
(a)
Interpretation:
The chemical formula of the magnesium sulfate is to be stated.
Concept Introduction:
A chemical formula of a given compound is used to describe the number of atoms present in it. Since a compound is made of two or more than two different elements in definite proportion, a chemical formula can best describe the number of elements it contains with their atomic symbols.
(a)
Answer to Problem 6.57FU
The chemical formula of magnesium sulfate is MgSO4.
Explanation of Solution
Magnesium is a second group element belonging to the alkaline earth metal family. To obtain stable noble gas configurationit loses two electrons and forms Mg2+.
Sulfate ion is a polyatomic ion which is composed of one sulfur and four oxygen atoms.It carries a charge of -2.
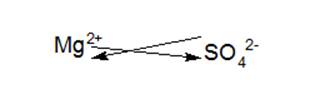
Hence, in magnesium sulfate, magnesium transfers two of its electrons to sulfate ion which results in the formation of an ionic compound known as MgSO4.
(b)
Interpretation:
The chemical formula of tin (II) fluoride is to be stated.
Concept Introduction:
A chemical formula of a given compound is used to describe the number of atoms present in it. Since a compound is made of two or more than two different elements in definite proportion, a chemical formula can best describe the number of elements it contains with their atomic symbols.
(b)
Answer to Problem 6.57FU
The chemical formula of tin (II) fluoride is SnF2.
Explanation of Solution
Tin or ‘Sn’ is a fourteen group element belonging to the Carbon family. Hence ‘Sn’looses two of its electrons to form Sn2+. Fluorine is a seventeen group element belonging to the halogens family and is non-metallic in nature therefore it gains one electron to form F-.When the chemical formula of an ionic compound is written, the superscript which denotes the charge on the cation becomes the subscript of the anion and vice-versa.
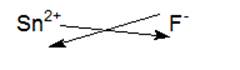
The chemical formula of the resulting compound is SnF2.
(c)
Interpretation:
The chemical formula of aluminum hydroxide is to be stated.
Concept Introduction:
A chemical formula of a given compound is used to describe the number of atoms present in it. Since a compound is made of two or more than two different elements in definite proportion, a chemical formula can best describe the number of elements it contain with their atomic symbols.
(c)
Answer to Problem 6.57FU
The chemical formula of aluminum hydroxide is Al (OH)3.
Explanation of Solution
Aluminum or ‘Al’is a thirteen group element belonging to the Boron family. After losing three electrons from its valence shell Al forms trivalent Al3+ cation. Hydroxide ion is a diatomic ion which consists of oxygen and hydrogen atom bonded covalently and it carries a negative charge. When Al loses three electrons and results in Al3+,three OH gains one electron each and results in OH- to form an ionic compound named as Al (OH)3 or commonly known as aluminum hydroxide. When the chemical formula of an ionic compound is written, the superscript which denotes the charge on the cation becomes the subscript of the anion and vice-versa.
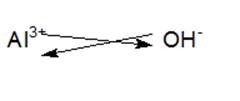
Thus, chemical formula of the resulting compound is Al (OH)3.
Want to see more full solutions like this?
Chapter 6 Solutions
Basic Chemistry (5th Edition)
- Determine the structures of the missing organic molecules in the following reaction: X+H₂O H* H+ Y OH OH Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. X Sarrow_forwardPredict the major products of this organic reaction. If there aren't any products, because nothing will happen, check the box under the drawing area instead. No reaction. HO. O :☐ + G Na O.H Click and drag to start drawing a structure. XS xs H₂Oarrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? H H C H- a -H b H Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal groups may have slightly different sizes. a = b = 0 °arrow_forward
- What are the angles a and b in the actual molecule of which this is a Lewis structure? :0: HCOH a Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal that might be caused by the fact that different electron groups may have slightly different sizes. a = 0 b=0° Sarrow_forwardDetermine the structures of the missing organic molecules in the following reaction: + H₂O +H OH O OH +H OH X Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structure of the missing organic molecule X. Click and drag to start drawing a structure.arrow_forwardIdentify the missing organic reactant in the following reaction: x + x O OH H* + ☑- X H+ O O Х Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H₂O) are not shown. In the drawing area below, draw the skeletal ("line") structure of the missing organic reactant X. Click and drag to start drawing a structure. Carrow_forward
- CH3O OH OH O hemiacetal O acetal O neither O 0 O hemiacetal acetal neither OH hemiacetal O acetal O neither CH2 O-CH2-CH3 CH3-C-OH O hemiacetal O acetal CH3-CH2-CH2-0-c-O-CH2-CH2-CH3 O neither HO-CH2 ? 000 Ar Barrow_forwardWhat would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 2 2. n-BuLi 3 Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. • Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure.arrow_forwardPredict the products of this organic reaction: NaBH3CN + NH2 ? H+ Click and drag to start drawing a structure. ×arrow_forward
- Predict the organic products that form in the reaction below: + OH +H H+ ➤ ☑ X - Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. Garrow_forwardPredict the organic products that form in the reaction below: OH H+ H+ + ☑ Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. ✓ marrow_forwardDetermine the structures of the missing organic molecules in the following reaction: + H₂O +H H+ Y Z ☑ ☑ Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X, Y, and Z. You may draw the structures in any arrangement that you like, so long as they aren't touching. Molecule X shows up in multiple steps, but you only have to draw its structure once. Click and drag to start drawing a structure. AP +arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





