
Concept explainers
(a)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to
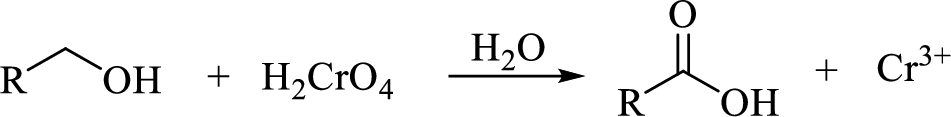
(b)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
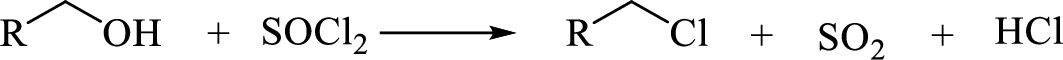
(c)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
Secondary alcohols may react by

(d)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
Secondary alcohols may react by

(e)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to carboxylic acid: A primary alcohol is oxidized to a carboxylic acid by chromic acid. The mechanism involves initial formation of an alkyl chromate intermediate, followed by reaction with base to remove a proton, generating the carbonyl group of an aldehyde and simultaneously reducing the chromium (VI) to chromium (IV). An initially formed aldehyde adds water, generating an aldehyde hydrate, which is oxidized according to the same mechanism to give the carboxylic acid.
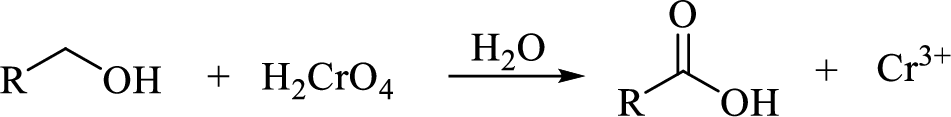
(f)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidative cleavage of a glycol:
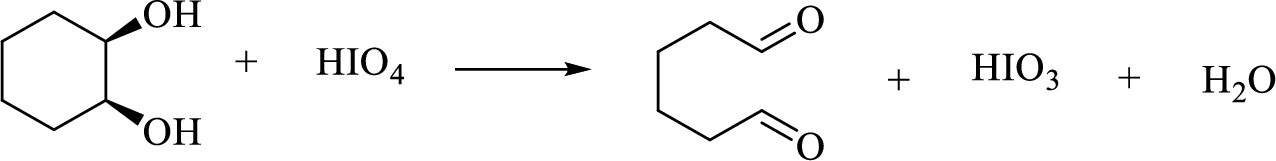
(g)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidative cleavage of a glycol:
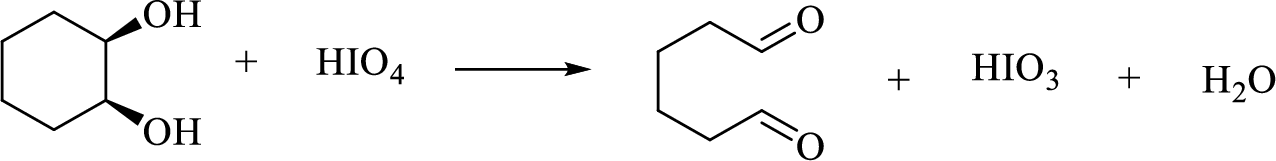
(h)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
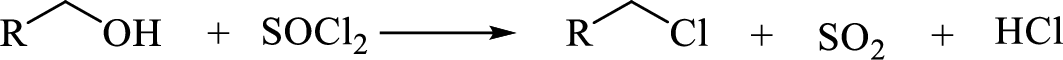
(i)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to an aldehyde: The oxidation of a primary alcohol to an aldehyde can be carried out using pyridinium chlorochromate (PCC). Because there is no water, the aldehyde does not form the hydrate, and the oxidation reaction stops at the aldehyde stage. Alternatively, Swern or Dess-martin oxidation can be used.

(j)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to an aldehyde: The oxidation of a primary alcohol to an aldehyde can be carried out using pyridinium chlorochromate (PCC). Because there is no water, the aldehyde does not form the hydrate, and the oxidation reaction stops at the aldehyde stage. Alternatively, Swern or Dess-martin oxidation can be used.

Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry, Loose-leaf Version
- Indicate the product that is obtained if the benzotriazol reacts with dimethyl sulfate.arrow_forwardIndicate how to obtain 2-metilbencimidazol from 1,2-diaminobenzene.arrow_forwardbreak down both reactions shown and explain it correctly using the bromonium ion mechanism, instead of the (disproven) carbocation-based mechanism.arrow_forward
- Indicate how from 1,2-diaminobenzene to obtain 1-metilbenzotriazol.arrow_forward-C = C - C - + Br₂ + I" -> -C-C-c -C = C -C- + Br² + I₂ -C=C Br I + Brū + Iz -7- C - C-C- I Br Mechanism; - C = c - c - + Br - Br > - C-c-c- Br -C-C-C- + 1 - - -Ċ-Ċ'-c' - Br Br Iarrow_forwardWrite the mechanism of the esterification reaction (please show the mechanism included line pairs and arrows)arrow_forward
- How do I break down the reaction shown on the chalkboard and explain it correctly using the bromonium ion mechanism, instead of the (disproven) carbocation-based mechanismarrow_forward¿Qué the product is obtained from tetraethoxypropano and hidrazina?. Indicate the reason why the corresponding dial is used.arrow_forwardIf CH3COCH2CH(OCH3)2 is reacted with hydrazine, two isomeric products are formed. Indicate their structures and the major product.arrow_forward
- Is it possible to obtain addition derivatives to nitrogen in position 2 of pyrazoles by reaction with electrophilic agents? Reason for this.arrow_forwardStarting from 1,3-dicarbonyl derivatives to obtain isooxazoles and isothiazoles. Indicate whether synthetic methods exist.arrow_forwardIn the synthesis of benzotriazole, adding NaNO2 heats the solution. State the reason.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





