
CHEMISTRY
2nd Edition
ISBN: 9781593995782
Author: OpenStax
Publisher: XANEDU PUBLISHING
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 30E
For each of the following structures, determine the hybridization requested and whether the electrons will be delocalized:
(a) Hybridization of each carbon
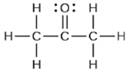
(b) Hybridization of sulfur
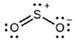
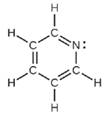
(c) All atoms
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Could you redraw these and also explain how to solve them for me pleas
None
Draw the curved-arrow mechanism with the drawings of the molecules, not
just abbreviations.
-NO₂
Sn, HCl (aq)
E
D
H
(CH3CO)₂O
-NH2
CH3
Chapter 8 Solutions
CHEMISTRY
Ch. 8 - Explain how and bonds are similar and how they...Ch. 8 - Draw a curve that describes the energy of a system...Ch. 8 - Explain why bonds occur at specific average bond...Ch. 8 - Use valence bond theory to explain the bonding in...Ch. 8 - Use valence bond theory to explain the bonding in...Ch. 8 - How many and bonds are present in the molecule...Ch. 8 - A friend tells you N2 has three bonds due to...Ch. 8 - Draw the Lewis structures for CO2 and CO, and...Ch. 8 - Why is the concept of hybridization required in...Ch. 8 - Give the shape that describes each hybrid orbital...
Ch. 8 - Explain why a carbon atom cannot form five bonds...Ch. 8 - What is the hybridization of the central atom in...Ch. 8 - A molecule with the formula AB3 could have one of...Ch. 8 - Methionine, CH3SCH2CH2CH(NH2)CO2H, is an amino...Ch. 8 - Sulfuric acid is manufactured by a series of...Ch. 8 - Two important industrial chemicals, ethene, C2H4,...Ch. 8 - For many years after they were discovered, it was...Ch. 8 - Consider nitrous acid, HNO2 (HONO). (a) Write a...Ch. 8 - Strike-anywhere matches contain a layer of KClO3...Ch. 8 - Identify the hybridization of each carbon atom in...Ch. 8 - Write Lewis structures for NF3 and PP5. On the...Ch. 8 - In addition to NF3, two other fluoro derivatives...Ch. 8 - The bond energy of a C—C single bond averages 347...Ch. 8 - For the carbonate ion, CO32, draw all of the...Ch. 8 - A useful solvent that will dissolve salts as well...Ch. 8 - For the molecule allene, H2C=C=CH2, give the...Ch. 8 - Identify the hybridization of the central atom in...Ch. 8 - Describe the molecular geometry and hybridization...Ch. 8 - For each of the following molecules, indicate the...Ch. 8 - For each of the following structures, determine...Ch. 8 - Draw the orbital diagram for carbon in CO2 showing...Ch. 8 - Sketch the distribution of electron density in the...Ch. 8 - How are the following similar, and how do they...Ch. 8 - If molecular orbitals are created by combining...Ch. 8 - Can a molecule with an odd number of electrons...Ch. 8 - Can a molecule with an even number of electrons...Ch. 8 - Why are bonding molecular orbitals lower in energy...Ch. 8 - Calculate the bond order for an ion with this...Ch. 8 - Explain why an electron in the bonding molecular...Ch. 8 - Predict the valence electron molecular orbital...Ch. 8 - Determine the bond order of each member of the...Ch. 8 - For the first ionization energy for an N2...Ch. 8 - Compare the atomic and molecular orbital diagrams...Ch. 8 - Which of the period 2 homonuclear diatomic...Ch. 8 - A friend tells you that the 2s orbital for...Ch. 8 - True or false: Boron contains 2s22p1 valence...Ch. 8 - What charge would be needed on F2 to generate an...Ch. 8 - Predict whether the MO diagram S2 would show s-p...Ch. 8 - Explain why N22+ is diamagnetic, while O24+, which...Ch. 8 - Using the MO diagrams, predict the bond order for...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which type of cartilage is most plentiful in the adult body?
Anatomy & Physiology (6th Edition)
Use a globe or map to determine, as accurately as possible, the latitude and longitude of Athens, Greece.
Applications and Investigations in Earth Science (9th Edition)
4. Three groups of nonvascular plants are _______, ______, and _______. Three groups of seedless vascular plant...
Biology: Life on Earth (11th Edition)
Match the following examples of mutagens. Column A Column B ___a. A mutagen that is incorporated into DNA in pl...
Microbiology: An Introduction
In pea plants, plant height, seed shape, and seed color are governed by three independently assorting genes. Th...
Genetic Analysis: An Integrated Approach (3rd Edition)
Match each of the following items with all the terms it applies to:
Human Physiology: An Integrated Approach (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is/are the product(s) of the following reaction? Select all that apply. * HI A B C OD OH A B OH D Carrow_forwardIn the image, the light blue sphere represents a mole of hydrogen atoms, the purple or teal spheres represent a mole of a conjugate base. A light blue sphere by itself is H+. Assuming there is 2.00 L of solution, answer the following: The Ka of the left & right solution is? The pH of the left & right solution is? The acid on the left & right is what kind of acid?arrow_forwardNonearrow_forward
- Nonearrow_forwardNonearrow_forwardWhat spectral features allow you to differentiate the product from the starting material? Use four separate paragraphs for each set of comparisons. You should have one paragraph each devoted to MS, HNMR, CNMR and IR. 2) For MS, the differing masses of molecular ions are a popular starting point. Including a unique fragmentation is important, too. 3) For HNMR, CNMR and IR state the peaks that are different and what makes them different (usually the presence or absence of certain groups). See if you can find two differences (in each set of IR, HNMR and CNMR spectra) due to the presence or absence of a functional group. Include peak locations. Alternatively, you can state a shift of a peak due to a change near a given functional group. Including peak locations for shifted peaks, as well as what these peaks are due to. Ideally, your focus should be on not just identifying the differences but explaining them in terms of functional group changes.arrow_forward
- Nonearrow_forwardNonearrow_forwardIn the solid state, oxalic acid occurs as a dihydrate with the formula H2C2O4 C+2H2O. Use this formula to calculate the formula weight of oxalic acid. Use the calculated formula weight and the number of moles (0.00504mol) of oxalic acid in each titrated unknown sample recorded in Table 6.4 to calculate the number of grams of pure oxalic acid dihydrate contained in each titrated unknown sample.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY