
Concept explainers
(a)
Interpretation:
The term “acid” should be defined by giving an example.
Concept Introduction:
The chemical properties of an acid and a base are different from each other because both are chemically opposite. Acid and base reacts with each other to form water and salt. The reaction is known as neutralization reaction. In aqueous solution, acid gives hydrogen ion/s 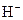 and base gives hydroxide ion/s
and base gives hydroxide ion/s 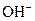 these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
(a)
Answer to Problem 1RQ
Solution:
A compound that gives H+ ion/s in the aqueous solution and turns litmus indicator dye to red is termed as an acid. For example: Hydrogen chloride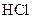
Explanation of Solution
An acid is a compound with some particular properties such as sour in taste and its reaction with base results in the formation of salt and water. It turns litmus indicator dye to red. On reaction with metals, hydrogen gas H2 is released. They also produce H+ ion/s in aqueous solution.
Example of an acid is hydrogen chloride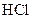 It is a strong acid and completely dissociates into H+ and
It is a strong acid and completely dissociates into H+ and 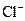 ions in the aqueous solution.
ions in the aqueous solution.
Hydrogen chloride is an acid because it gives H+ ion in the aqueous solution. Also, it turns litmus indicator dye to red. On reaction with metals such as zinc and iron it produces H2 gas.
(b)
Interpretation:
The term “base” should be defined by giving an example.
Concept Introduction:
The chemical properties of an acid and a base are different from each other because both are chemically opposite. Acid and base reacts with each other to form water and salt. The reaction is known as neutralization reaction. In aqueous solution, acid gives hydrogen ion/s 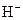 and base gives hydroxide ion/s
and base gives hydroxide ion/s 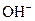 these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
(b)
Answer to Problem 1RQ
Solution:
A compound that gives OH- ion/s in the aqueous solution and turns litmus indicator dye to blue is termed as a base. For example: Sodium hydroxide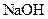
Explanation of Solution
A base is a compound with some particular properties such as bitter in taste and its reaction with acid results in the formation of salt and water. It turns litmus indicator dye to blue. It feels slippery on the skin. It produces OH- ion/s in the aqueous solution.
Example of a base is sodium hydroxide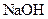 It is a strong base and completely dissociates into Na+ and
It is a strong base and completely dissociates into Na+ and 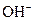 ions in the aqueous solution.
ions in the aqueous solution.
Sodium hydroxide is a base because it produces OH- ion in the aqueous solution. Also, it turns litmus indicator dye to blue. It reacts with acid to form salt and water.
(c)
Interpretation:
The term “salt” should be defined by giving an example.
Concept Introduction:
The chemical properties of an acid and a base are different from each other because both are chemically opposite. Acid and base reacts with each other to form water and salt. The reaction is known as neutralization reaction. In aqueous solution, acid gives hydrogen ion/s 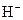 and base gives hydroxide ion/s
and base gives hydroxide ion/s 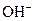 these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
these positive and negative ions combined to form H2O and affect the concentration of aqueous solution.
(c)
Answer to Problem 1RQ
Solution:
Salt is defined as an ionic compound formed from the reaction of an acid and a base. For example: sodium chloride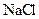 which is formed from the reaction of
which is formed from the reaction of 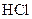 and
and 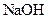 as follows:
as follows:
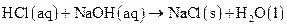
Explanation of Solution
If an acid reacts with a base, salt is formed. It is an ionic compound because it is made up of ions of acid and base. A salt is formed from neutralization reaction of an acid and a base therefore, it is neutral in nature.
For example: Sodium chloride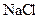 which is formed from the reaction of
which is formed from the reaction of 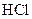 and
and 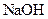 as follows:
as follows:
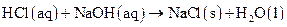
Thus, salt is an ionic compound such as sodium chloride which is formed from the reaction of 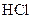 (acid) and
(acid) and 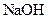 (base).
(base).
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry for Changing Times
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardPlease help me answer a. Please and thank you I advance.arrow_forwardDraw both of the chair flips for both the cis and trans isomers for the following compounds: 1,4-diethylcyclohexane 1-methyl-3-secbutylcyclohexanearrow_forward
- Ppplllleeeaaasssseeee hellppp wiithhh thisss physical chemistryyyyy I talked like this because AI is very annoyingarrow_forwardFor this question, if the product is racemic, input both enantiomers in the same Marvin editor. A) Input the number that corresponds to the reagent which when added to (E)-but-2-ene will result in a racemic product. Input 1 for Cl, in the cold and dark Input 2 for Oy followed by H₂O, Zn Input 3 for D₂ with metal catalyst Input 4 for H₂ with metal catalyst B) Draw the skeletal structure of the major organic product made from the reagent in part A Marvin JS Help Edit drawing C) Draw the skeletal structure of the major organic product formed when (2)-but-2-ene is treated with peroxyacetic acid. Marvin 35 Helparrow_forwardMichael Reactions 19.52 Draw the products from the following Michael addition reactions. 1. H&C CH (a) i 2. H₂O* (b) OEt (c) EtO H₂NEt (d) ΕΙΟ + 1. NaOEt 2. H₂O' H H 1. NaOEt 2. H₂O*arrow_forward
- Rank the labeled protons (Ha-Hd) in order of increasing acidity, starting with the least acidic. НОН НЬ OHd Онсarrow_forwardCan the target compound at right be efficiently synthesized in good yield from the unsubstituted benzene at left? ? starting material target If so, draw a synthesis below. If no synthesis using reagents ALEKS recognizes is possible, check the box under the drawing area. Be sure you follow the standard ALEKS rules for submitting syntheses. + More... Note for advanced students: you may assume that you are using a large excess of benzene as your starting material. C :0 T Add/Remove step Garrow_forwardThe following equations represent the formation of compound MX. What is the AH for the electron affinity of X (g)? X₂ (g) → 2X (g) M (s) → M (g) M (g) M (g) + e- AH = 60 kJ/mol AH = 22 kJ/mol X (g) + e-X (g) M* (g) +X (g) → MX (s) AH = 118 kJ/mol AH = ? AH = -190 kJ/mol AH = -100 kJ/mol a) -80 kJ b) -30 kJ c) -20 kJ d) 20 kJ e) 156 kJarrow_forward
- A covalent bond is the result of the a) b) c) d) e) overlap of two half-filled s orbitals overlap of a half-filled s orbital and a half-filled p orbital overlap of two half-filled p orbitals along their axes parallel overlap of two half-filled parallel p orbitals all of the abovearrow_forwardCan the target compound at right be efficiently synthesized in good yield from the unsubstituted benzene at left? starting material target If so, draw a synthesis below. If no synthesis using reagents ALEKS recognizes is possible, check the box under the drawing area. Be sure you follow the standard ALEKS rules for submitting syntheses. + More... Note for advanced students: you may assume that you are using a large excess of benzene as your starting material. C T Add/Remove step X ноarrow_forwardWhich one of the following atoms should have the largest electron affinity? a) b) c) d) 으으 e) 1s² 2s² 2p6 3s¹ 1s² 2s² 2p5 1s² 2s² 2p 3s² 3p² 1s² 2s 2p 3s² 3p6 4s2 3ds 1s² 2s² 2p6arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





