
Concept explainers
a)
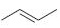
Interpretation:
The reaction of the alkene, 2-butene, with a proton to give a carbocation is to be shown using curved arrows. The structure of the carbocation formed as also to be given.
Concept introduction:
The double bond in
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To show:
Using curved arrows the reaction of the alkene, 2-butene, with a proton to give a carbocation and also to give the structure of the carbocation.
b)
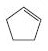
Interpretation:
The reaction of the alkene, cyclopentene, with a proton to give a carbocation is to be shown using curved arrows. The structure of the carbocation formed as also to be given.
Concept introduction:
The double bond in alkenes is nucleophilic. The π electrons can be donated to a proton. By donating the π electrons one of the carbons in the double bond forms a new C-H bond while the other carbon gets a positive charge resulting in a carbocation.
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To show:
Using curved arrows the reaction of the alkene, cyclopentene, with a proton to give a carbocation and also to give the structure of the carbocation.
c)
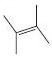
Interpretation:
The reaction of the alkene, 2,3-dimethyl-2-butene, with a proton to give a carbocation is to be shown using curved arrows. The structure of the carbocation formed as also to be given.
Concept introduction:
The double bond in alkenes is nucleophilic. The π electrons can be donated to a proton. By donating the π electrons one of the carbons in the double bond forms a new C-H bond while the other carbon gets a positive charge resulting in a carbocation.
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To show:
Using curved arrows the reaction of the alkene, 2,3-dimethyl-2-butene, with a proton to give a carbocation and also to give the structure of the carbocation.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Study Guide with Student Solutions Manual for McMurry's Organic Chemistry, 9th
- Hydrochloric acid (HCl) reacts with sodium hydroxide (NaOH) to form sodium chloride (NaCl) and water. If ΔH ° = −56.13 kJ/mol and ΔS ° = 79.11 J/mol ⋅ K, what is the temperature of the reaction if ΔG ° = −80.89 kJ/mol?arrow_forwardFor a particular hypothetical reaction, A+B →2C, the value of AG° is -125 kJ/mol. What is the value of AG for this reaction at 35°C when [A] = 0.10 M, [B] = 0.05 M, and [C] = 2.0 × 10¹ M?arrow_forwardIn an experiment, 74.3 g of metallic copper was heated to 100.0°C and then quickly dropped into 200.0 mL of water in a calorimeter. The heat capacity of the calorimeter with the water was 875 J/°C. The initial temperature of the calorimeter was 27.5°C, and the final temperature after addition of the metal was 29.8°C. What is the value of the molar heat capacity of copper?arrow_forward
- The Haber-Bosch process permits the direct conversion of molecular nitrogen to ammonia, which can be used in large-scale fertilizer production. Given the balanced Haber-Bosch reaction and using the bond energies in the table below, estimate the enthalpy change associated with the reaction. N2(g) + 3H2(g) → 2NH3(g) Bond N=N N = N Energy (kJ/mol) 941 418 N-N H-H N-H 163 435 388arrow_forwardBenzoic acid is used to determine the heat capacity of bomb calorimeters because it can be obtained in pure form and its energy of combustion is known very accurately (−26.43 kJ/g). Determine the heat capacity of a calorimeter that had a temperature increase of 9.199°C when 3.500 g of benzoic acid was used.arrow_forwardGiven the standard enthalpies of formation for the following substances, determine the reaction enthalpy for the following reaction. 2N2H4(g) + 2NO2(g) → 3N2(g) + 4H2O(g) AHrxn ? kJ Substance AH in kJ/mol N2H4(g) +95.4 NO2(g) +33.1 H2O(g) -241.8arrow_forward
- If 7.3 kJ of energy are required to change the temperature of water from 5.0 to 70.0, what was the volume of water? (cs = 4.184 J/(g ⋅ ), d = 1.00 g/mL)arrow_forwardBALANCE CHEMICAL REACTIONarrow_forwardPredict the product(s) of the following reactions. If no reaction, write "NR". a) Cl₂ FeCl3 e) HNO3 H2SO4 b) NO2 CI. HNO3 f) Br Br2 OH H2SO4 HO3S. FeBr3 c) Cl2 g) FeCl3 F d) O₂N Br2 FeBr3 O₂N OH HNO3 CH3 H2SO4arrow_forward
- ulating the pH salt solution Calculate the pH at 25 °C of a 0.75M solution of anilinium chloride (C6H5NH3C1). Note that aniline (C6H5NH2) is a weak base with a pK of 4.87. Round your answer to 1 decimal place. pH = ☐ ☑ ⑤ ? olo 18 Ararrow_forwardI apologize, but the app is not allowing me to post the other 4 pictures of the thermodynamics chart. But I believe the values are universal. Please help!arrow_forwardCalculating the pH of a salt solution Calculate the pH at 25 °C of a 0.29M solution of potassium butanoate (KC3H,CO2). Note that butanoic acid (HC3H,CO2) is a weak acid with a pKa of 4.82. Round your answer to 1 decimal place. pH = -0 Х olo 18 Ararrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
