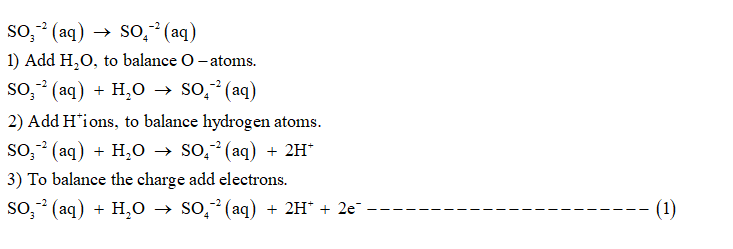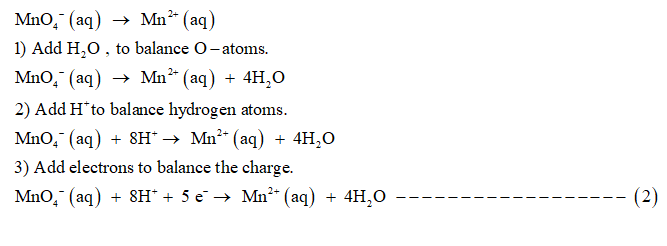Chemistry 9th Edition
ISBN: 9781133611097
Author: Steven S. Zumdahl
Publisher: Steven S. Zumdahl
1 Chemical Foundations 2 Atoms, Molecules, And Ions 3 Stoichiometry 4 Types Of Chemical Reactions And Solution Stoichiometry 5 Gases 6 Thermochemistry 7 Atomic Structure And Periodicity 8 Bonding: General Concepts 9 Covalent Bonding: Orbitals 10 Liquids And Solids 11 Properties Of Solutions 12 Chemical Kinetics 13 Chemical Equilibrium 14 Acids And Bases 15 Acid-base Equilibria 16 Solubility And Complex Ion Equilibria 17 Spontaneity, Entropy, And Free Energy 18 Electrochemistry 19 The Nucleus: A Chemist's View 20 The Representative Elements 21 Transition Metals And Coordination Chemistry 22 Organic And Biological Molecules Chapter4: Types Of Chemical Reactions And Solution Stoichiometry
Chapter Questions Section: Chapter Questions
Problem 1RQ: The (aq) designation listed after a solute indicates the process of hydration. Using KBr(aq) and... Problem 2RQ: Characterize strong electrolytes versus weak electrolytes versus nonelectrolytes. Give examples of... Problem 3RQ: Distinguish between the terms slightly soluble and weak. electrolyte. Problem 4RQ: Molarity is a conversion factor relating moles of solute in solution to the volume of the solution.... Problem 5RQ: What is a dilution? What stays constant in a dilution? Explain why the equation M1 V1 = M2 V2 works... Problem 6RQ: When the following beakers are mixed, draw a molecular-level representation of the product mixture... Problem 7RQ: Differentiate between the formula equation, the complete ionic equation, and the net ionic equation.... Problem 8RQ: What is an acid-base reaction? Strong bases are soluble ionic compounds that contain the hydroxide... Problem 9RQ: Define the terms oxidation, reduction, oxidizing agent, and reducing agent. Given a chemical... Problem 10RQ Problem 1ALQ: Assume you have a highly magnified view of a solution of HCl that allows you to see the HCl. Draw... Problem 2ALQ: You have a solution of table salt in water. What happens to the salt concentration (increases,... Problem 3ALQ: You have a sugar solution (solution A) with concentration x. You pour one-fourth of this solution... Problem 4ALQ: You add an aqueous solution of lead nitrate to an aqueous solution of potassium iodide. Draw highly... Problem 5ALQ: Order the following molecules from lowest to highest oxidation state of the nitrogen atom: HNO3,... Problem 6ALQ: Why is it that when something gains electrons, it is said to be reduced? What is being reduced? Problem 7ALQ: Consider separate aqueous solutions of HCl and H2SO4 with the same molar concentrations. You wish to... Problem 8ALQ Problem 9ALQ Problem 10ALQ: The exposed electrodes of a light bulb are placed in a solution of H2SO4 in an electrical circuit... Problem 13Q: Differentiate between what happens when the following are added to water. a. polar solute versus... Problem 14Q: A typical solution used in general chemistry laboratories is 3.0 M HCl. Describe, in detail, the... Problem 15Q Problem 16Q: A student wants to prepare 1.00 L of a 1.00-M solution of NaOH (molar mass = 40.00 g/mol). If solid... Problem 17Q: List the formulas of three soluble bromide salts and three insoluble bromide salts. Do the same... Problem 18Q: When 1.0 mole of solid lead nitrate is added to 2.0 moles of aqueous potassium iodide, a yellow... Problem 19Q: What is an acid and what is a base? An acid-base reaction is sometimes called a proton-transfer... Problem 20Q: A student had 1.00 L of a 1.00-M acid solution. Much to the surprise of the student, it took 2.00 L... Problem 21Q: Differentiate between the following terms. a. species reduced versus the reducing agent b. species... Problem 22Q: How does one balance redox reactions by the oxidation states method? Problem 23E Problem 24E: Match each name below with the following microscopic pictures of that compound in aqueous solution.... Problem 25E: Calcium chloride is a strong electrolyte and is used to salt streets in the winter to melt ice and... Problem 26E: Commercial cold packs and hot packs are available for treating athletic injuries. Both types contain... Problem 27E: Calculate the molarity of each of these solutions. a. A 5.623-g sample of NaHCO3 is dissolved in... Problem 28E: A solution of ethanol (C2H5OH) in water is prepared by dissolving 75.0 mL of ethanol (density = 0.79... Problem 29E: Calculate the concentration of all ions present in each of the following solutions of strong... Problem 30E Problem 31E Problem 32E Problem 33E Problem 34E: If 10. g of AgNO3 is available, what volume of 0.25 M AgNO3 solution can be prepared? Problem 37E: A solution is prepared by dissolving 10.8 g ammonium sulfate in enough water to make 100.0 mL of... Problem 38E: A solution was prepared by mixing 50.00 mL of 0.100 M HNO3 and 100.00 mL of 0.200 M HNO3. Calculate... Problem 39E: Calculate the sodium ion concentration when 70.0 mL of 3.0 M sodium carbonate is added to 30.0 mL of... Problem 40E: Suppose 50.0 mL of 0.250 M CoCl2 solution is added to 25.0 mL of 0.350 M NiCl2 solution. Calculate... Problem 41E Problem 42E: A stock solution containing Mn2+ ions was prepaned by dissolving 1.584 g pure manganese metal in... Problem 43E: On the basis of the general solubility rules given in Table 6-1, predict which of the following... Problem 44E: On the basis of the general solubility rules given in Table 6-1, predict which of the following... Problem 45E: When the following solutions are mixed together, what precipitate (if any) will form? a. FeSO4(aq) +... Problem 46E: When the following solutions are mixed together, what precipitate (if any) will form? a.... Problem 47E: For the reactions in Exercise 47, write the balanced formula equation, complete ionic equation, and... Problem 48E: For the reactions in Exercise 48, write the balanced formula equation, complete ionic equation, and... Problem 49E: Write the balanced formula and net ionic equation for the reaction that occurs when the contents of... Problem 50E: Give an example how each of the following insoluble ionic compounds could be produced using a... Problem 51E: Write net ionic equations for the reaction, if any, that occurs when aqueous solutions of the... Problem 52E: Write net ionic equations for the reaction, if any, that occurs when aqueous solutions of the... Problem 53E: Separate samples of a solution of an unknown soluble ionic compound are treated with KCl, Na2SO4,... Problem 54E: A sample may contain any or all of the following ions: Hg22+, Ba2+, and Mn2+. a. No precipitate... Problem 55E: What mass of Na2CrO4 is required to precipitate all of the silver ions from 75.0 mL of a 0.100-M... Problem 56E: What volume of 0.100 M Na3PO4 is required to precipitate all the lead(II) ions from 150.0 mL of... Problem 57E: What mass of iron(III) hydroxide precipitate can be produced by reacting 75.0 mL of 0.105 M... Problem 60E: What mass of silver chloride can be prepared by the reaction of 100.0 mL of 0.20 M silver nitrate... Problem 61E: A 100.0-mL aliquot of 0.200 M aqueous potassium hydroxide is mixed with 100.0 mL of 0.200 M aqueous... Problem 63E: A 1.42-g sample of a pure compound, with formula M2SO4, was dissolved in water and treated with an... Problem 64E: You are given a 1.50-g mixture of sodium nitrate and sodium chloride. You dissolve this mixture into... Problem 65E: Write the balanced formula, complete ionic, and net ionic equations for each of the following... Problem 66E: Write the balanced formula, complete ionic, and net ionic equations for each of the following... Problem 67E: Write the balanced formula equation for the acid-base reactions that occur when the following are... Problem 68E Problem 69E: What volume of each of the following acids will react completely with 50.00 mL of 0.200 M NaOH? a.... Problem 70E Problem 71E: Hydrochloric acid (75.0 mL of 0.250 M) is added to 225.0 mL of 0.0550 M Ba(OH)2 solution. What is... Problem 72E: A student mixes four reagents together, thinking that the solutions will neutralize each other. The... Problem 73E: A 25.00-mL sample of hydrochloric acid solution requires 24.16 mL of 0.106 M sodium hydroxide for... Problem 74E: A 10.00-mL sample of vinegar, an aqueous solution of acetic acid (HC2H3O2), is titrated with 0.5062... Problem 75E: What volume of 0.0200 M calcium hydroxide is required to neutralize 35.00 mL of 0.0500 M nitric... Problem 76E: A 30.0-mL sample of an unknown strong base is neutralized after the addition of 12.0 mL of a 0.150 M... Problem 77E: A student titrates an unknown amount of potassium hydrogen phthalate (KHC8H4O4, often abbreviated... Problem 78E: The concentration of a certain sodium hydroxide solution was determined by using the solution to... Problem 79E: Assign oxidation states for all atoms in each of the following compounds. a. KMnO4 b. NiO2 c.... Problem 81E: Assign the oxidation state for nitrogen in each of the following. a. Li3N b. NH3 c. N2H4 d. NO e.N2O... Problem 82E: Assign oxidatioo numbers to all the atoms in each of the following. a. SrCr2O7 b. CuCl2 c. O2 d.... Problem 83E: Specify which of the following are oxidationreduction reactions, and identify the oxidizing agent,... Problem 84E: Specify which of the following equations represent oxidationreduction reactions, and indicate the... Problem 85E: Consider the reaction between sodium metal and fluorine (F2) gas to form sodium fluoride. Using... Problem 86E: Consider the reaction between oxygen (O2) gas and magnesium metal to form magnesium oxide. Using... Problem 87E: Balance each of the following oxidationreduction reactions by using the oxidation states method.... Problem 88E: Balance each of the following oxidationreduction reactions by using the oxidation states method.... Problem 89AE: You wish to prepare 1 L of a 0.02-M potassium iodate solution. You require that the final... Problem 90AE: The figures below are molecular-level representations of four aqueous solutions of the same solute.... Problem 91AE Problem 92AE Problem 93AE: Using the general solubility rules given in Table 6-1. name three reagents that would form... Problem 94AE: Consider a 1.50-g mixture of magnesium nitrate and magnesium chloride. After dissolving this mixture... Problem 95AE: A 1.00-g sample of an alkaline earth metal chloride is treated with excess silver nitrate. All of... Problem 96AE: A mixture contains only NaCl and Al2(SO4)3. A 1.45-g sample of the mixture is dissolved in water and... Problem 98AE: A mixture contains only NaCl and Fe(NO3)3. A 0.456-g sample of the mixture is dissolved in water,... Problem 99AE: A student added 50.0 mL of an NaOH solution to 100.0 mL of 0.400 M HCl. The solution was then... Problem 100AE: Some of the substances commonly used in stomach antacids are MgO, Mg(OH)2, and Al(OH)3. a. Write a... Problem 101AE: Acetylsalicylic acid is the active ingredient in aspirin. It took 35.17 mL of 0.5065 M sodium... Problem 102AE: When hydrochloric acid reacts with magnesium metal, hydrogen gas and aqueous magnesium chloride are... Problem 103AE: A 2.20-g sample of an unknown acid (empirical formula = C3H4O3) is dissolved in 1.0 L of water. A... Problem 104AE: Carminic acid, a naturally occurring red pigment extracted from the cochineal insect, contains only... Problem 105AE: Chlorisondamine chloride (C14H20Cl6N2) is a drug used in the treatment of hypertension. A 1.28-g... Problem 106AE: Saccharin (C7H5NO3S) is sometimes dispensed in tablet form. Ten tablets with a total mass of 0.5894... Problem 107AE: Douglasite is a mineral with the formula 2KC1 FeCl2 2H2O. Calculate the mass percent of douglasite... Problem 108AE: Many oxidationreduction reactions can be balanced by inspection. Try to balance the following... Problem 109AE: The blood alcohol (C2H5OH) level can be determined by titrating a sample of blood plasma with an... Problem 110CWP: Calculate the concentration of all ions present when 0.160 g of MgCl2 is dissolved in 100.0 mL of... Problem 111CWP: A solution is prepared by dissolving 0.6706 g oxalic acid (H2C2O4) in enough water to make 100.0 mL... Problem 112CWP: For the following chemical reactions, determine the precipitate produced when the two reactants... Problem 113CWP: What volume of 0.100 M NaOH is required to precipitate all of the nickel(II) ions from 150.0 mL of a... Problem 114CWP Problem 115CWP: A 450.0-mL sample of a 0.257-M solution of silver nitrate is mixed with 400.0 mL of 0.200 M calcium... Problem 116CWP: The zinc in a 1.343-g sample of a foot powder was precipitated as ZnNH4PO4. Strong heating of the... Problem 117CWP: A 50.00-mL sample of aqueous Ca(OH)2 requires 34.66 mL of a 0.944-M nitric acid for neutralization.... Problem 118CWP: When organic compounds containing sulfur are burned, sulfurdioxide is produced. The amount of SO2... Problem 119CWP: Assign the oxidation state for the element Listed in each of the following compounds: Oxidation... Problem 120CP: A 10.00-g sample consisting of a mixture of sodium chloride and potassium sulfate is dissolved in... Problem 121CP: The units of parts per million (ppm) and parts per billion (ppb) are commonly used by environmental... Problem 122CP: In the spectroscopic analysis of many substances, a series of standard solutions of known... Problem 123CP: In most of its ionic compounds, cobalt is either Co(II) or Co(III). One such compound, containing... Problem 124CP: Polychlorinated biphenyls (PCBs) have been used extensively as dielectric materials in electrical... Problem 125CP: Consider the reaction of 19.0 g of zinc with excess silver nitrite to produce silver metal and zinc... Problem 126CP: A mixture contains only sodium chloride and potassium chloride. A 0.1586-g sample of the mixture was... Problem 127CP Problem 128CP: Zinc and magnesium metal each react with hydrochloric acid according to the following equations:... Problem 129CP: You made 100.0 mL of a lead(II) nitrate solution for lab but forgot to cap it. The next lab session... Problem 130CP: Consider reacting copper(II) sulfate with iron. Two possible reactions can occur, as represented by... Problem 131CP: Consider an experiment in which two burets, Y and Z, are simultaneously draining into a beaker that... Problem 132CP: Complete and balance each acid-base reaction. a. H3PO4(aq) + NaOH(aq) Contains three acidic... Problem 133CP: What volume of 0.0521 M Ba(OH)2 is required to neutralize exactly 14.20 mL of 0.141 M H3PO4?... Problem 134CP: A 10.00-mL sample of sulfuric acid from an automobile battery requires 35.08 mL of 2.12 M sodium... Problem 135CP: A 0.500-L sample of H2SO4 solution was analyzed by taking a 100.0-mL aliquot and adding 50.0 mL of... Problem 136CP: A 6.50-g sample of a diprotic acid requires 137.5 mL of a 0.750 M NaOH solution for complete... Problem 137CP: Citric acid, which can be obtained from lemon juice, has the molecular formula C6H8O7. A 0.250-g... Problem 138CP Problem 139CP: It took 25.06 0.05 mL of a sodium hydroxide solution to titrate a 0.4016-g sample of KHP (see... Problem 140IP Problem 141IP: In a 1-L beaker, 203 mL of 0.307 M ammonium chromate was mixed with 137 mL of 0.269 M chromium(III)... Problem 142IP Problem 143IP: The unknown acid H2X can be neutralized completely by OH according to the following (unbalanced)... Problem 144MP: Three students were asked to find the identity of the metal in a particular sulfate salt. They... Problem 145MP: You have two 500.0-mL aqueous solutions. Solution A is a solution of a metal nitrate that is 8.246%... Problem 88E: Balance each of the following oxidationreduction reactions by using the oxidation states method....
Related questions
I. Balance the ff. redox reactions. Balance them using water in acidic medium if necessary.
SO3 −2 (aq) + MnO−4 (aq) → SO4 −2 (aq) + Mn2+ (aq)
Definition Definition Chemical reactions involving both oxidation and reduction processes. During a redox reaction, electron transfer takes place in such a way that one chemical compound gets reduced and the other gets oxidized.
Expert Solution
To balance the redox reactions in acidic medium, consider both the oxidation and reduction half cell reactions.
1) Add H+ ions to balance hydrogen atoms.
2) To balance O-atoms , add H2 O molecules.
3) To balance the charge add electrons.
Given unbalanced chemical equation is:
SO3 −2 (aq) + MnO4 − (aq) → SO4 −2 (aq) + Mn2+ (aq)
Oxidation half reaction is:
Reduction half reaction is:
Step by step
Solved in 5 steps with 4 images
UNLOCK THE REST