
To write:
The complete and balanced
a) 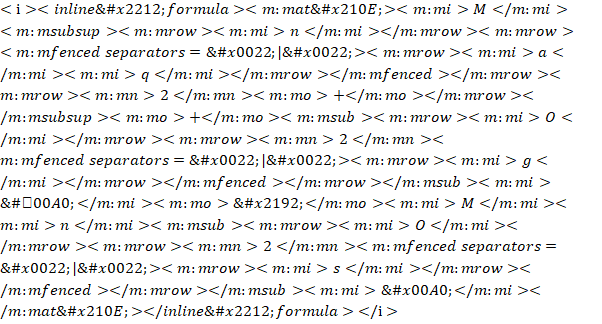 (basic solution)
(basic solution)
b)  (acidic solution)
(acidic solution)
c)
Answer to Problem 8.96QA
Solution:
Complete and balanced redox reaction is
a)
b)
c)
Explanation of Solution
1) Concept:
To balance a redox reaction, first we have to assign oxidation numbers to each of the elements in the compound and decide which element is going to be reduced and oxidized. Oxidation is loss of electrons or increase in oxidation number while reduction is gain of electrons or decrease in oxidation number.
2) Given:
i)
ii)  (acidic solution)
(acidic solution)
iii)
3) Calculations:
a)
i) The following preliminary expression relates the known reactant and products:
There are equal number of
ii) We perform an
As
iii) To balance these
iv) The ionic charge on the reactant is
v) Taking an inventory of atoms on both sides of the reaction arrow, we find that the left side has four more
Now the reaction is balanced.
b)
i) The following preliminary expression relates the known reactant and products:
There are equal numbers of  on both product and reactant sides, but there are unequal numbers of
on both product and reactant sides, but there are unequal numbers of
ii) We perform an
The
The change in the oxidation number is the same for oxidation and reduction.
iii) The ionic charge on the reactant is
iv) Taking an inventory of atoms on both sides of the reaction arrow, we find that the left side has four more
Now the reaction is balanced
c)
i) The following preliminary expression relates the known reactant and products:
There are unequal numbers of
ii) We perform an
The
The
The change in the oxidation number is the same for oxidation and reduction.
Also there are same number of charges on the reactant and product sides, so the reaction is balanced.
Conclusion:
In a redox reaction, substances either gain electrons or thereby undergo reduction, or they lose electrons and undergo oxidation. The given reaction is redox as the oxidation number of elements in the reactants changes during the reaction.
Want to see more full solutions like this?
Chapter 8 Solutions
CHEM:ATOM FOC 2E CL (TEXT)
- Using a cell of known pathlength b = 1.25115 x 10-3 cm, a water absorption spectrum was measured. The band at 1645 cm-1, assigned to the O-H bending, showed an absorbance, A, of 1.40. a) Assuming that water density is 1.00 g/mL, calculate the water molar concentration c (hint: M= mole/L) b) Calculate the molar absorptivity, a, of the 1645 cm-1 band c) The transmitted light, I, can be written as I= Ioexp(-xb), where x is the absorption coefficient (sometimes designated as alpha), Io is the input light, and b is the cell pathlength. Prove that x= (ln10)*x*c d) Calculate x for the 1645 cm-1 bandarrow_forwardConvert 1.38 eV into wavelength (nm) and wavenumber (cm-1) (c = 2.998 x 108 m/s; h = 6.626 x 10-34 J*s).arrow_forwardCan you help me understand the CBC method on metal bridging by looking at this problem?arrow_forward
- A partir de Aluminio y Co(NO3)2ꞏ6H2O, indicar las reacciones a realizar para obtener Azul de Thenard (Al2CoO4).arrow_forwardTo obtain Thenard Blue (Al2CoO4), the following reaction is correct (performed in an oven):Al(OH)3 + Co(OH)2 → Al2CoO4 + 4 H2Oarrow_forwardProblem 38 can u explain and solve thanks april 24arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





