
Concept explainers
(a)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given molecule is
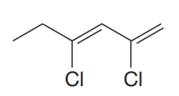
In the given molecule, the longest carbon chain containing all
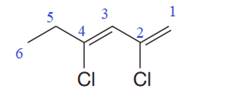
At carbon atoms C2 and C4 of the root, two chlorine atoms are present. Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(b)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is
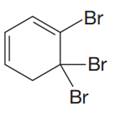
In the given molecule, the largest carbon ring containing all two
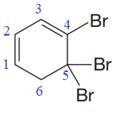
Three bromine atoms are present as substituents at carbon atoms C4, C5, and C5 to the root. The prefix ‘tri’ must be used to indicate the number of substituents present to the root. Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(c)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given molecule is
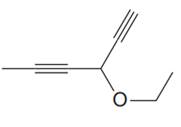
In the given molecule, the longest carbon chain containing all two
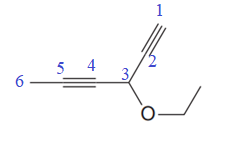
One methoxy substituent is attached to the C3 carbon atom of the root. Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(d)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given molecule is
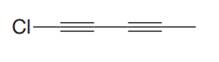
In the given molecule, the longest carbon chain containing all
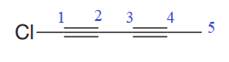
At carbon atom C1of the root, one chlorine atom is present.
Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(e)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is
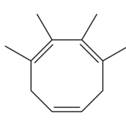
In the given molecule, the largest carbon ring containing all three
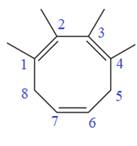
Four methyl substituents are attached at C1, C2, C3, and C4 carbon atoms of the root. The prefix ‘tetra’ should be used to indicate four methyl substituents.
Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(f)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given molecule is
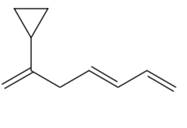
In the given molecule, the longest carbon chain containing all
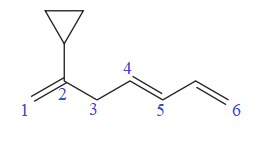
At carbon atom C2 of the root, one cyclopropyl group is attached.
Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
(g)
Interpretation:
The IUPAC name for the given compound is to be determined.
Concept introduction:
For a molecule that has more than one double bond or triple bond, the IUPAC name must indicate the number of double bonds or triple bonds present as well as their locations. To name the molecule with multiple double/triple bonds, establish the root as the longest carbon chain or the largest carbon ring that contains the greatest number of entire
Answer to Problem B.32P
The IUPAC name for the given compound is
Explanation of Solution
The given molecule is
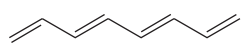
In the given molecule, the longest carbon chain containing all
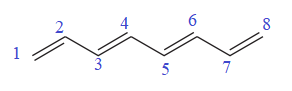
There are no substituents attached to the root. Thus, the complete IUPAC name of this molecule is
The IUPAC name of the compound is written according to the rules for nomenclature.
Want to see more full solutions like this?
Chapter B Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 5. Write the formation reaction of the following complex compounds from the following reactants: 6. AgNO₃ + K₂CrO₂ + NH₄OH → 7. HgNO₃ + excess KI → 8. Al(NO₃)₃ + excess NaOH →arrow_forwardIndicate whether the product formed in the reaction exhibits tautomerism. If so, draw the structure of the tautomers. CO₂C2H5 + CH3-NH-NH,arrow_forwardDraw the major product of this reaction N-(cyclohex-1-en-1-yl)-1-(pyrrolidino) reacts with CH2=CHCHO, heat, H3O+arrow_forward
- Draw the starting material that would be needed to make this product through an intramolecular Dieckmann reactionarrow_forwardDraw the major product of this reaction. Nitropropane reacts + pent-3-en-2-one reacts with NaOCH2CH3, CH3CHOHarrow_forwardIndicate whether the product formed in the reaction exhibits tautomerism. If so, draw the structure of the tautomers. OC2H5 + CoHs-NH-NH,arrow_forward
- Explain how substitutions at the 5-position of barbituric acid increase the compound's lipophilicity.arrow_forwardExplain how substitutions at the 5-position of phenobarbital increase the compound's lipophilicity.arrow_forwardName an interesting derivative of barbituric acid, describing its structure.arrow_forward
- Briefly describe the synthesis mechanism of barbituric acid from the condensation of urea with a β-diketone.arrow_forwardGiven the hydrazones indicated, draw the structures of the enamines that can be formed. Indicate the most stable enamine (explain). C6H5 C6H5 H C6H5 Harrow_forward4. Propose a Synthesis for the molecule below. You may use any starting materials containing 6 carbons or less (reagents that aren't incorporated into the final molecule such as PhзP do not count towards this total, and the starting material can have whatever non-carbon functional groups you want), and any of the reactions you have learned so far in organic chemistry I, II, and III. Your final answer should show each step separately, with intermediates and conditions clearly drawn.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

