![OWLv2 for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th Edition, [Instant Access], 1 term (6 months)](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
Concept explainers
6-111 As noted in Section 6-8C, the amount of external pressure that must be applied to a more concentrated solution to stop the passage of solvent molecules across a semipermeable membrane is known as the osmotic pressure 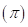 The osmotic pressure obeys a law similar in form to the
The osmotic pressure obeys a law similar in form to the 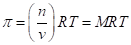 Substituting for pressure and solving for osmotic pressures gives the following equation: RT MRT, where M is the concentration or molarity of the solution.
Substituting for pressure and solving for osmotic pressures gives the following equation: RT MRT, where M is the concentration or molarity of the solution.
(a) Determine the osmotic pressure at 25°C of a 0.0020 M sucrose (C12H22O11) solution.
(b) Seawater contains 3.4 g of salts for every liter of solution. Assuming the solute consists entirely of NaCl (and complete dissociation of the NaCI salt), calculate the osmotic pressure of seawater at 25°C.
(c) The average osmotic pressure of blood is 7.7 atm at 25°C. What concentration of glucose (C6H12O6) will be isotonic with blood?
(d) Lysozyme is an enzyme that breaks bacterial cell walls. A solution containing 0.150 g of this enzyme in 210. mL of solution has an osmotic pressure of 0.953 torr at 25°C. What is the molar mass of lysozyme?
(e) The osmotic pressure of an aqueous solution of a certain protein was measured in order to determine the protein's molar mass. The solution contained 3.50 mg of protein dissolved in sufficient water to form 5.00 mL of solution. The osmotic pressure of the solution at 25°C was found to be 1.54 torr. Calculate the molar mass of the protein.
(a)
Interpretation:
The osmotic pressure of given sucrose solution should be calculated.
Concept Introduction:
Isotonic solutions are solutions in which both the solutions contain same osmolarity. Osmolarity is a term used for multiplication of molarity of the solution with numbers of each particles of the solute. It depends on one gram of solute present in 1000 grams of solution. Osmotic pressure is the pressure which is applied externally on more concentrated solution that stops the movement of solute form semipermeable membrane. Osmotic pressure follows the ideal gas law and following is the equation for it:
Where,
- Π= osmotic pressure
- M= molarity of compound
- R= the gas constant
- T= temperature in Kelvin
Answer to Problem 6.111P
The sucrose solution contains 0.049atm at
Explanation of Solution
The data given is as follow.
Temperature =
Molarity =0.0020M.
From above mentioned equation for osmotic pressure,
The osmotic pressure is 0.049atm for given sucrose solution.
(b)
Interpretation:
The osmotic pressure of seawater at
Concept Introduction:
Isotonic solutions are solutionsin which both the solutions contain same osmolarity. Osmolarity is a term used for multiplication of molarity of the solution with numbers of each particles of the solute. It depends on one gram of solute present in 1000 grams of solution. Osmotic pressure is the pressure which is applied externally on more concentrated solution that stops the movement of solute form semipermeable membrane. Osmotic pressure follows the ideal gas law and following is the equation for it:
Where,
- Π= osmotic pressure
- M= molarity of compound
- R= the gas constant
- T= temperature in Kelvin
Answer to Problem 6.111P
The osmotic pressure of given NaCl containing seawater solution is 2.845atm.
Explanation of Solution
The data given is follow,
Temperature =
NaCl=3.4g per liter.
First calculating molarity of NaCl.
For each formula, NaCl dissociate into two ions;
Molarity of NaCl solution=
For osmotic pressure of NaCl solution,
The osmotic pressure of given NaCl containing seawater solution is 2.845atm.
(c)
Interpretation:
The concentration of glucose should be calculated to make it isotonic with blood.
Concept Introduction:
Isotonic solutions are solutionsin which both the solutions contain same osmolarity. Osmolarity is a term used for multiplication of molarity of the solution with numbers of each particles of the solute. It depends on one gram of solute present in 1000 grams of solution. Osmotic pressure is the pressure which is applied externally on more concentrated solution that stops the movement of solute form semipermeable membrane. Osmotic pressure follows the ideal gas law and following is the equation for it:
Where,
- Π= osmotic pressure
- M= molarity of compound
- R= the gas constant
- T= temperature in Kelvin
Answer to Problem 6.111P
The solution should contain 0.314M of glucose to become isotonic with blood.
Explanation of Solution
The osmotic pressure of blood is 7.7atm at 298K temperature.
To find out the concentration of glucose that should be isotonic with blood,
The solution should contain 0.314M of glucose to become isotonic with blood.
(d)
Interpretation:
The molar mass of lysozymes in solution should be calculated.
Concept Introduction:
Isotonic solutions are solutionsin which both the solutions contain same osmolarity. Osmolarity is a term used for multiplication of molarity of the solution with numbers of each particles of the solute. It depends on one gram of solute present in 1000 grams of solution. Osmotic pressure is the pressure which is applied externally on more concentrated solution that stops the movement of solute form semipermeable membrane. Osmotic pressure follows the ideal gas law and following is the equation for it:
Where,
- Π= osmotic pressure
- M= molarity of compound
- R= the gas constant
- T= temperature in kelvin
Answer to Problem 6.111P
The molar mass of lysozymes is
Explanation of Solution
Osmotic pressure =0.953torr= 0.00125atm (1 torr =0.001315atm).
Temperature=298K.
Calculating the moles of lysozyme in solution,
Now calculating molar mass for lysozyme,
(e)
Interpretation:
The molar mass of protein in solution should be calculated.
Concept Introduction:
Isotonic solutions are solutionsin which both the solutions contain same osmolarity. Osmolarity is a term used for multiplication of molarity of the solution with numbers of each particles of the solute. It depends on one gram of solute present in 1000 grams of solution. Osmotic pressure is the pressure which is applied externally on more concentrated solution that stops the movement of solute form semipermeable membrane. Osmotic pressure follows the ideal gas law and following is the equation for it:
Where,
- Π= osmotic pressure
- M= molarity of compound
- R= the gas constant
- T= temperature in Kelvin
Answer to Problem 6.111P
The molar mass of protein is
Explanation of Solution
Osmotic pressure =1.54torr=0.002026atm (1 torr =0.001315atm).
Temperature=298K.
Calculating the moles of protein in solution,
Now calculating molar mass for lysozyme,
Want to see more full solutions like this?
Chapter 6 Solutions
OWLv2 for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th Edition, [Instant Access], 1 term (6 months)
- 6-74 An osmotic semipermeable membrane that allows only water to pass separates two compartments, A and B. Compartment A contains 0.9% NaCI, and compartment B contains 3% glycerol C3H8O3. (a) In which compartment will the level of solution rise? (b) Which compartment (if either) has the higher osmotic pressure?arrow_forward6-113 List the following aqueous solutions in order of decreasing freezing point: 0.040 M glycerin (C3H8O3) 0.025 M NaBr, and 0.015 M AI(NO3)3 Assume complete dissociation of any salts.arrow_forward6-60 Predict which of these covalent compounds is soluble in water. (a) C2H6 (b) CH3OH (c) HF (d) NH3 (e) CCI4arrow_forward
- 6-67 Calculate the freezing points of solutions made by dissolving 1.00 mole of each of the following ionic solutes in 1000. g of H2O. (a) NaCI (b) MgCI2 (c) (NH4)2CO3 (d) AI(HCO3)3arrow_forward6-68 If we add 175 g of ethylene glycol, C2H6O2, per 1000. g of water to a car radiator, what will be the freezing point of the solution?arrow_forward6-26 On the basis of polarity and hydrogen bonding, which solute would be the most soluble in benzene C6H6? (a) CH3OH (b) H2O (c) CH3CH2CH2CH3 (d) H2SO4arrow_forward
- 6-75 Calculate the osmolarity of each of the following solutions. (a) 0.39 M Na2CO3 (b) 0.62 M AI(NO3)3 (c) 4.2 M LiBr (d) 0.009 M K3PO4arrow_forward6-43 The label on a sparkling cider says it contains 22.0 g glucose (C6H12O6) 190. mg K+ , and 4.00 mg Na+ per serving of 240. mL of cider. Calculate the molarities of these ingredients in the sparkling cider.arrow_forward6-18 Suppose you prepare a solution by dissolving glucose in water. Which is the solvent, and which is the solute?arrow_forward
- Will red blood cells swell, remain the same size, or shrink when placed in each of the solutions in Problem 8-101? Classify each of the following solutions as hypotonic, isotonic, or hypertonic relative to red blood cells? a. 0.92%(m/v) glucose solution b. 0.92%(m/v) NaCl solution c. 2.3%(m/v) glucose solution d. 5.0%(m/v) NaCl solutionarrow_forward6-20 Give a familiar example of solutions of each of these types: (a) Liquid in liquid (b) Solid in liquid (c) Gas in liquid (d) Gas in gasarrow_forward6-32 Would you expect the solubility of ammonia gas in water at 2 atm pressure to be: (a) greater than, (b) the same as, or (c) smaller than at 0.5 atm pressure?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





