The palladium acetate complex, [Pd(OAc)2], is useful for performing catalytic reactions such as the Heck reaction and the Cope reaction. (a) Briefly describe the Cope reaction. Illustrate your answer with suitable chemical equations. (b) Comment on two features of this complex that would make it an effective catalyst.
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
The palladium acetate complex, [Pd(OAc)2], is useful for performing catalytic reactions such as the Heck reaction and the Cope reaction.
(a) Briefly describe the Cope reaction. Illustrate your answer with suitable chemical equations.
(b) Comment on two features of this complex that would make it an effective catalyst.
(a) Cope reaction: The cope reaction is the reaction in which an amine is oxidized to an intermediate called an N-oxide which when heated acts as a base in an intermolecular elimination.
Step 1 : Oxidation of a 30 amine to an N-oxide. The reaction mechanism step is shown below,
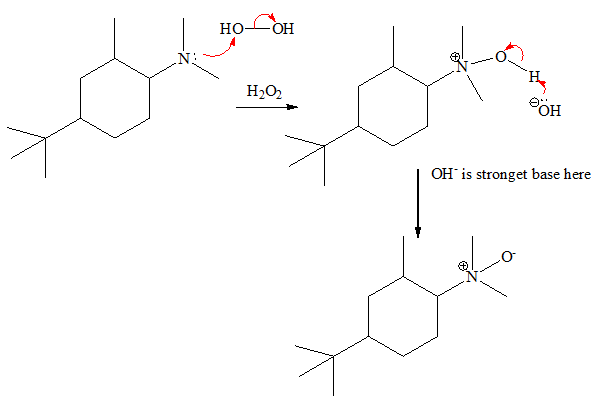
Step 2 : Heat induced elimination
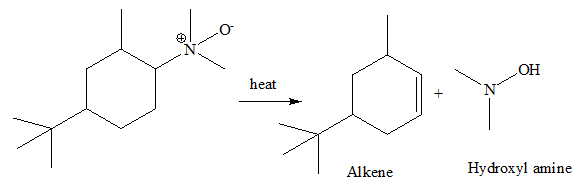
The C=C and O-H bonds are formed whereas the C-H and C-N bonds are broken in above step.
Step by step
Solved in 4 steps with 4 images









