
Us Solutions Manual To Accompany Elements Of Physical Chemistry 7e
7th Edition
ISBN: 9780198802259
Author: ATKINS
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 8, Problem 8B.2E
(a)
Interpretation Introduction
Interpretation:
The ground-state electronic configuration of carbon atom has to be predicted.
Concept-Introduction:
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled. Greater the number of parallel spins of electrons more stable the orbital. This is known as Hund’s rule.
- The order of electrons filling in an atom is given as
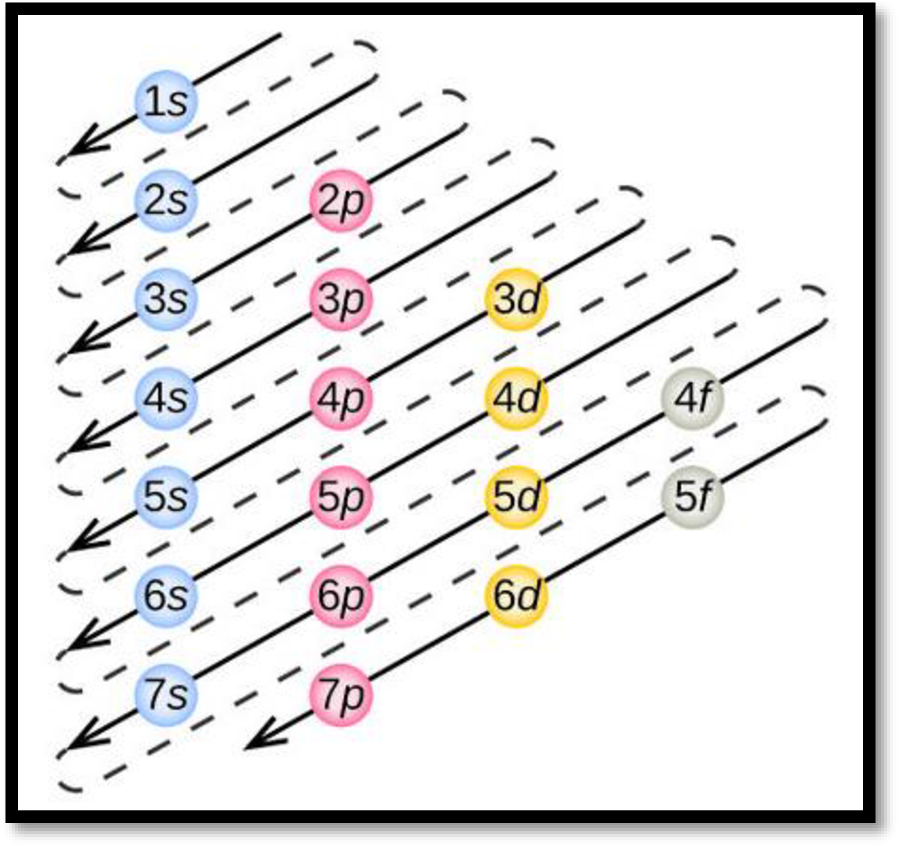
Figure 1
(b)
Interpretation Introduction
Interpretation:
The ground-state electronic configuration of titanium ion has to be predicted.
Concept-Introduction:
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled. Greater the number of parallel spins of electrons more stable the orbital. This is known as Hund’s rule.
- The order of electrons filling in an atom is given as
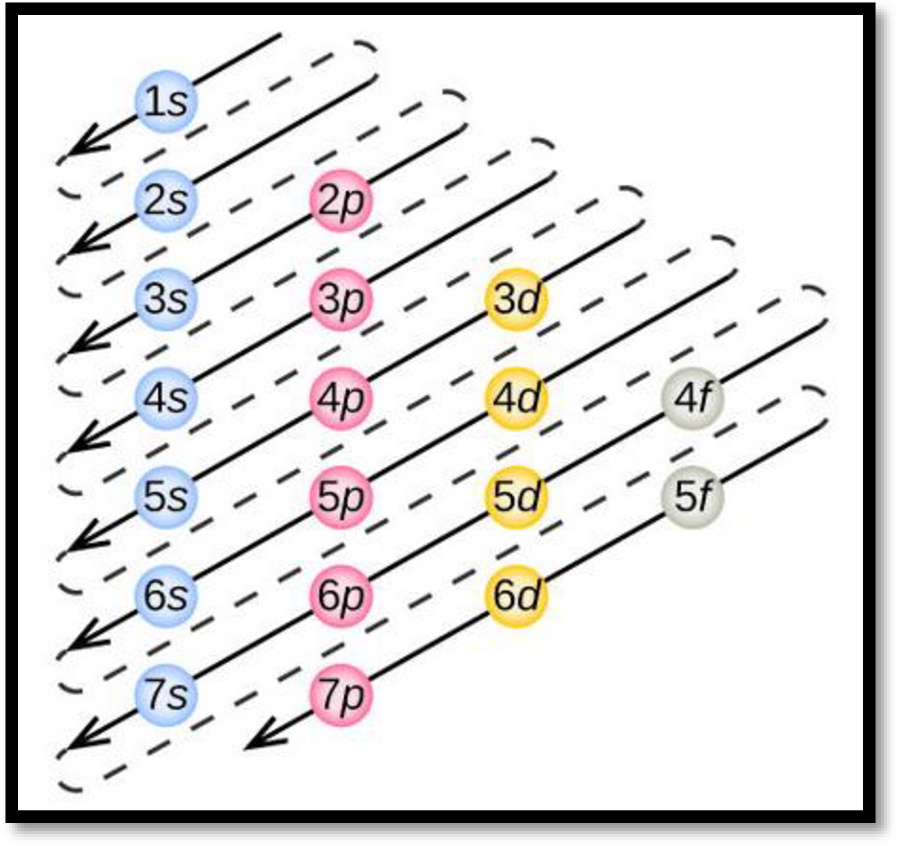
Figure 1
(b)
Interpretation Introduction
Interpretation:
The ground-state electronic configuration of manganese ion has to be predicted.
Concept-Introduction:
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled. Greater the number of parallel spins of electrons more stable the orbital. This is known as Hund’s rule.
- The order of electrons filling in an atom is given as
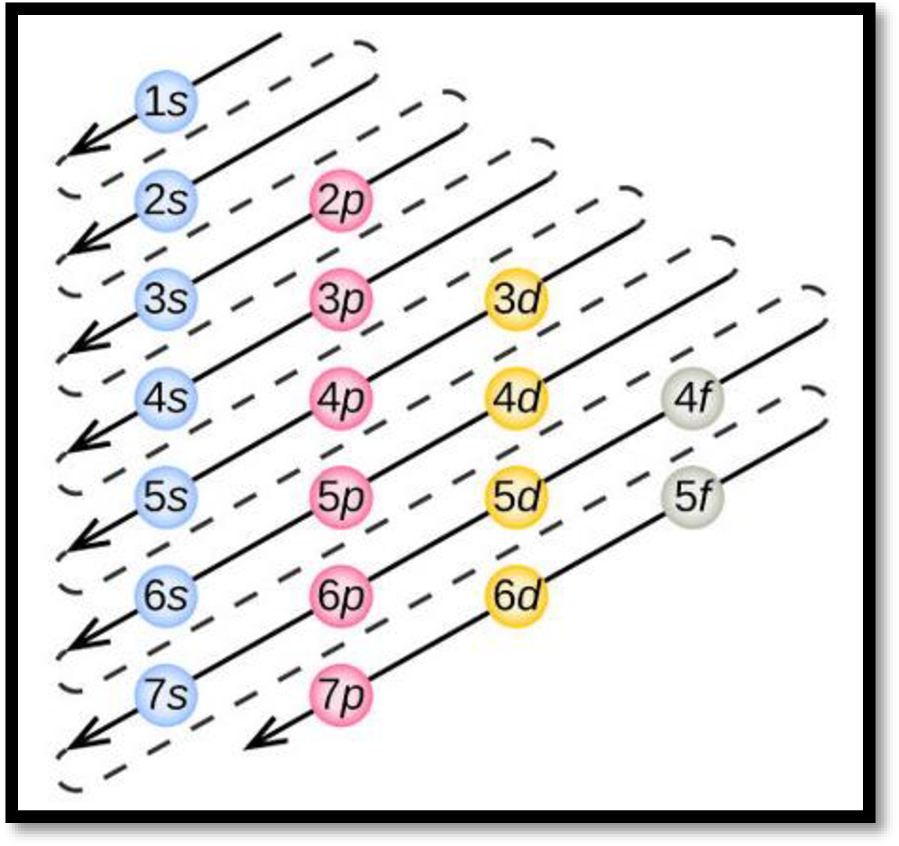
Figure 1
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please help with the curved arrow mechanism of this reaction, thank you
Concentration (mg/l)
Peak Area
0
158
10
10241
20
18425
30
26457
40
37125
50
44256
60
56124
Question: Determine the regression equation (a and b coefficients) from first principles
Concentration (mg/l)
Peak Area
0
158
10
10241
20
18425
30
26457
40
37125
50
44256
60
56124
You have been asked to determine the concentration of citral in a highly valued magnolia essential oil.
QUESTION: Calculate the concentration of citral in your highly valued magnolia essential oil which returns a peak area of 41658
Chapter 8 Solutions
Us Solutions Manual To Accompany Elements Of Physical Chemistry 7e
Ch. 8 - Prob. 8A.1STCh. 8 - Prob. 8A.2STCh. 8 - Prob. 8A.3STCh. 8 - Prob. 8A.4STCh. 8 - Prob. 8A.5STCh. 8 - Prob. 8B.2STCh. 8 - Prob. 8B.3STCh. 8 - Prob. 8B.4STCh. 8 - Prob. 8C.1STCh. 8 - Prob. 8C.2ST
Ch. 8 - Prob. 8D.1STCh. 8 - Prob. 8D.2STCh. 8 - Prob. 8D.3STCh. 8 - Prob. 8D.4STCh. 8 - Prob. 8D.5STCh. 8 - Prob. 8D.6STCh. 8 - Prob. 8D.7STCh. 8 - Prob. 8A.1ECh. 8 - Prob. 8A.2ECh. 8 - Prob. 8A.3ECh. 8 - Prob. 8A.6ECh. 8 - Prob. 8A.7ECh. 8 - Prob. 8A.8ECh. 8 - Prob. 8A.9ECh. 8 - Prob. 8A.10ECh. 8 - Prob. 8A.12ECh. 8 - Prob. 8B.1ECh. 8 - Prob. 8B.2ECh. 8 - Prob. 8B.3ECh. 8 - Prob. 8C.1ECh. 8 - Prob. 8C.2ECh. 8 - Prob. 8D.1ECh. 8 - Prob. 8D.2ECh. 8 - Prob. 8D.3ECh. 8 - Prob. 8D.4ECh. 8 - Prob. 8D.5ECh. 8 - Prob. 8D.6ECh. 8 - Prob. 8D.7ECh. 8 - Prob. 8D.8ECh. 8 - Prob. 8D.9ECh. 8 - Prob. 8D.11ECh. 8 - Prob. 8D.12ECh. 8 - Prob. 8.1DQCh. 8 - Prob. 8.2DQCh. 8 - Prob. 8.3DQCh. 8 - Prob. 8.4DQCh. 8 - Prob. 8.5DQCh. 8 - Prob. 8.6DQCh. 8 - Prob. 8.7DQCh. 8 - Prob. 8.8DQCh. 8 - Prob. 8.2PCh. 8 - Prob. 8.4PCh. 8 - Prob. 8.7PCh. 8 - Prob. 8.8PCh. 8 - Prob. 8.9PCh. 8 - Prob. 8.11PCh. 8 - Prob. 8.12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Need help with these problems...if you can please help me understand problems E & F.arrow_forwardPlease help me solve these problems. Thank you in advance.arrow_forwardPredict the products of this organic reaction: O N IN A N + H2O + HCI ? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. 田 C + Explanation Check Click and drag to start drawing a structure. C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward
- 6. For each of the following, fill in the synthesis arrows with reagents and show the intermediates. You DO NOT need to use the same number of arrows that are shown (you may use more or less), but the product must be formed from the reactant. Then write the mechanism of one step in the synthesis (you can choose which step to write the mechanism for), including all reagents required, clearly labeling the nucleophile and electrophile for each step, and using curved arrows to show the steps in the mechanism. a. b. OHarrow_forwardDraw the productsarrow_forwardDraw the correct productsarrow_forward
- E Organic Chemistry Maxwell Draw the correct products, in either order, for the ozonolysis reaction: 1) O3, CH2Cl2, -78 °C Product 1 + Product 2 2) Zn, HOAc Draw product 1. Select Draw Templates More C H O presented by M Draw product 2. Erase Select Draw Templates M / # # carrow_forward✓ edict the products of this organic reaction: ---- ။ A CH3–C−NH–CH2–C−CH3 + KOH ? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. C 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibiliarrow_forwardPredict the product of this organic reaction: A HO-C-CH3 + CH3NH2 P+ H2O Specifically, in the drawing area below draw the condensed structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. marrow_forward
- H 1) OsO4, pyridine 2) Na2SO3 or NaHSO3 in H₂O 2 productsarrow_forward● Biological Macromolecules Naming and drawing cyclic monosaccharides Your answer is incorrect. • Row 1: Your answer is incorrect. Row 3: Your answer is incorrect. • Row 4: Your answer is incorrect. Try again... 0/5 Give the complete common name, including anomer and stereochemistry labels, of the following molecules. You will find helpful information in the ALEKS resource. CH2OH OH OH H H I H OH OH H] H CH2OH H OH ẞ-L-sorbose HOCH2 OH OH H HOCH2 H OH OH H OH H H CH2OH OH H H OH H I- H OH H OH Explanation Recheck W E R % 25 α B Y X & 5 D F G H McGraw Hill LLC. All Rights Reserved. Terms of Use | Pr Parrow_forwardWhat is the missing reactant in this organic reaction? + R -A HO IN + H₂O Specifically, in the drawing area below draw the skeletal ("line") structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. Explanation Check Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY