
Concept explainers
(a)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charges on oxygen atom and chlorine atom are
Explanation of Solution
The given structure is shown in figure 1.
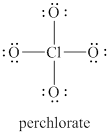
Figure 1
The formal charge on atom is calculated by the formula given below as,
The valence electrons of oxygen atoms in given species are
Thus, the formal charge on each oxygen atom is
The valence electrons of chlorine atom in given species are
Thus, the formal charge on chlorine atom is
The total charge on the given species is calculated as follows.
Hence, total charge on the species is
The formal charges on oxygen atom and chlorine atom are
(b)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charges on oxygen atom, nitrogen atom and carbon atom are
Explanation of Solution
The given structure is shown in figure 2.
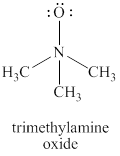
Figure 2
The formal charge on atom is calculated by the formula given below as,
The valence electrons of oxygen atoms in given species are
Thus, the formal charge on each oxygen atom is
The valence electrons of nitrogen atom in given species are
Thus, the formal charge on nitrogen atom is
The valence electrons of carbon atom in given species are
Thus, the formal charge on carbon atom is
The total charge on the given species is calculated as follows.
Hence, total charge on the species is
The formal charges on oxygen atom, nitrogen atom and carbon atom are
(c)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charges on left oxygen atom, right oxygen atom and central oxygen atom are
Explanation of Solution
The given structure is shown in figure 3.
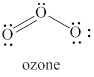
Figure 3
The formal charge on atom is calculated by the formula given below as,
The valence electrons of left oxygen atom in given species are
Thus, the formal charge on each oxygen atom is
The valence electrons of right oxygen atom in given species are
Thus, the formal charge on each oxygen atom is
The valence electrons of central oxygen atom in given species are
Thus, the formal charge on each oxygen atom is
The total charge on the given species is calculated as follows.
Hence, total charge on the species is
The formal charges on left oxygen atom, right oxygen atom and central oxygen atom are
(d)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charge on carbon atom is
Explanation of Solution
The given structure is shown in figure 4.
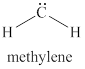
Figure 4
The formal charge on atom is calculated by the formula given below as,
The valence electrons of carbon atom in given species are
Thus, the formal charge on carbon atom is
Hence, total charge on the species is also
The formal charge on carbon atom is
(e)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charge on carbon atom and carbon radical is
Explanation of Solution
The given structure is shown in figure 5.
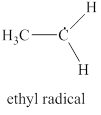
Figure 5
The formal charge on atom is calculated by the formula given below as,
The valence electrons of carbon atom in given species are
Thus, the formal charge on carbon atom is
The valence electrons of carbon radical in given species are
Thus, the formal charge on carbon radical is
Hence, total charge on the species is also
The formal charge on carbon atom and carbon radical is
(f)
Interpretation:
The formal charge on each atom and the net charge in the given species are to be stated.
Concept introduction:
Chemical compounds contain two types of bonds. These are known as ionic and covalent bonds. In ionic bonds, the ions are held by the electrostatic interaction between them. In covalent bonds, the atoms are held together by the sharing of electrons. The formal charge is the charge on the constituent atoms in a molecule. It is calculated by using valence electrons of the atom.
Answer to Problem 1.25AP
The formal charge on oxygen atom and chlorine atom is
Explanation of Solution
The given structure is shown in figure 6.
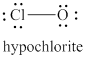
Figure 6
The formal charge on atom is calculated by the formula given below as,
The valence electrons of oxygen atoms in given species are
Thus, the formal charge on each oxygen atom is
The valence electrons of chlorine atom in given species are
Thus, the formal charge on chlorine atom is
The total charge on the given species is calculated as follows.
Hence, total charge on the species is
The formal charge on oxygen atom and chlorine atom is
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry Study Guide and Solutions
- For each reaction below, decide if the first stable organic product that forms in solution will create a new CC bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. དྲ。 ✗MgBr ? O CI Will the first product that forms in this reaction create a new C-C bond? Yes No • ? Will the first product that forms in this reaction create a new CC bond? Yes No × : ☐ Xarrow_forwardPredict the major products of this organic reaction: OH NaBH4 H ? CH3OH Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. ☐ : Sarrow_forwardPredict the major products of this organic reaction: 1. LIAIHA 2. H₂O ? Note: be sure you use dash and wedge bonds when necessary, for example to distinguish between major products with different stereochemistry. Click and drag to start drawing a structure. X : ☐arrow_forward
- For each reaction below, decide if the first stable organic product that forms in solution will create a new C - C bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. NH2 tu ? ? OH Will the first product that forms in this reaction create a new CC bond? Yes No Will the first product that forms in this reaction create a new CC bond? Yes No C $ ©arrow_forwardAs the lead product manager at OrganometALEKS Industries, you are trying to decide if the following reaction will make a molecule with a new C-C bond as its major product: 1. MgCl ? 2. H₂O* If this reaction will work, draw the major organic product or products you would expect in the drawing area below. If there's more than one major product, you can draw them in any arrangement you like. Be sure you use wedge and dash bonds if necessary, for example to distinguish between major products with different stereochemistry. If the major products of this reaction won't have a new CC bond, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. This reaction will not make a product with a new CC bond. G marrow_forwardIncluding activity coefficients, find [Hg22+] in saturated Hg2Br2 in 0.00100 M NH4 Ksp Hg2Br2 = 5.6×10-23.arrow_forward
- give example for the following(by equation) a. Converting a water insoluble compound to a soluble one. b. Diazotization reaction form diazonium salt c. coupling reaction of a diazonium salt d. indacator properties of MO e. Diazotization ( diazonium salt of bromobenzene)arrow_forward2-Propanone and ethyllithium are mixed and subsequently acid hydrolyzed. Draw and name the structures of the products.arrow_forward(Methanesulfinyl)methane is reacted with NaH, and then with acetophenone. Draw and name the structures of the products.arrow_forward
- 3-Oxo-butanenitrile and (E)-2-butenal are mixed with sodium ethoxide in ethanol. Draw and name the structures of the products.arrow_forwardWhat is the reason of the following(use equations if possible) a.) In MO preperation through diazotization: Addition of sodium nitrite in acidfied solution in order to form diazonium salt b.) in MO experiment: addition of sodium hydroxide solution in the last step to isolate the product MO. What is the color of MO at low pH c.) In MO experiment: addition of sodium hydroxide solution in the last step to isolate the product MO. What is the color of MO at pH 4.5 d.) Avoiding not cooling down the reaction mixture when preparing the diazonium salt e.) Cbvcarrow_forwardA 0.552-g sample of an unknown acid was dissolved in water to a total volume of 20.0 mL. This sample was titrated with 0.1103 M KOH. The equivalence point occurred at 29.42 mL base added. The pH of the solution at 10.0 mL base added was 3.72. Determine the molar mass of the acid. Determine the Ka of the acid.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





