
Concept explainers
(a)
Interpretation:
Lewis structure for the
Concept introduction:
Lewis structure is a convenient way to convey information such as which atoms are bonded to each other by which type of bond. Only valence electrons take part to draw Lewis structure.
Valence electrons that participate in bond formation are called bonding electron pairs whereas electrons that do not take part in bonding and that remain as non-bonding electrons are termed as lone pair of electrons.
Lewis structure is drawn from the total valence electron count of each atom in the molecule. Skeleton structure for the given molecule is drawn with atoms bonded with a single bond, the central atom is always less electronegative atom. Electrons that participate in forming the bond are bonding electrons. Remaining electrons are distributed first to outer atoms such that each atom completes its octet other than hydrogen.
Answer to Problem 1.44P
Lewis structure for
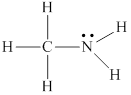
Explanation of Solution
The given molecule
Thus the skeleton structure for
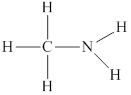
Remaining two valence electrons are contributed as lone pair of electrons to the outer atom which is nitrogen.
Hence the complete Lewis structure for

Lewis structure for
(b)
Interpretation:
Lewis structure for
Concept introduction:
Lewis structure is drawn from the total valence electron count of each atom in the molecule. Skeleton structure for the given molecule is drawn with atoms bonded with a single bond, the central atom is always less electronegative atom. Electrons that participate in forming the bond are bonding electrons. Remaining electrons are distributed first to outer atoms such that each atom complete its octet other than hydrogen.
Answer to Problem 1.44P
Lewis structure for
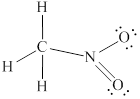
Explanation of Solution
In
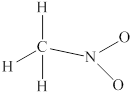
Remaining
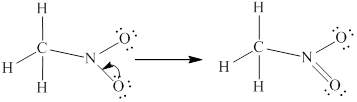
Lewis structure for
(c)
Interpretation:
Lewis structure for
Concept introduction:
Lewis structure is drawn from the total valence electron count of each atom in the molecule. Valence electrons participate in bond formation are called bonding electron pairs whereas electrons that do not take part in bonding and that remain as non-bonding electrons are termed as lone pair of electrons. Skeleton structure for the given molecule is drawn with atoms bonded with a single bond, the central atom is always less electronegative atom.
Lone pair of electrons contributed to outer atoms to complete octet except hydrogen.
Answer to Problem 1.44P
The Lewis structure for
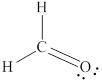
Explanation of Solution
Carbon is the central atom in
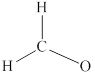
Remaining six electrons are contributed as lone pairs to the outer atom oxygen but one electron is involved in bonding with complete octet of oxygen atom.
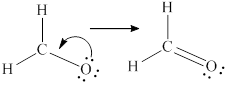
Lewis structure is drawn from the total valence electron count.
(d)
Interpretation:
Lewis structure for
Concept introduction:
Lewis structure is drawn from the total valence electron count of each atom in the molecule. Skeleton structure for the given molecule is drawn with atoms bonded with a single bond, the central atom is always less electronegative atom. Electrons that participate in forming the bond are bonding electrons. Remaining electrons are distributed first to outer atoms such that each atom completes its octet other than hydrogen.
Answer to Problem 1.44P
Lewis structure for
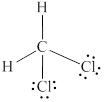
Explanation of Solution
In
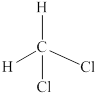
Eight electrons from total
Therefore the Lewis structure for

Lewis structure is drawn from total valence electron count.
(e)
Interpretation:
Lewis structure for
Concept introduction:
Lewis structure is drawn from the total valence electron count of each atom in the molecule. Valence electrons that participate in bond formation are called bonding electron pairs whereas electrons that do not take part in bonding and that remain as non-bonding electrons are termed as lone pair of electrons. Skeleton structure for the given molecule is drawn with atoms bonded with a single bond, the central atom is always less electronegative atom.
Lone pair of electrons contributed to outer atoms to complete octet except hydrogen.
Answer to Problem 1.44P
Lewis structure for
![]()
Explanation of Solution
The given molecule
![]()
Remaining
Hence the Lewis structure for
![]()
Lewis structure is drawn from total valence electron count.
Want to see more full solutions like this?
Chapter 1 Solutions
ORG CHEM W/ EBOOK & SW5 + STUDY GUIDE
- In an experiment, 74.3 g of metallic copper was heated to 100.0°C and then quickly dropped into 200.0 mL of water in a calorimeter. The heat capacity of the calorimeter with the water was 875 J/°C. The initial temperature of the calorimeter was 27.5°C, and the final temperature after addition of the metal was 29.8°C. What is the value of the molar heat capacity of copper?arrow_forwardThe Haber-Bosch process permits the direct conversion of molecular nitrogen to ammonia, which can be used in large-scale fertilizer production. Given the balanced Haber-Bosch reaction and using the bond energies in the table below, estimate the enthalpy change associated with the reaction. N2(g) + 3H2(g) → 2NH3(g) Bond N=N N = N Energy (kJ/mol) 941 418 N-N H-H N-H 163 435 388arrow_forwardBenzoic acid is used to determine the heat capacity of bomb calorimeters because it can be obtained in pure form and its energy of combustion is known very accurately (−26.43 kJ/g). Determine the heat capacity of a calorimeter that had a temperature increase of 9.199°C when 3.500 g of benzoic acid was used.arrow_forward
- Given the standard enthalpies of formation for the following substances, determine the reaction enthalpy for the following reaction. 2N2H4(g) + 2NO2(g) → 3N2(g) + 4H2O(g) AHrxn ? kJ Substance AH in kJ/mol N2H4(g) +95.4 NO2(g) +33.1 H2O(g) -241.8arrow_forwardIf 7.3 kJ of energy are required to change the temperature of water from 5.0 to 70.0, what was the volume of water? (cs = 4.184 J/(g ⋅ ), d = 1.00 g/mL)arrow_forwardBALANCE CHEMICAL REACTIONarrow_forward
- Predict the product(s) of the following reactions. If no reaction, write "NR". a) Cl₂ FeCl3 e) HNO3 H2SO4 b) NO2 CI. HNO3 f) Br Br2 OH H2SO4 HO3S. FeBr3 c) Cl2 g) FeCl3 F d) O₂N Br2 FeBr3 O₂N OH HNO3 CH3 H2SO4arrow_forwardulating the pH salt solution Calculate the pH at 25 °C of a 0.75M solution of anilinium chloride (C6H5NH3C1). Note that aniline (C6H5NH2) is a weak base with a pK of 4.87. Round your answer to 1 decimal place. pH = ☐ ☑ ⑤ ? olo 18 Ararrow_forwardI apologize, but the app is not allowing me to post the other 4 pictures of the thermodynamics chart. But I believe the values are universal. Please help!arrow_forward
- Calculating the pH of a salt solution Calculate the pH at 25 °C of a 0.29M solution of potassium butanoate (KC3H,CO2). Note that butanoic acid (HC3H,CO2) is a weak acid with a pKa of 4.82. Round your answer to 1 decimal place. pH = -0 Х olo 18 Ararrow_forward: At a certain temperature, the equilibrium constant K for the following reaction is 1.58 × 10-12 N2(g) + O2(g) = 2 NO(g) Use this information to complete the following table. Suppose a 38. L reaction vessel is filled with 0.93 mol of N2 and 0.93 mol of O2. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little N2 and O2. There will be very little NO. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 NO(g) N2(9)+02(9) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 3 N2(9)+302(g) 6 NO(g) Neither of the above is true. K = ☐ K = ☐ ☐ X10 Х D ? 000 18 Ar Barrow_forwardwhen performing the reaction that involves 2 equivalents of 3-(diethylamino)-phenol and Phthalic anhydride with sulfuric acid and water react to form rhodamine b where the Phthalic anhydride cleaves in acid and how does Excessive Washing (w/ Base) & Subsequent Resonance Structure get affectedarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
