862218 |
| 304.5 | 399.5 | 5.9902137652 |
| 362.7 | 383.7 | 5.9498609973 |
| 429.5 | 366.4 | 5.9037256328 |
| 509.7 | 346.6 | 5.8481713773 |
| 606.3 | 324.1 | 5.7810521101 |
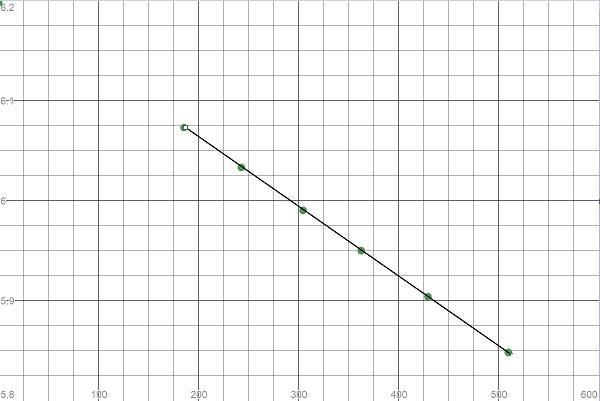
Here we see the curve is linear and thus the reaction is a first order reaction.
To calculate the rate constant, we need the negative slope of the line in the plot
Hence, the rate constant is the negative of the slope obtained. It is equal to
The concept of integrated rate law and the manipulation the data into a plot helps in determining the order of the decomposition of
Want to see more full solutions like this?
Chapter 11 Solutions
Bundle: Chemistry for Engineering Students, 3rd, Loose-Leaf + OWLv2 with QuickPrep 24-Months Printed Access Card
- draw the enolate anion and the carbonyl that would be needed to make this product through an aldol addition reaction.arrow_forwardDraw the Michael Adduct and the final product of the Robinson annulation reaction. Ignore inorganic byproducts.arrow_forwardDraw the Michael adduct and final product of the Robinson annulation reaction. Ignore inorganic byproductsarrow_forward
- Post Lab Questions. 1) Draw the mechanism of your Diels-Alder cycloaddition. 2) Only one isomer of product is formed in the Diels-Alder cycloaddition. Why? 3) Imagine that you used isoprene as diene - in that case you don't have to worry about assigning endo vs exo. Draw the "endo" and "exo" products of the Diels-Alder reaction between isoprene and maleic anhydride, and explain why the distinction is irrelevant here. 4) This does not hold for other dienes. Draw the exo and endo products of the reaction of cyclohexadiene with maleic anhydride. Make sure you label your answers properly as endo or exo. 100 °C Xylenes ??? 5) Calculate the process mass intensity for your specific reaction (make sure to use your actual amounts of reagent).arrow_forwardIndicate the product(s) A, B C and D that are formed in the reaction: H + NH-NH-CH [A+B] [C+D] hydrazonesarrow_forwardHow can you prepare a 6 mL solution of 6% H2O2, if we have a bottle of 30% H2O2?arrow_forward
- How many mL of H2O2 from the 30% bottle must be collected to prepare 6 mL of 6% H2O2.arrow_forwardIndicate the product(s) B and C that are formed in the reaction: HN' OCH HC1 B + mayoritario C minoritario OCH3arrow_forwardIndicate the product(s) that are formed in the reaction: NH-NH, OCH3 -H₂O OCH3arrow_forward
- 21.38 Arrange the molecules in each set in order of increasing acidity (from least acidic to most acidic). OH OH SH NH2 8 NH3 OH (b) OH OH OH (c) & & & CH3 NO2 21.39 Explain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH OH OH OH PK 10.0 PK 8.81 PK 9.28 PK 9.81arrow_forwardidentify which spectrum is for acetaminophen and which is for phenacetinarrow_forwardThe Concept of Aromaticity 21.15 State the number of 2p orbital electrons in each molecule or ion. (a) (b) (e) (f) (c) (d) (h) (i) DA (k) 21.16 Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hückel criteria? Which, if planar, would be antiaromatic? 21.17 Which of the following structures are considered aromatic according to the Hückel criteria? ---0-0 (a) (b) (c) (d) (e) (h) H -H .8.0- 21.18 Which of the molecules and ions from Problem 21.17 have electrons donated by a heteroatom?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning  ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





