
a)
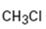
Interpretation:
Whether CH3Cl is likely to behave as a nucleophile or electrophile or both to be stated.
Concept introduction:
A nucleophile (either negatively charged or a neutral molecule containing an atom with at least one lone pair of electrons) has an electron rich-site. An electrophile (either positively charged or a neutral molecule containing an electron deficient atom) has an electron poor-site.
To state:
Whether CH3Cl will behave as nucleophile or electrophile or both.
b)
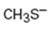
Interpretation:
Whether CH3S- is likely to behave as a nucleophile or electrophile or both to be stated.
Concept introduction:
A nucleophile (either negatively charged or a neutral molecule containing an atom with at least one lone pair of electrons) has an electron rich-site. An electrophile (either positively charged or a neutral molecule containing an electron deficient atom) has an electron poor-site.
To state:
Whether CH3S- will behave as nucleophile or electrophile or both.
c)
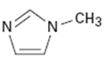
Interpretation:
Whether the compound given is likely to behave as a nucleophile or electrophile or both to be stated.
Concept introduction:
A nucleophile (either negatively charged or a neutral molecule containing an atom with at least one lone pair of electrons) has an electron rich-site. An electrophile (either positively charged or a neutral molecule containing an electron deficient atom) has an electron poor-site.
To state:
Whether the compound given will behave as nucleophile or electrophile or both.
d)
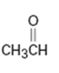
Interpretation:
Whether CH3CHO is likely to behave as a nucleophile or electrophile or both to be stated.
Concept introduction:
A nucleophile (either negatively charged or a neutral molecule containing an atom with at least one lone pair of electrons) has an electron rich-site. An electrophile (either positively charged or a neutral molecule containing an electron deficient atom) has an electron poor-site.
To state:
Whether CH3CHO will behave as nucleophile or electrophile or both.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Student Value Bundle: Organic Chemistry, + OWLv2 with Student Solutions Manual eBook, 4 terms (24 months) Printed Access Card (NEW!!)
- Beer’s Law is A = εbc, where A is absorbance, ε is the molar absorptivity (which is specific to the compound and wavelength in the measurement), and c is concentration. The absorbance of a 2.31 × 10-5 M solution of a compound is 0.822 at a wavelength of 266 nm in a 1.00-cm cell. Calculate the molar absorptivity at 266 nm.arrow_forwardHow to calculate % of unknown solution using line of best fit y=0.1227x + 0.0292 (y=2.244)arrow_forwardGiven a 1,3-dicarbonyl compound, state the (condensed) formula of the compound obtaineda) if I add hydroxylamine (NH2OH) to give an isooxazole.b) if I add thiosemicarbazide (NH2-CO-NH-NH2) to give an isothiazole.arrow_forward
- Complete the following acid-base reactions and predict the direction of equilibrium for each. Justify your prediction by citing pK values for the acid and conjugate acid in each equilibrium. (a) (b) NHs (c) O₂N NH NH OH H₁PO₁arrow_forward23.34 Show how to convert each starting material into isobutylamine in good yield. ཅ ནད ཀྱི (b) Br OEt (c) (d) (e) (f) Harrow_forwardPlease help me Please use https://app.molview.com/ to draw this. I tried, but I couldn't figure out how to do it.arrow_forward
- Propose a synthesis of 1-butanamine from the following: (a) a chloroalkane of three carbons (b) a chloroalkane of four carbonsarrow_forwardSelect the stronger base from each pair of compounds. (a) H₂CNH₂ or EtzN (b) CI or NH2 NH2 (c) .Q or EtzN (d) or (e) N or (f) H or Harrow_forward4. Provide a clear arrow-pushing mechanism for each of the following reactions. Do not skip proton transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted without ambiguity. a. 2. 1. LDA 3. H3O+ HOarrow_forward
- b. H3C CH3 H3O+ ✓ H OHarrow_forward2. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases. a. CH3arrow_forwardIdentify and provide an explanation that distinguishes a qualitative and quantitative chemical analysis. Provide examples.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


