
a)
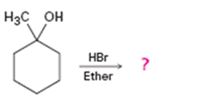
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
Alcohols gets converted into the corresponding bromides on treatment with HBr in ether solution.
To predict:
The product of the reaction shown.
Answer to Problem 26AP
The product of the reaction shown is
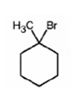
Explanation of Solution
When 1-methylcyclohexanol is treated with HBr, the OH group in it is replaced by the bromine to yield 1-chloro-1-methylcyclohexane and water as products.
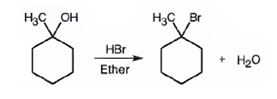
The product of the reaction shown is
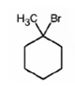
b)
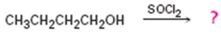
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
Alcohols when treated with SOCl2 yield the corresponding alkyl chlorides along with SO2 and HCl.
To predict:
The product of the reaction in which 1-butanol reacts with SOCl2 .
Answer to Problem 26AP
The product of the reaction in which 1-butanol reacts with SOCl2 is 1-chlorobutane.
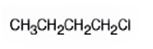
Explanation of Solution
When 1-butanol reacts with SOCl2, the OH group in is replaced by chlorine to yield 1-chlorobutane. SO2 and HCl are obtained as other products.
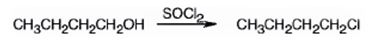
The product of the reaction in which 1-butanol reacts with SOCl2 is 1-chlorobutane.
c)
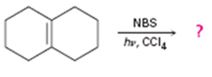
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
Cycloalkenes on treatment with NBS in CCl4 in the presence of light undergo bromination at the allyl position.
To predict:
The product of the reaction shown.
Answer to Problem 26AP
The products of the reaction shown are
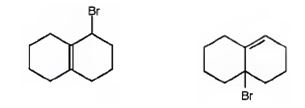
Explanation of Solution
NBS is normally used for allylic bromination. The reaction occurs through a radical mechanism. The radical produced by the homolytic cleavage of the C-H bond in the allyl position gets delocalized with the π electrons of the double bond to yield another radical with an odd electron at the junction of the rings. The attack of bromine radical hence leads to two different products as shown.
The products of the reaction shown are

d)
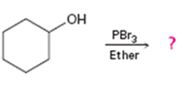
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
Alcohols give the corresponding bromides as products on treatment with PBr3 in ether solution.
To predict:
The product of the reaction in which cyclohexanol reacts with PBr3.
Answer to Problem 26AP
The product of the reaction in which cyclohexanol reacts with PBr3is
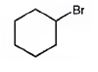
Explanation of Solution
When cyclohexanol is treated with PBr3, the OH group in it is replaced by the bromine to yield bromocyclohexane.
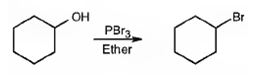
The product of the reaction in which cyclohexanol reacts with PBr3 is

e)
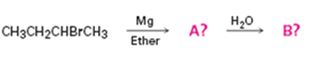
Interpretation:
The products of the reactions shown are to be predicted.
Concept introduction:
Alkyl bromides react with Mg in ether to give Grignard reagents. Grignard reagents when treated with water yield the corresponding
To predict:
The products of the reaction shown.
Answer to Problem 26AP
The products of the reaction shown are sec-butyl magnesium bromide (A) and n-butane (B).
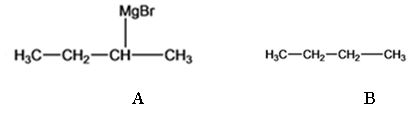
Explanation of Solution
sec-butyl bromide when reacted with Mg in ether yields sec-butyl magnesium bromide, a Grignard reagent. The sec-butyl magnesium bromide reacts with water to yield n-butane.
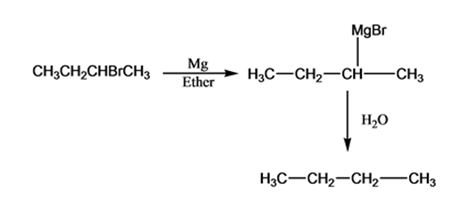
The products of the reaction shown are sec-butyl magnesium bromide (A) and n-butane (B).
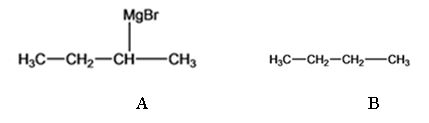
f)
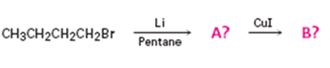
Interpretation:
The products of the reaction shown are to be predicted.
Concept introduction:
To predict:
The product of the reaction shown.
Answer to Problem 26AP
The products of the reaction shown are N-butyllithium (A) and Lithium n-dibutyl copper (B).
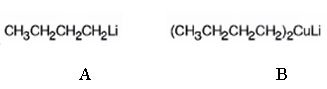
Explanation of Solution
n-Butyl bromide reacts with Li to yield n-butyl lithium and lithium bromide. The n-butyl lithium produced when reacted with CuI yields Lithium n-dibutyl copper as the product.
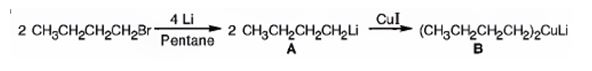
The products of the reaction shown are N-butyllithium (A) and Lithium n-dibutyl
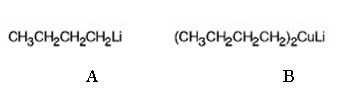
g)
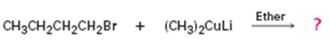
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
When alkyl halides are treated with lithium dimethyl copper, the halogens are displaced by an alkyl group and a higher alkane is produced as the product.
To predict:
The product of the reaction in which n-butyl bromide reacts with lithium dimethylcopper.
Answer to Problem 26AP
The product of the reaction in which n-butyl bromide reacts with lithium dimethylcopper is n-pentane.
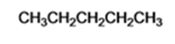
Explanation of Solution
When n-butyl bromide is treated with lithium dimethylcopper, the bromine is replaced by a methyl group to yield n-pentane as the product.
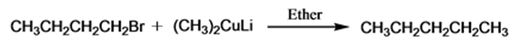
The product of the reaction in which n-butyl bromide reacts with lithium dimethylcopper is n-pentane.
Want to see more full solutions like this?
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY
- need help finding the product of these reactionsarrow_forwardPart 1. Draw monomer units of the following products and draw their reaction mechanism 1) Bakelite like polymer Using: Resorcinol + NaOH + Formalin 2) Polyester fiber Using a) pthalic anhydride + anhydrous sodium acetate + ethylene glycol B)pthalic anhydride + anhydrous sodium acetate + glycerol 3) Temporary cross-linked polymer Using: 4% polyvinyl alcohol+ methyl red + 4% sodium boratearrow_forwardUsing the table of Reactants and Products provided provide the correct letter that corresponds with the Carboxylic acid that is formed in the reaction below. 6 M NaOH Acid-workup WRITE THE CORRECT LETTER ONLY DO NOT WRITE EXTRA WORDS OR PHRASES A) Pool of Reagents for Part B CI B) OH C) E) CI J) racemic F) K) OH N) OH P) G) OH D) HO H) L) M) HO Q) R) CI Aarrow_forward
- In the table below, the exact chemical structures for Methyl salicylate can be represented by the letter WRITE THE CORRECT LETTER ONLY DO NOT WRITE EXTRA WORDS OR PHRASES CI B) A) E) Cl racemic F) J) CI K) N) OH P) Pool of Reagents for Part B OH OH G) L) OH D) HO H) M) HO Q) R) CIarrow_forwardDraw the stepwise mechanism for the reactionsarrow_forwardPart I. a) Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone b) Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone (3,3-dimethyl-2-butanone) and 2, 3-dimethyl - 1,3-butadiene. Give reasonable mechanism the formation of the products Forarrow_forward
- 3. The explosive decomposition of 2 mole of TNT (2,4,6-trinitrotoluene) is shown below: Assume the C(s) is soot-basically atomic carbon (although it isn't actually atomic carbon in real life). 2 CH3 H NO2 NO2 3N2 (g)+7CO (g) + 5H₂O (g) + 7C (s) H a. Use bond dissociation energies to calculate how much AU is for this reaction in kJ/mol.arrow_forwardPart I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone and (3,3-dimethyl-2-butanone) 2,3-dimethyl-1,3-butadiene. Give reasonable mechanism the formation of the products Forarrow_forwardShow the mechanism for these reactionsarrow_forward
- Draw the stepwise mechanismarrow_forwardDraw a structural formula of the principal product formed when benzonitrile is treated with each reagent. (a) H₂O (one equivalent), H₂SO₄, heat (b) H₂O (excess), H₂SO₄, heat (c) NaOH, H₂O, heat (d) LiAlH4, then H₂Oarrow_forwardDraw the stepwise mechanism for the reactionsarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

