
Concept explainers
Classify each transformation as substitution, elimination, or addition.
a. 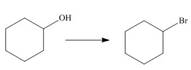 c.
c. 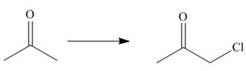
b.  d.
d. 
(a)
Interpretation: The given transformation is to be classified as substitution, elimination, or addition reaction.
Concept introduction: The substitution reactions involve the substitution or replacement of an atom or group of atoms in a compound by another atom or groups of atoms. In the substitution reactions, the replacement takes place by the break down of sigma bonds.
In elimination reaction, the formation of
Answer to Problem 6.1P
The given transformation is an example of a substitution reaction.
Explanation of Solution
The given transformation is shown as,
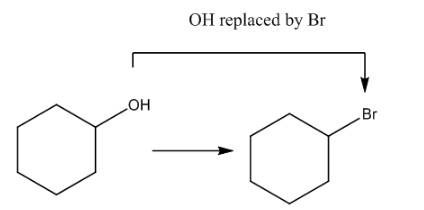
Figure 1
In the above reaction, the hydroxyl group is replaced by a bromine atom. The breakdown of the sigma bond precedes the mechanism. On replacement of the hydroxyl group, one sigma bond breaks and another is formed at the same position. Therefore, it is classified as the substitution reaction.
The given transformation is classified as a substitution reaction.
(b)
Interpretation: The given transformation is to be classified as substitution, elimination, or addition reaction.
Concept introduction: The substitution reactions involve the substitution or replacement of an atom or group of atoms in a compound by another atom or groups of atoms. In the substitution reactions, the replacement takes place by the break down of sigma bonds.
In elimination reaction, the formation of
Answer to Problem 6.1P
The given transformation is an example of an addition reaction.
Explanation of Solution
The given transformation is shown as,
Figure 2
The above reaction involves the breakdown of
The given transformation is classified as an addition reaction.
(c)
Interpretation: The given transformation is to be classified as substitution, elimination, or addition.
Concept introduction: The substitution reactions involve the substitution or replacement of an atom or group of atoms in a compound by another atom or groups of atoms. In the substitution reactions, the replacement takes place by the break down of sigma bonds.
In elimination reaction, the formation of
Answer to Problem 6.1P
The given transformation is an example of a substitution reaction.
Explanation of Solution
The given transformation is shown as,
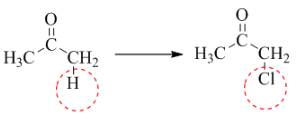
Figure 3
In the above reaction, the hydrogen is replaced by chlorine. The breakdown of the sigma bond precedes the mechanism. On the replacement of hydrogen, one sigma bond breaks and another sigma bond is formed between carbon and substituent atom.
The given transformation is classified as a substitution reaction.
(d)
Interpretation: The given transformation as substitution, elimination, or addition is to be classified.
Concept introduction: The substitution reactions involve the substitution or replacement of an atom or group of atoms in a compound by another atom or groups of atoms. In the substitution reactions, the replacement takes place by the break down of sigma bonds.
In elimination reaction, the formation of
Answer to Problem 6.1P
The given transformation is an example of an elimination reaction.
Explanation of Solution
The given transformation is shown as,
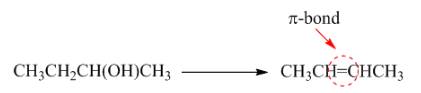
Figure 4
In this reaction, the formation of
The given transformation is classified as an elimination reaction.
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry
Additional Science Textbook Solutions
College Physics: A Strategic Approach (3rd Edition)
Cosmic Perspective Fundamentals
Biology: Life on Earth with Physiology (11th Edition)
SEELEY'S ANATOMY+PHYSIOLOGY
Loose Leaf For Integrated Principles Of Zoology
Organic Chemistry
- Please help me figure out the mechanism with arrows of the following reactionarrow_forwardOrganic Functional Groups Predicting the reactants or products of acetal hydrolysis termine the structures of the missing organic molecules in the following reaction: H* H* + H₂O Y ☑ Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X, Y, and Z. You may draw that you like, so long as they aren't touching. Molecule X shows up in multiple steps, but you only have to draw its structure Explanation Check @2 W Click and drag to start drawing a structure. #4 # 3 LU E % 67 olo 5 66 R T Y & 7 AcGraw Hill LLC. All Rights R Xarrow_forward8. (16 pts) Provide the stepwise mechanism for the synthesis of the following compound via an enaminearrow_forward
- Draw the titration curve of (i) weak acid vs. strong base; (ii) weak acid vs. weakbase; (iii) diprotic acid with strong base (iii) triprotic acid with strong base.arrow_forwardComplete the reaction in the drawing area below by adding the major products to the right-hand side. If there won't be any products, because nothing will happen under these reaction conditions, check the box under the drawing area instead. Note: if the products contain one or more pairs of enantiomers, don't worry about drawing each enantiomer with dash and wedge bonds. Just draw one molecule to represent each pair of enantiomers, using line bonds at the chiral center. More... No reaction. my ㄖˋ + 1. Na O Me Click and drag to start drawing a structure. 2. H +arrow_forwardPredict the intermediate 1 and final product 2 of this organic reaction: NaOMe H+ + 1 2 H H work up You can draw 1 and 2 in any arrangement you like. Note: if either 1 or 2 consists of a pair of enantiomers, just draw one structure using line bonds instead of 3D (dash and wedge) bonds at the chiral center. Click and drag to start drawing a structure. X $ dmarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





