
Concept explainers
Given, iodine azide adds to 1- butane only one product shown results.
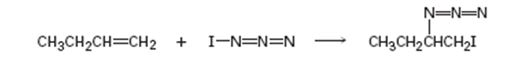
a) Add lone-pair electrons to the structure shown for IN3, and draw a second resonance form for the molecule.
Interpretation:
Lone pair of electrons is to be added to the structure of IN3 and another resonance form is to be drawn for it.
Concept introduction:
Lone pairs of electrons are those electrons which remain unshared on an atom in a molecule. Resonance forms differ only in the placements of their π or nonbonding electrons. Neither the position nor the hybridization of the atoms change in different resonance forms. Normal valence rules have to be followed.
To add:
Lone pair of electrons to the structure of IN3 and to draw another resonance form for it.
Answer to Problem 34MP
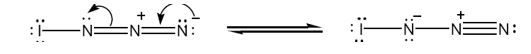
The structure of IN3 with lone pair of electrons added on each atom with another resonance form is shown below.
Explanation of Solution
Iodine has seven valence electrons (5s25p5) and nitrogen (2s22p3) has five (2s22p3) valence electrons. In the structure given, iodine has shared an electron with nitrogen in I-N bond. The other six electrons remain on it as lone pairs. Nitrogen is trivalent. The left nitrogen atom utilized three of its five electrons, one in bonding with iodine and other two in bonding with middle nitrogen. So it has a lone pair. The middle nitrogen has formed four bonds, two each with, left and right nitrogen. It has lost an additional electron to the nitrogen at right and has a positive charge. The nitrogen in the right, in addition to gaining an electron, has utilized only two of its five electrons for bonding with middle nitrogen. So it has a negative charge with two lone pair of electrons.
The structure of IN3 with lone pair of electrons added on each atom with another resonance form is shown below.

b) Calculate formal charges for the atoms in both resonance structures you drew for IN3 in part (a).
Interpretation:
The formal charges for the atoms in both resonance structures drawn for IN3 are to be calculated.
Concept introduction:
The formal charge on different atoms in a molecule can be calculated using the relation

To calculate:
The formal charges for the atoms in both resonance structures drawn for IN3
Answer to Problem 34MP
Two resonance structures for IN3 with lone pair of electrons on each atom are shown below.
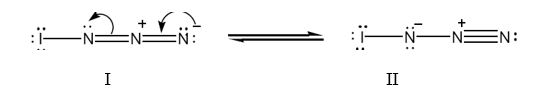
Formal charge for atoms in structure I:
Formal charge on iodine = 7-(2/2)-6 = 0
Formal charge on left nitrogen = 5-(6/2)-2 = 0
Formal charge on middle nitrogen = 5-(8/2)-0 = +1
Formal charge on right nitrogen = 5-(4/2)-4 = -1
Formal charge for atoms in structure II:
Formal charge on iodine = 7- (2/2)-6 = 0
Formal charge on left nitrogen = 5- (4/2)-4 = -1
Formal charge on middle nitrogen = 5-(8/2)-0 = +1
Formal charge on right nitrogen = 5-(6/2)-2 = 0
Explanation of Solution
The iodine atom has the outer electronic configuration 5s25p5. It has seven valence electrons. In structure I, the iodine atom has utilized one electron for forming a single bond with left nitrogen. It has six electrons as three lone pairs. Thus it has no formal charge.
Nitrogen has the outer electronic configuration 2s22p3. It has five valence electrons. It is trivalent. In structure I, the left nitrogen has used one electron for forming N-N bond and another electron for forming N-I bond. It has the remaining two electrons as a lone pair. Thus it has no formal charge.
The middle nitrogen has used four electrons two each in the two in N=N bonds. The middle nitrogen has lost one electron to the nitrogen in right and thus has a formal positive charge.
The right nitrogen has used two electrons for forming N=N bonds. It has gained one electron from middle nitrogen. Thus it has four electrons as two lone pairs. Thus it has a formal negative charge.
In structure II, the iodine atom has utilized one electron for forming a single bond with left nitrogen. It has six electrons as three lone pairs. Thus it has no formal charge.
Nitrogen has the outer electronic configuration 2s22p3. It has five valence electrons. It is trivalent. In structure II, the left nitrogen has used two electrons for forming N=N and an electron for forming N-I bond. It has four electrons as two lone pairs. Thus it has gained one electron and has a formal negative charge.
The middle nitrogen has used four electrons one for bonding with left nitrogen and three with right nitrogen. It has no lone pair of electrons. Thus it has lost one electron and has a formal positive charge.
The right nitrogen has used three electrons for forming three bonds with middle nitrogen. It has two electrons as a lone pair. Thus it has no a formal charge.
Two resonance structures for IN3 with lone pair of electrons on each atom are shown below.
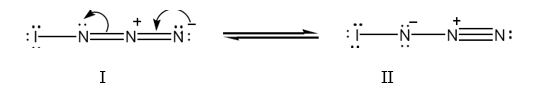
Formal charge for atoms in structure I:
Formal charge on iodine = 7- (2/2)-6 = 0
Formal charge on left nitrogen = 5- (6/2)-2 = 0
Formal charge on middle nitrogen = 5-(8/2)-0 = +1
Formal charge on right nitrogen = 5-(4/2)-4 = -1
Formal charge for atoms in structure II:
Formal charge on iodine = 7- (2/2)-6 = 0
Formal charge on left nitrogen = 5- (4/2)-4 = -1
Formal charge on middle nitrogen = 5-(8/2)-0 = +1
Formal charge on right nitrogen = 5-(6/2)-2 = 0
c) In light of the result observed when IN3 adds to 1-butane, what is the polarity of the I-N3 bond? Propose a mechanism for the reaction using curved arrows to show the electron flow in each step.
Interpretation:
The polarity of I-N3 bond when it adds to 1-butene is to be stated. A mechanism is to be proposed for the reaction using curved arrows to show the electron flow in each step.
Concept introduction:
If the two atoms in a covalent bond differ in their electronegativity values, then the bond becomes polar with the less electronegative atom at the negative end and the more electronegative atom at the positive end of the dipole. The addition follows Markovnikov regiochemistry, the negative part gets added to the more alkyl substituted carbon in the double bond and the positive part gets added to the less alkyl substituted carbon in the double bond.
To state:
The polarity of I-N3 bond when it adds to 1-butene. To propose a mechanism for the reaction using curved arrows to show the electron flow in each step.
Answer to Problem 34MP
The polarity of I-N3 bond is
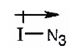
A mechanism for the reaction using curved arrows to show the electron flow in each step when I-N3 adds to 1-butene is shown below.
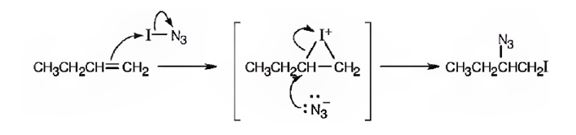
Explanation of Solution
In the product the iodine atom is attached to the less alkyl substituted carbon in double bond. If the addition occurs as per the Markovnikov regiochemistry, iodine must be the positive part in N-I3. The reaction is initiated by the attack of the π electrons of the double bond on the positively polarized iodine of N-I3 leading to the formation of an iodonium ion. In the second step, the N3- ion attacks the iodonium ion from the least hindered side to give the product.
The polarity of I-N3 bond is
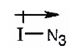
A mechanism for the reaction of using curved arrows to show the electron flow in each step when I-N3 adds to 1-butene is shown below.

Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry - With Access (Custom)
- A 0.10 M solution of acetic acid (CH3COOH, Ka = 1.8 x 10^-5) is titrated with a 0.0250 M solution of magnesium hydroxide (Mg(OH)2). If 10.0 mL of the acid solution is titrated with 10.0 mL of the base solution, what is the pH of the resulting solution?arrow_forwardGiven a complex reaction with rate equation v = k1[A] + k2[A]2, what is the overall reaction order?arrow_forwardPlease draw the structure in the box that is consistent with all the spectral data and alphabetically label all of the equivalent protons in the structure (Ha, Hb, Hc....) in order to assign all the proton NMR peaks. The integrations are computer generated and approximate the number of equivalent protons. Molecular formula: C13H1802 14 13 12 11 10 11 (ppm) Structure with assigned H peaks 2.08 3.13arrow_forward
- CHEMICAL KINETICS. One of the approximation methods for solving the rate equation is the steady-state approximation method. Explain what it consists of.arrow_forwardCHEMICAL KINETICS. One of the approximation methods for solving the rate equation is the limiting or determining step approximation method. Explain what it consists of.arrow_forwardCHEMICAL KINETICS. Indicate the approximation methods for solving the rate equation.arrow_forward
- TRANSMITTANCE เบบ Please identify the one structure below that is consistent with the 'H NMR and IR spectra shown and draw its complete structure in the box below with the protons alphabetically labeled as shown in the NMR spectrum and label the IR bands, including sp³C-H and sp2C-H stretch, indicated by the arrows. D 4000 OH LOH H₂C CH3 OH H₂C OCH3 CH3 OH 3000 2000 1500 HAVENUMBERI-11 1000 LOCH3 Draw your structure below and label its equivalent protons according to the peak labeling that is used in the NMR spectrum in order to assign the peaks. Integrals indicate number of equivalent protons. Splitting patterns are: s=singlet, d=doublet, m-multiplet 8 3Hb s m 1Hd s 3Hf m 2Hcd 2Had 1He 鄙视 m 7 7 6 5 4 3 22 500 T 1 0arrow_forwardRelative Transmittance 0.995 0.99 0.985 0.98 Please draw the structure that is consistent with all the spectral data below in the box and alphabetically label the equivalent protons in the structure (Ha, Hb, Hc ....) in order to assign all the proton NMR peaks. Label the absorption bands in the IR spectrum indicated by the arrows. INFRARED SPECTRUM 1 0.975 3000 2000 Wavenumber (cm-1) 1000 Structure with assigned H peaks 1 3 180 160 140 120 100 f1 (ppm) 80 60 40 20 0 C-13 NMR note that there are 4 peaks between 120-140ppm Integral values equal the number of equivalent protons 10.0 9.0 8.0 7.0 6.0 5.0 4.0 3.0 2.0 1.0 0.0 fl (ppm)arrow_forwardCalculate the pH of 0.0025 M phenol.arrow_forward
- In the following reaction, the OH- acts as which of these? NO2-(aq) + H2O(l) ⇌ OH-(aq) + HNO2(aq)arrow_forwardUsing spectra attached, can the unknown be predicted? Draw the predicition. Please explain and provide steps. Molecular focrmula:C16H13ClOarrow_forwardCalculate the percent ionization for 0.0025 M phenol. Use the assumption to find [H3O+] first. K = 1.0 x 10-10arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

