
Concept explainers
(a)
Interpretation:
Constitutional isomers that has molecular formula
Concept Introduction:
Halogenation reaction is the one where atom or atoms of halogens get substituted in a carbon chain. Halogenation is a type of substitution reaction.
In
IUPAC rules for naming alkanes:
There are about five rules that has to be followed for naming an alkane and they are,
- The longest continuous carbon chain in the compound has to be identified. This is known as parent compound. From this the parent name is obtained. Suffix “–ane” (for alkane) is added at the end of the prefix which gives information about the number of carbon atoms.
- Numbering has to be done so that the lowest number is given to the first group that is encountered in the parent chain.
- Naming and numbering has to be given for each atom or group that is attached to the parent chain. Numbering has to be done in a way that substituents get the least numbering.
- If the same substitution is present in the parent chain more than once, a separate prefix is added which tells about the number of times the substituent occurs. Prefixes used are di-, tri-, tetra-, penta- etc.
- Name of the substituents has to be placed in an alphabetical order before the parent compound name.
(a)
Explanation of Solution
Given molecular formula of constitutional isomer is
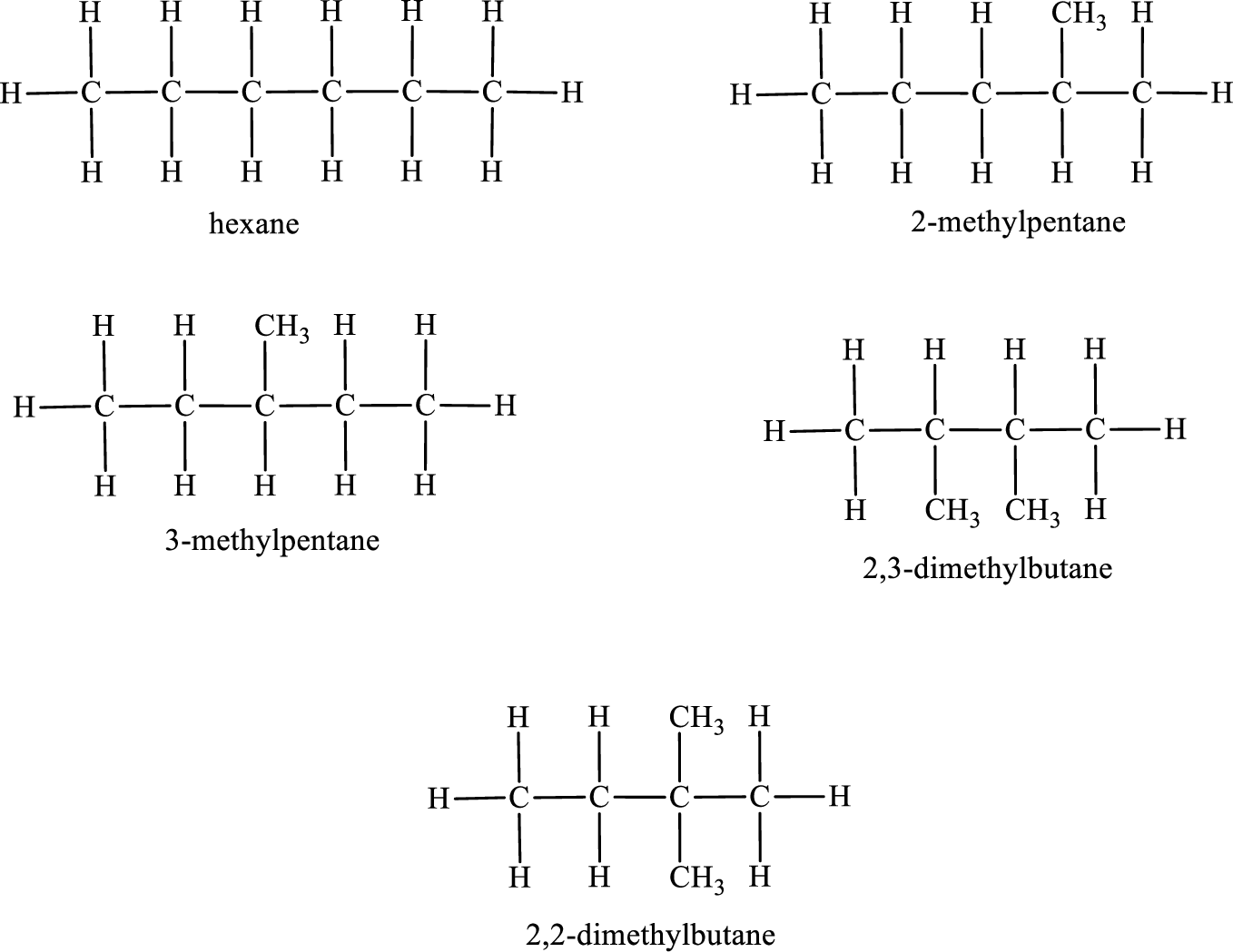
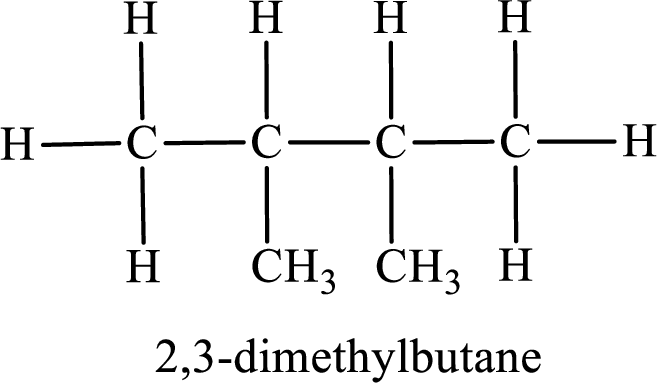
Among the constitutional isomers, the one that gives only two monobromo derivatives can be identified by looking into the structure that contain carbon atoms that has only two different environment. The structure is found to be 2,3-dimethylbutane.
The reaction can be given as,
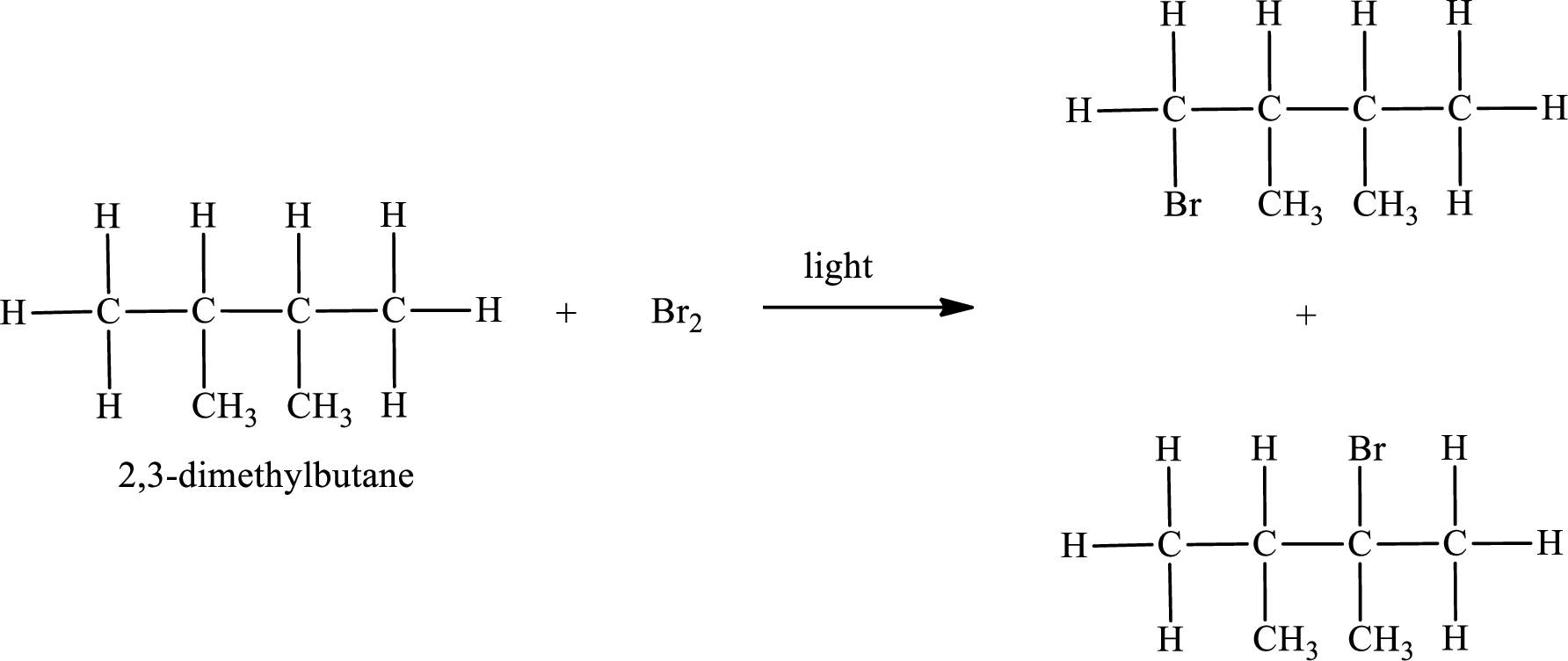
IUPAC names:
First monobromo derivative:
In the given compound, the longest carbon chain is found to contain four carbon atoms. Therefore, the parent alkane name is butane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
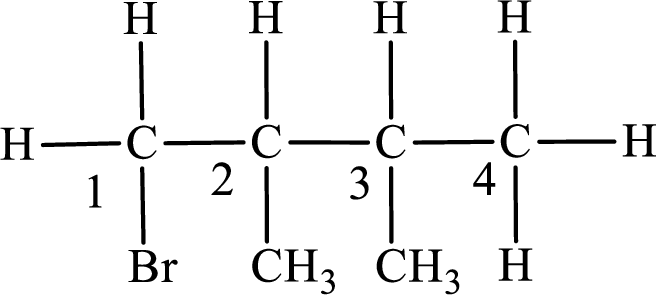
The substituent present in the given compound are a bromine atom and two methyl groups. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
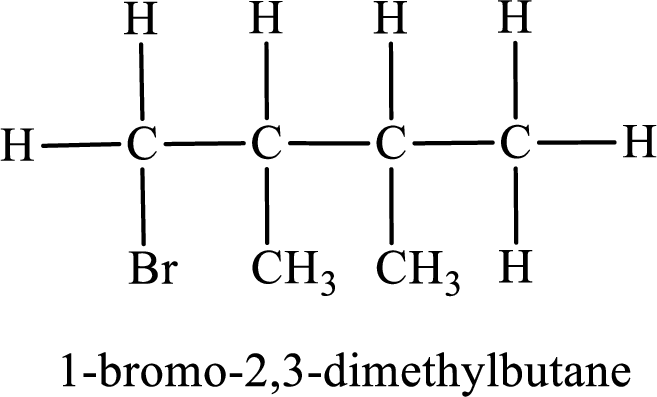
Parent chain is butane and the substituent present is 1-bromo-2,3-dimethyl. Hence, the IUPAC name is given as 1-bromo-2,3-dimethylbutane.
Second monobromo derivative:
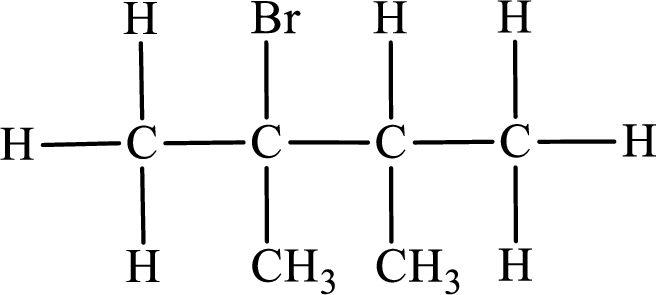
In the given compound, the longest carbon chain is found to contain four carbon atoms. Therefore, the parent alkane name is butane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
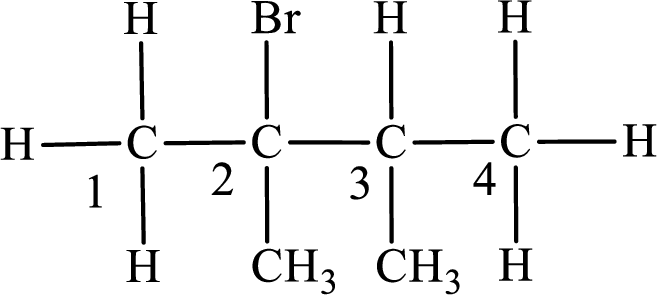
The substituent present in the given compound are a bromine atom and two methyl groups. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
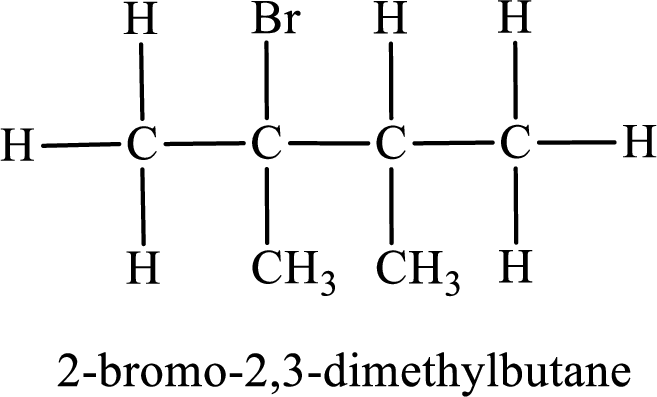
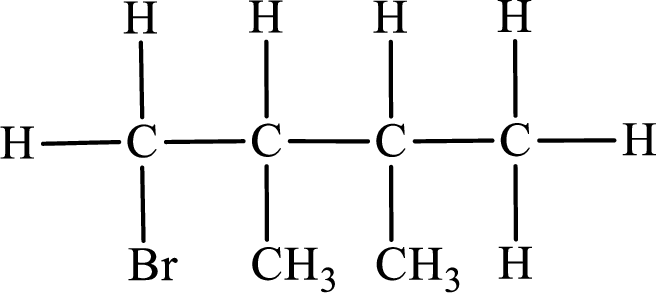
Parent chain is butane and the substituent present is 2-bromo-2,3-dimethyl. Hence, the IUPAC name is given as 2-bromo-2,3-dimethylbutane.
(b)
Interpretation:
Constitutional isomers that has molecular formula
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Given molecular formula of constitutional isomer is
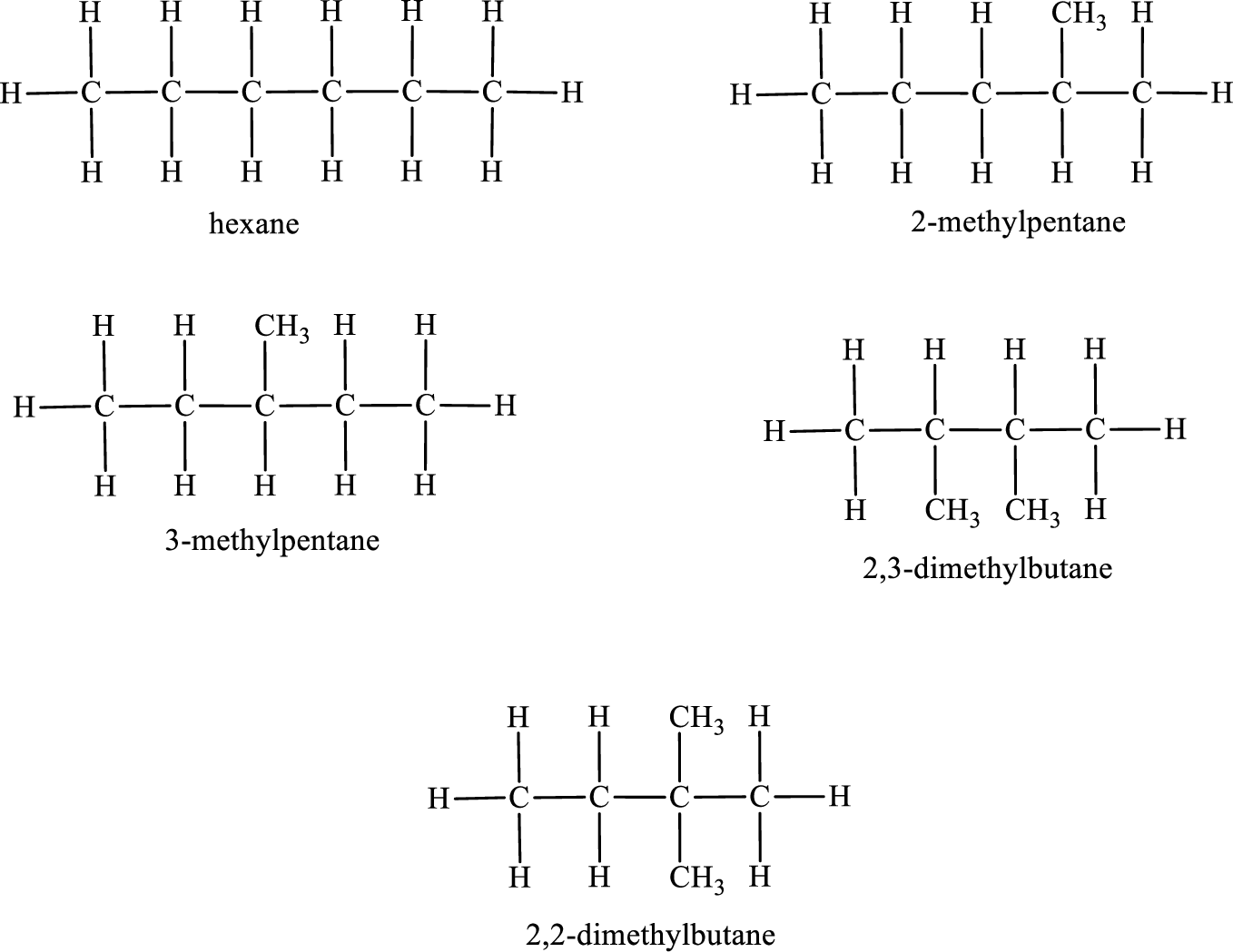
Among the constitutional isomers, the one that gives only three monobromo derivatives can be identified by looking into the structure that contain carbon atoms that has only three different environment. The structure is found to be hexane.
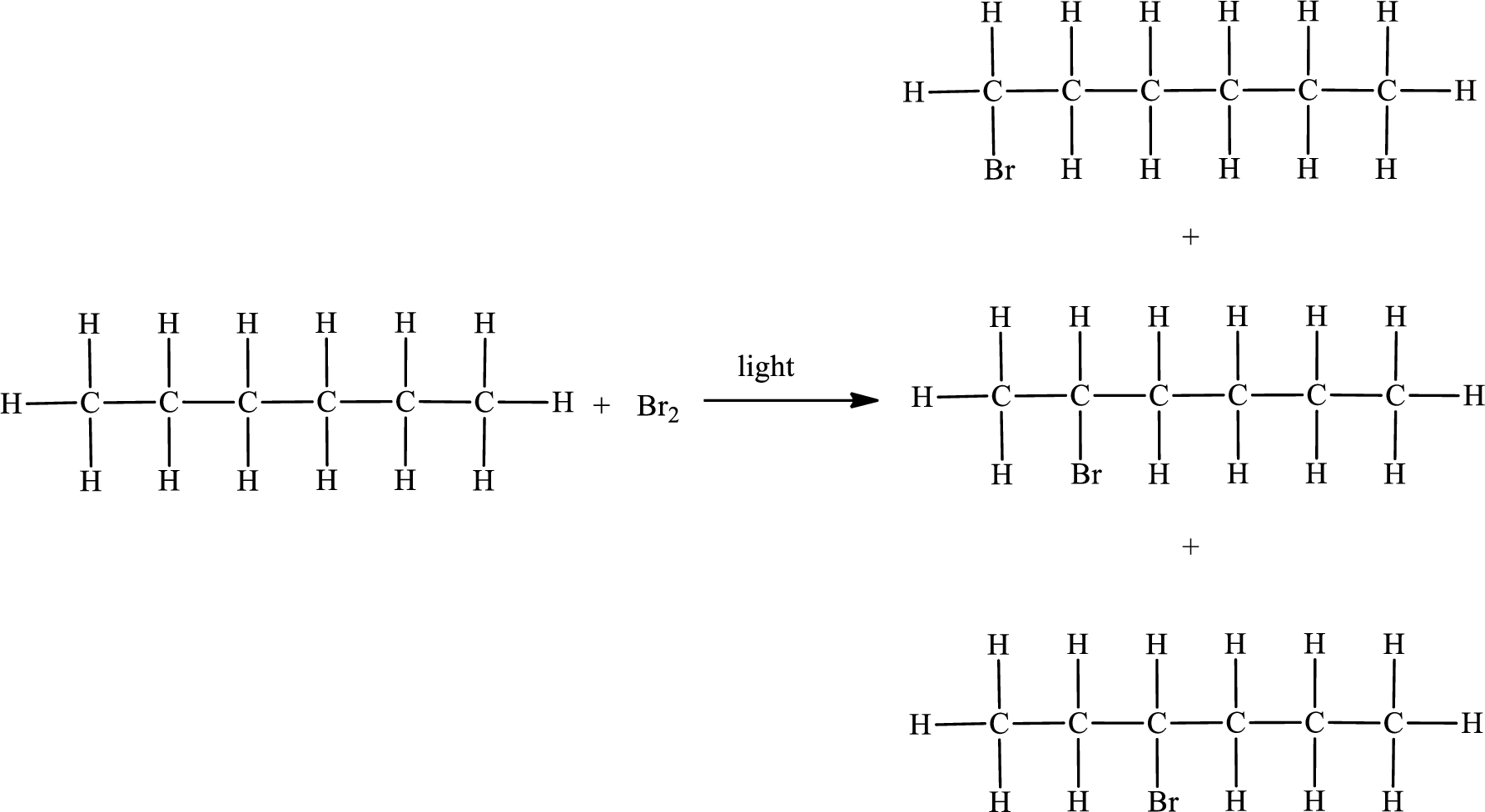
The reaction can be given as,
IUPAC names:
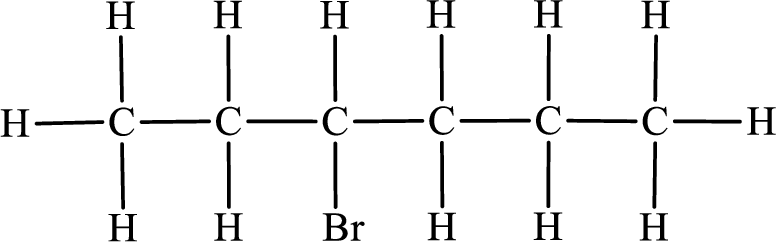
First monobromo derivative:
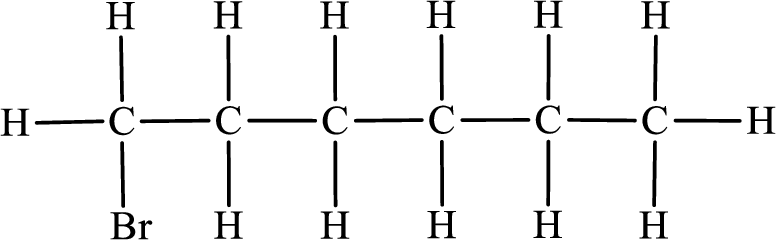
In the given compound, the longest carbon chain is found to contain six carbon atoms. Therefore, the parent alkane name is hexane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
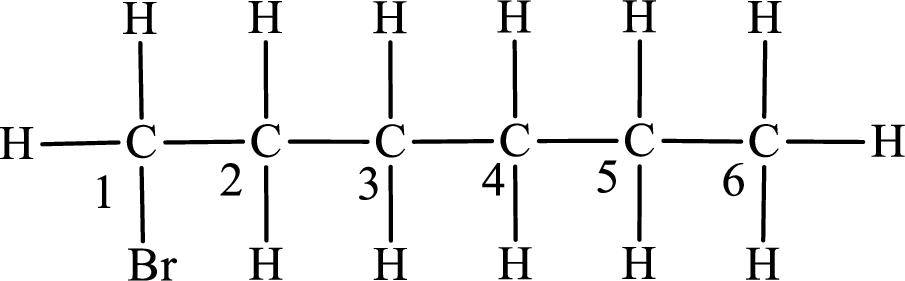
The substituent present in the given compound is a bromine atom. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
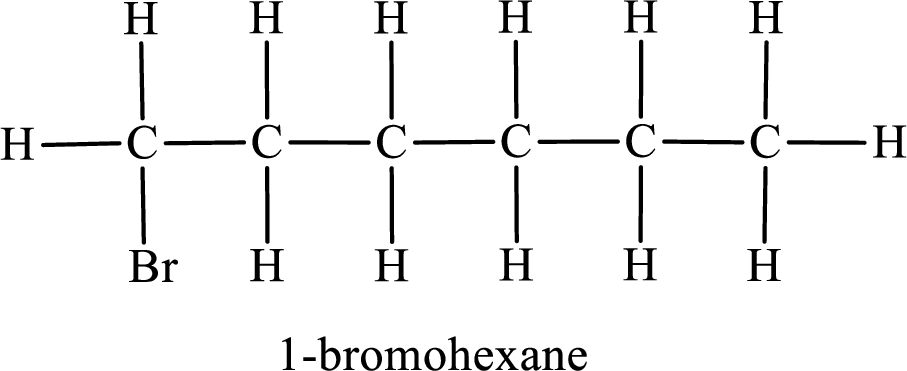
Parent chain is hexane and the substituent present is 1-bromo. Hence, the IUPAC name is given as 1-bromohexane.
Second monobromo derivative:
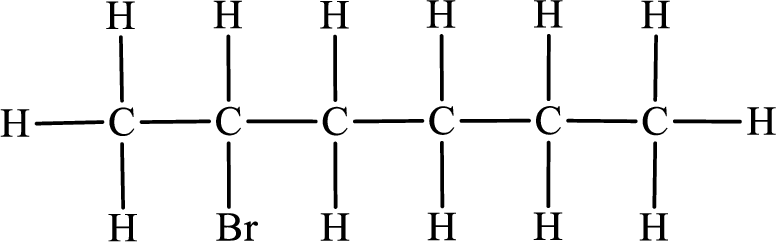
In the given compound, the longest carbon chain is found to contain six carbon atoms. Therefore, the parent alkane name is hexane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
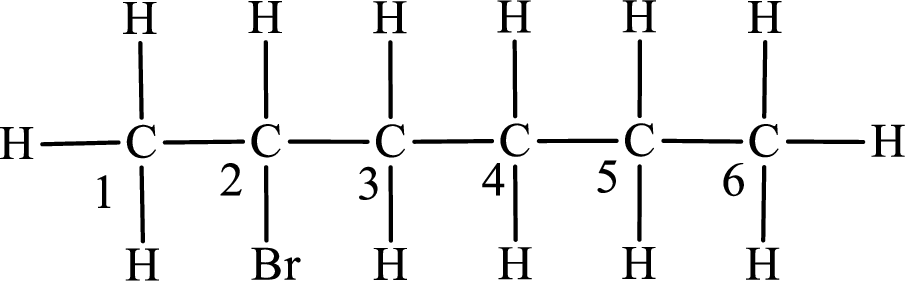
The substituent present in the given compound is a bromine atom. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
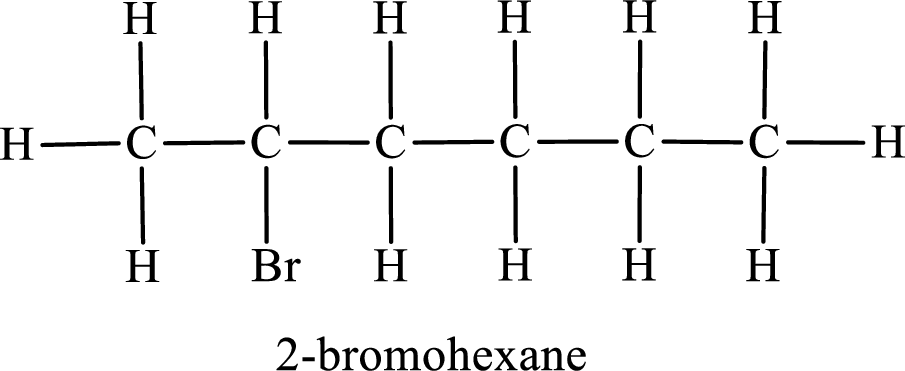
Parent chain is hexane and the substituent present is 2-bromo. Hence, the IUPAC name is given as 2-bromohexane.
Third monobromo derivative:
In the given compound, the longest carbon chain is found to contain six carbon atoms. Therefore, the parent alkane name is hexane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
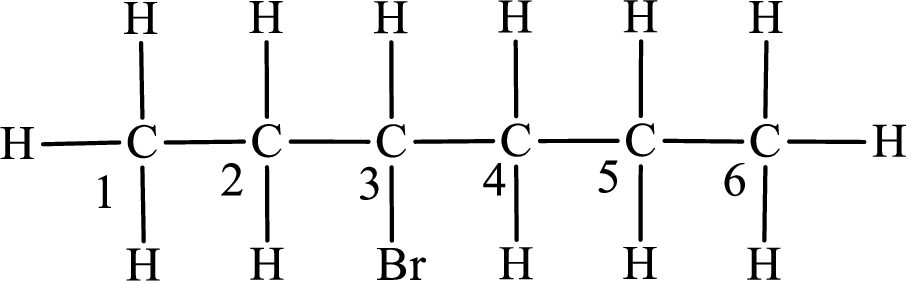
The substituent present in the given compound is a bromine atom. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
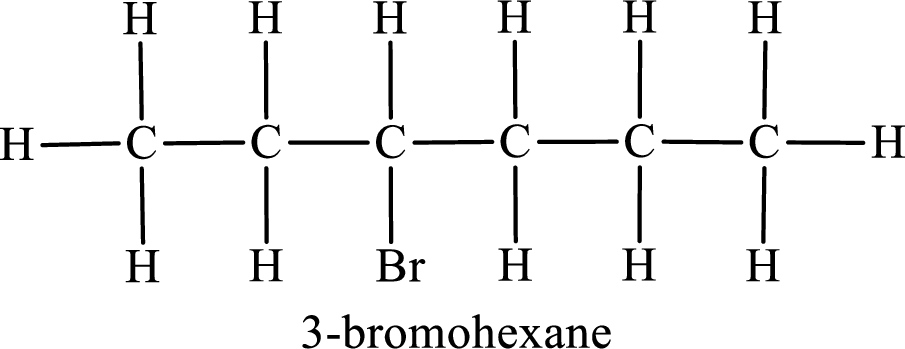
Parent chain is hexane and the substituent present is 3-bromo. Hence, the IUPAC name is given as 3-bromohexane.
(c)
Interpretation:
Constitutional isomers that has molecular formula
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
Given molecular formula of constitutional isomer is
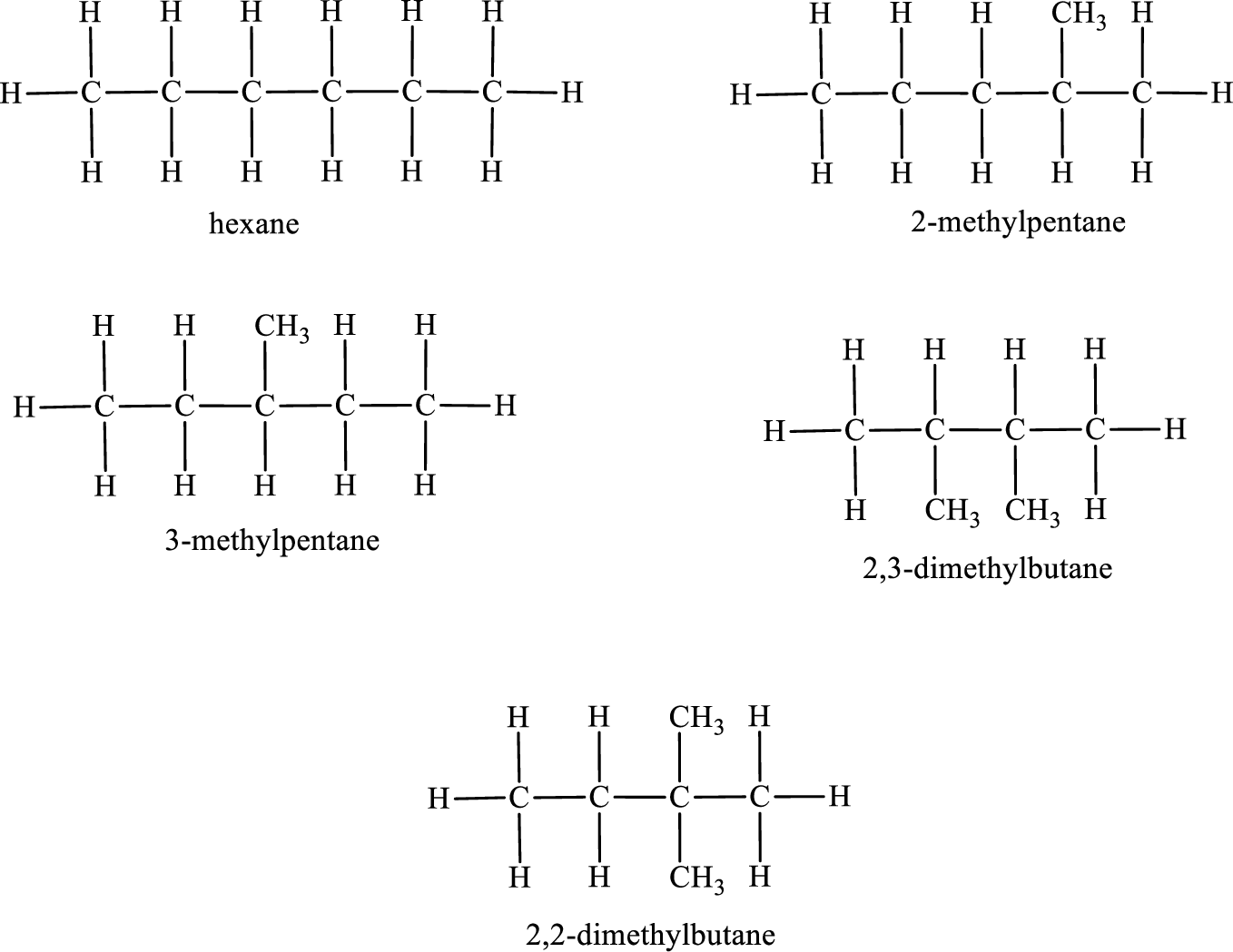
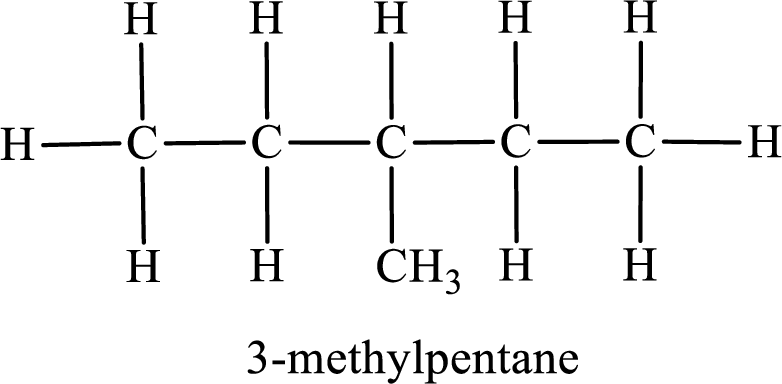
Among the constitutional isomers, the one that gives only four monobromo derivatives can be identified by looking into the structure that contain carbon atoms that has only four different environment. The structure is found to be 3-methylpentane.
The reaction can be given as,
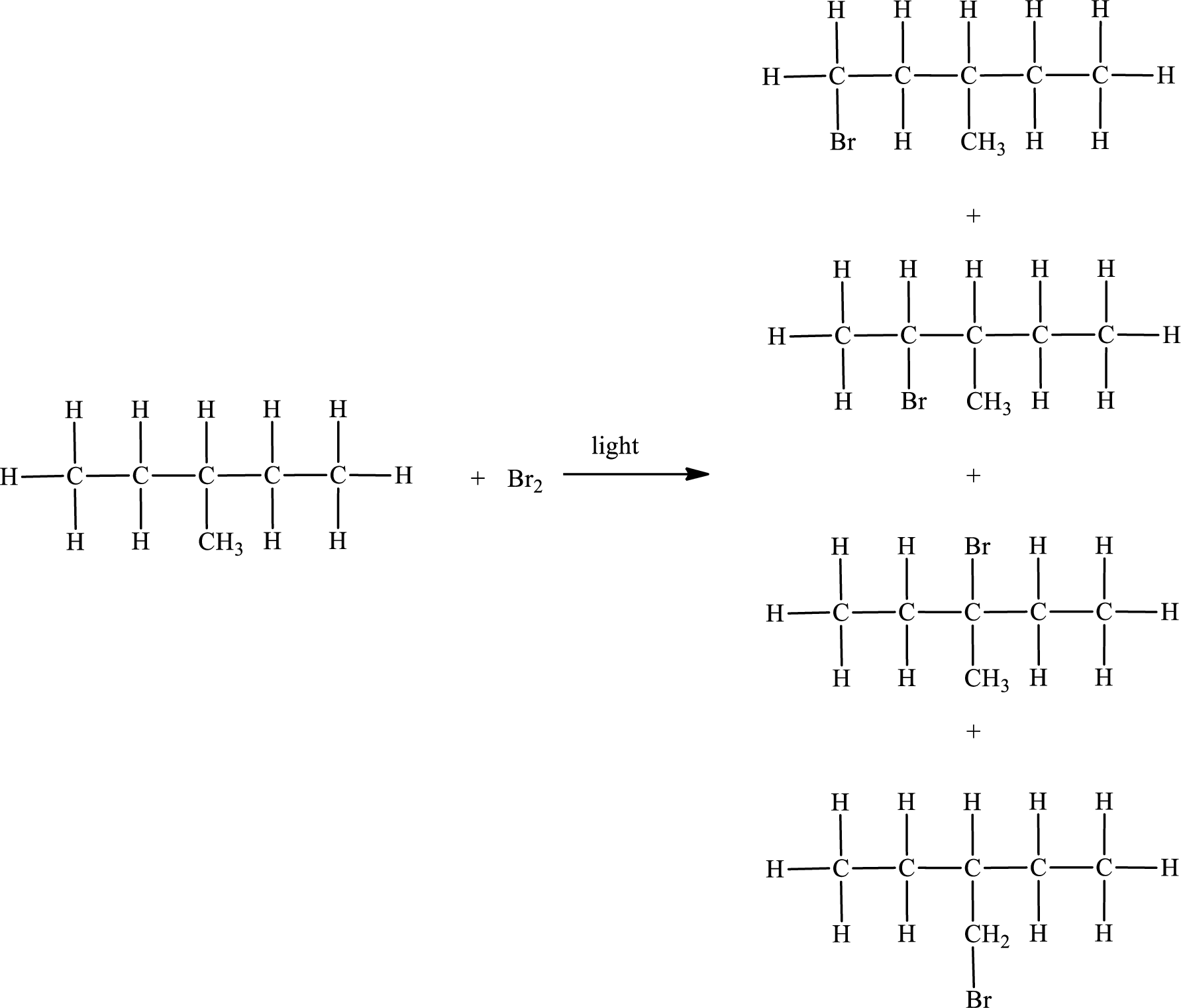
IUPAC names:
First monobromo derivative:
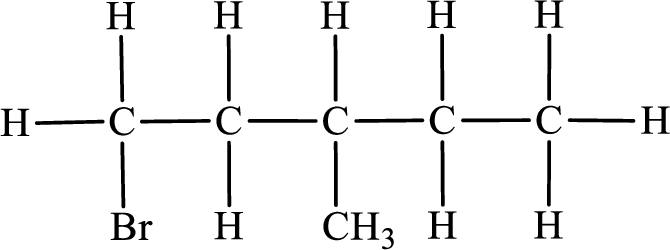
In the given compound, the longest carbon chain is found to contain five carbon atoms. Therefore, the parent alkane name is pentane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
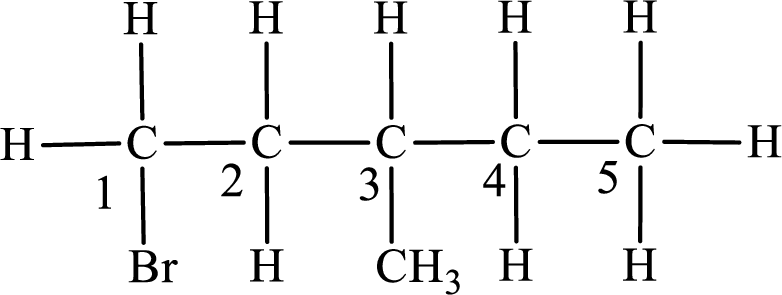
The substituent present in the given compound are a bromine atom and a methyl group. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
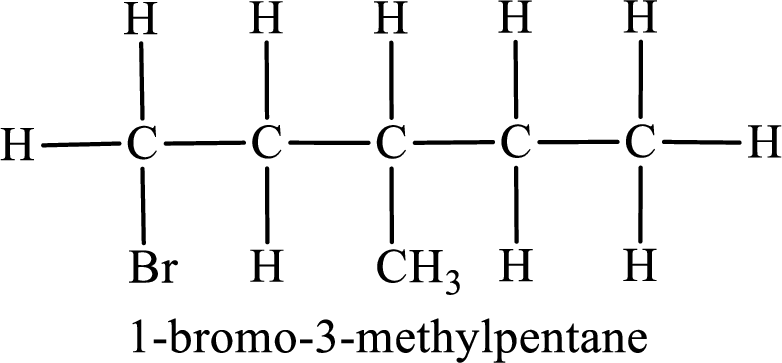
Parent chain is pentane and the substituent present is 1-bromo-3-methyl. Hence, the IUPAC name is given as 1-bromo-3-methylpentane.
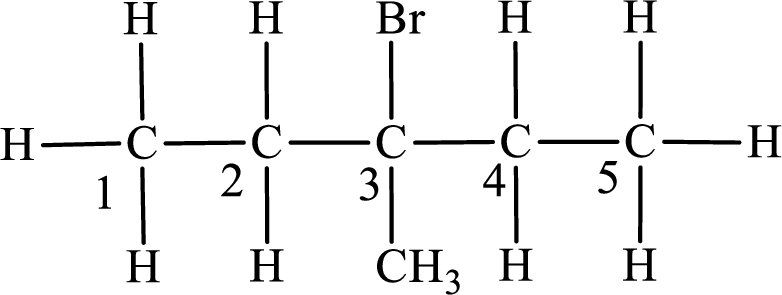
Second monobromo derivative:
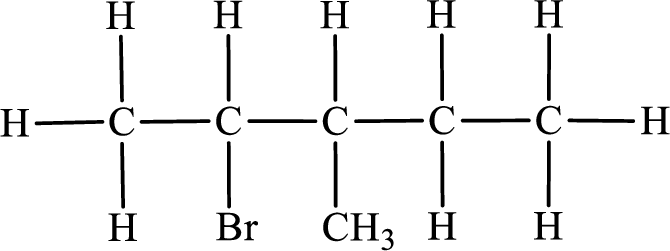
In the given compound, the longest carbon chain is found to contain five carbon atoms. Therefore, the parent alkane name is pentane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
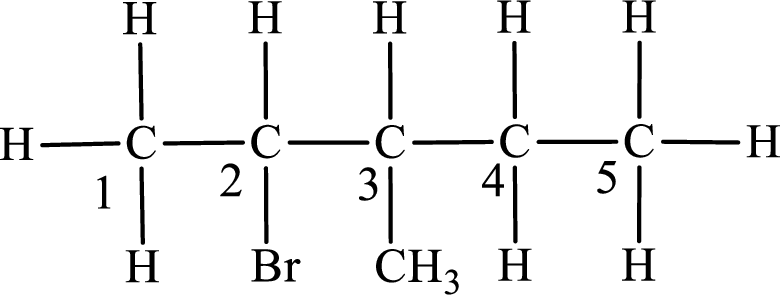
The substituent present in the given compound are a bromine atom and a methyl group. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
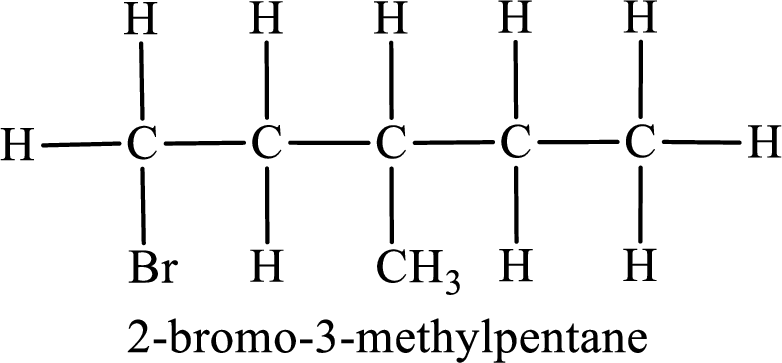
Parent chain is pentane and the substituent present is 2-bromo-3-methyl. Hence, the IUPAC name is given as 2-bromo-3-methylpentane.
Third monobromo derivative:
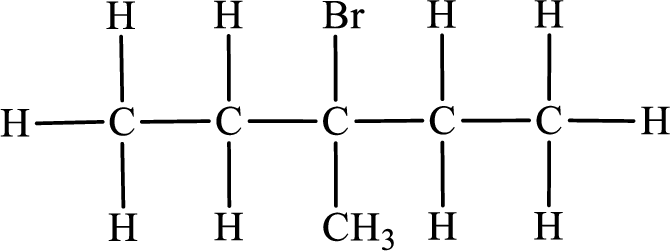
In the given compound, the longest carbon chain is found to contain five carbon atoms. Therefore, the parent alkane name is pentane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
The substituent present in the given compound are a bromine atom and a methyl group. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
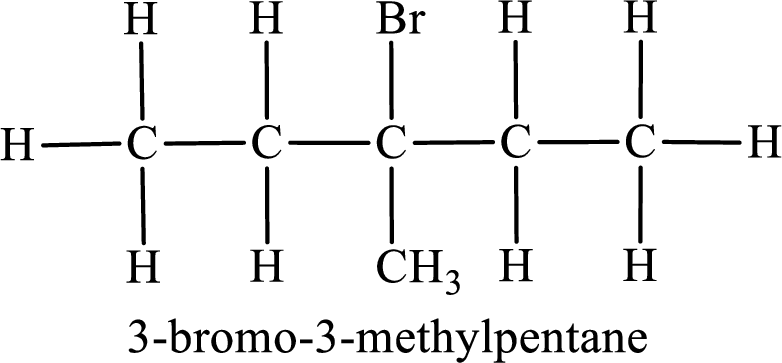
Parent chain is pentane and the substituent present is 3-bromo-3-methyl. Hence, the IUPAC name is given as 3-bromo-3-methylpentane.
Fourth monobromo derivative:
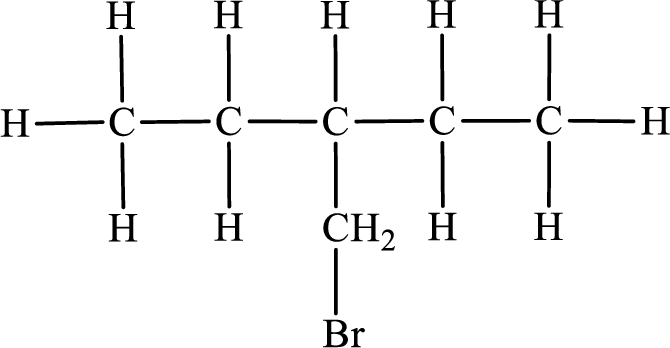
In the given compound, the longest carbon chain is found to contain five carbon atoms. Therefore, the parent alkane name is pentane.
Numbering of carbon atoms has to be done in a way that the substituents present in the longest carbon chain get the least numbering.
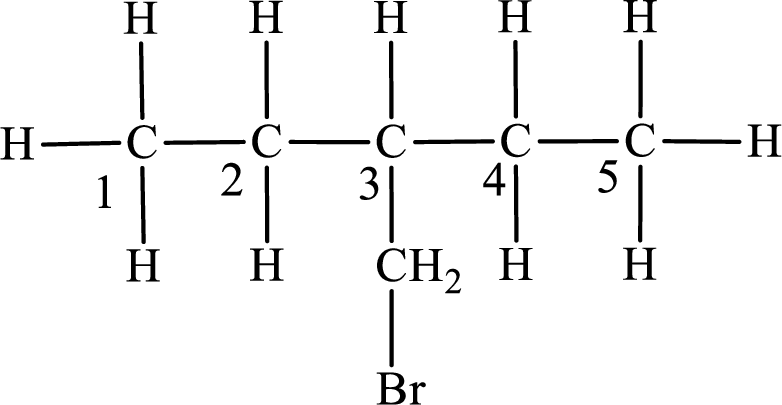
The substituent present in the given compound is a bromomethyl group. Number has to be added before the substituent indicating the carbon in which it is attached. Therefore, IUPAC name is obtained as,
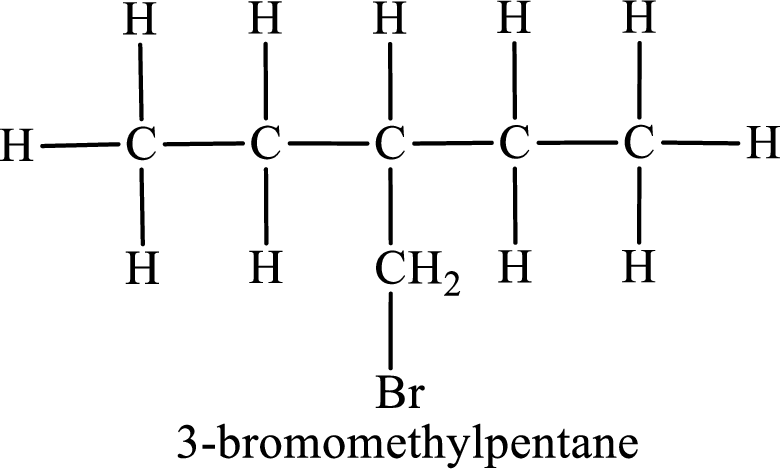
Parent chain is pentane and the substituent present is 3-bromomethyl. Hence, the IUPAC name is given as 3-bromomethylpentane.
Want to see more full solutions like this?
Chapter 10 Solutions
GENERAL,ORGANIC,+BIOCHEMISTRY
- Give the organic product: O A O B Ос ○ D -NH–CH3 + CH3 CH3 NEN C ? A CH3 CH3 NH- CH3 B CH3 CH3 N=N- C CH3 CH3 N=NNH CH3 D CH3 N=N CH3 NHCH3 LNH CHOarrow_forwardFinish the reaction- hand written pleasearrow_forwardGive the organic products: (benzyne) Br ? CH3 + K* :NH, liq NH3 HINT: Two products are formed. Each is a substituted aniline; they are isomers of each other. NH2 II I H₂N. CH3 CH3 III Select one: ○ A. I and II ○ B. I and III O C. I and IV O D. II and III O E. III and IV H₂N CH3 IV CH₂-NH2arrow_forward
- Predict the major products of this organic reaction: HBr (1 equiv) cold ? Some important notes: • Draw the major product, or products, of this reaction in the drawing area below. • You can draw the products in any arrangement you like. • Pay careful attention to the reaction conditions, and only include the major products. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. • Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions. Erase something Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terarrow_forwardQ14. Fill this chart: (please refer to ppt notes/browser to answer these questions) What alcohol is also called wood alcohol? What is the common name of ethanol? Draw the structure of phenol and thiophene? Are bigger chain alcohol like heptanol and octanol are soluble or insoluble in water and explain it ? Are ethers soluble or insoluble in water? What suffix and prefix are used for alcohol while naming alcohol and ether? What the process called when we add water to any alkene to make alcohol? Q16. Draw the diagram of following aromatic compound (practice from previous module) Aniline Phenol Benzoic acid Methyl benzoate Q17. a. Write the oxidation reactions for the 2 propanol. b. Write the oxidation reaction of the ethanol.arrow_forwardQuestion 11 of 18 (1 point) Question Attempt: 3 of How many signals do you expect in the 'H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. 1 For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box…arrow_forward
- Organic Chemistry Esterification reactions 1. Write the steps to prepare ester. 2. Write complete reaction of ethanol and acetic acid to make ester. 3. What does ester smell like? What are the uses of ester. 4. What the role of sulfuric acid in the esterification reactionarrow_forward11. Complete the following esterification reaction with names of all the reactants and products under. Hint: Remove the water and end up with ester R-C-OH + ROH R-C-OR + H₂O A carboxylic acid An alcohol An ester Water BYJU'S H-C-C O-H Нин C-C-C-H HAAA H O-C-C-C-H AAA Ethanoic acid Propanol Water Propyl ethanoate By com CH3COOH + CH3CH2CH2CH₂CH₂OH → Practice for alcohols aldehydes and ketones: 12. Draw the structures from the following names mixed of alcohol/aldehyde and ketone: a. 4-methyl cyclohexanone b. 3-methyl-2-pentenal c. 2,3-dimethylcyclohexanone d. 1,3propanediol or Propane 1,3 diol 13. Write systematic names for the following compounds identify functional group: a. b. (CH3)2CH-C OH c) CH(CH₂)-- OH -,-,arrow_forwardmay you please show all steps! i am having a hard time understanding and applying in this format, thank you!arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





