
In this chapter, we have learned about the
suppose it is hypothesized that it requires more energy to remove an electron from a metal that has atoms with one or more half-filled shells than from those that do not.
- Design a series of experiments involving the photoelectric effect that would test the hypothesis.
- What experimental apparatus would be needed to test the hypothesis? Its not necessary that you name actual equipment but rather that you imagine how the apparatus would work-think in terms of the types of measurements that would be needed, and what capability you would need in your apparatus.
- Describe the type of data you would collect and how you would analyze the data to see whether the hypothesis were correct.
- Could your experiments be extended to test the hypothesis for other parts of the periodic table, such as the lanthanide or actinide elements?
Interpretation: The experiments, apparatus and the type of data to test the given hypothesis is to be determined.
(a) A series of experiment involving the photoelectric effect that would test the hypothesis needs to be designed.
(b) The experimental apparatus required to test the given hypothesis should be determined.
(c)The type of data required to conclude whether the given hypothesis is correct or not should be determined.
(d) If the given hypothesis can be tested on lanthanides or actinides or not should be identified.
Concept Introduction: The light falls on the surface of the metal and results in the ejection of electron form its surface. This process is known as the photoelectric effect.
Answer to Problem 1DE
Solution: (a) When the light incident on the metal plate having one or more half-filled orbital; one does not observe the collection of electrons on the collector plate. But as the energy increases the electrons started collecting on the collector plate. This proves the given hypothesis.
(b) The experimental apparatus to study the photoelectric effect consists of the metal plate, collector plate, battery and voltmeter.
(c) The plot of stopping potential as a function of frequency is used to conclude that the given hypothesis is correct.
(d) The given hypothesis is not applicable on lanthanides and actinides because of the presence of d and f orbitals.
(a)
Explanation of Solution
The hypothesis says that that it requires more energy to remove an electron from the metal that has one or more half filled shells.
The ejection of electrons from the metal surface is tested by using the photoelectric effect by considering the apparatus consists of the metal plate on which the light is incident and the collector plate on which the electrons get collected. These two plates are connected to the electric circuit consists of battery, photodiode with amplifier and the voltmeter with reverse voltage.
When the light incident on the metal plate having one or more half filled orbital; one does not observed the collection of electrons on the collector plate. But as the energy increases the electrons starting collecting on the collector plate. This proves the given hypothesis.
(b)
Explanation:
The apparatus to study the photoelectric effect has the following parts,
- A photodiode with an amplifier.
- A digital voltmeter with reverse voltage.
- Batteries to operate amplifier and to provide reverse voltage.
- A monochromatic light source.
- The incident light beam intensity must adjust using a neutral filter.
The apparatus for testing the given hypothesis is shown below:
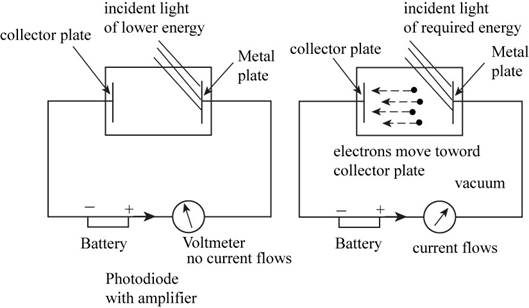
Figure 1
(c)
Explanation:
The data of frequency and wave length of different light source is collected and it is used in the apparatus of photoelectric effect. The different percentage transmission values as the function of intensity will be observed. The plot of stopping potential as a function of frequency will be observed from this data which conclude whether the hypothesis is correct or not.
(d)
Explanation:
The given hypothesis says that more energy is required to eject the electron form a metal having half filled orbitals as compared to those who have not. But the orbital of lanthanides and actinides are diffused in nature and they are larger in size. Therefore, they can easily accept and eject electrons by using lower energy radiation. Therefore, the given hypothesis cannot be tested on the lanthanides and actinides.
- The ejection of electrons is tested by using the apparatus consists of metal plate and collector plate.
- The experimental apparatus to study the photoelectric effect consists of the metal plate, collector plate, battery and voltmeter.
- The plot of stopping potential as a function of frequency is used to conclude that the given hypothesis is correct.
- The given hypothesis is not applicable on lanthanides and actinides because of the presence of d and f orbitals.
Want to see more full solutions like this?
Chapter 6 Solutions
MAST F/ CHEM: THE CENTRAL SCI CODE ALON
Additional Science Textbook Solutions
Organic Chemistry (8th Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Campbell Essential Biology with Physiology (5th Edition)
Campbell Biology (11th Edition)
Living By Chemistry: First Edition Textbook
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
- Li+ is a hard acid. With this in mind, which if the following compounds should be most soluble in water? Group of answer choices LiBr LiI LiF LiClarrow_forwardQ4: Write organic product(s) of the following reactions and show the curved-arrow mechanism of the reactions. Br MeOH OSO2CH3 MeOHarrow_forwardProvide the correct IUPAC name for the compound shown here. Reset cis- 5- trans- ☑ 4-6- 2- 1- 3- di iso tert- tri cyclo sec- oct but hept prop hex pent yl yne ene anearrow_forward
- Q6: Predict the major product(s) for the following reactions. Note the mechanism (SN1, SN2, E1 or E2) the reaction proceeds through. If no reaction takes place, indicate why. Pay attention to stereochemistry. NaCN DMF Br σ Ilm... Br H Br H H NaCN CH3OH KOtBu tBuOH NaBr H₂O LDA Et2O (CH3)2CHOH KCN DMSO NaOH H₂O, A LDA LDA Systemarrow_forwardQ7: For the following reactions, indicate the reaction conditions that would provide the indicated product in a high yield. Note the major reaction pathway that would take place (SN1, SN2, E1, or E2) Note: There may be other products that are not shown. There maybe more than one plausible pathway. Br H3C OH H3C CI ... H3C SCH2CH3 CI i SCH2CH3 ཨ་ Br System Settarrow_forwardQ2: Rank the compounds in each of the following groups in order of decreasing rate of solvolysis in aqueous acetone. OSO2CF3 OSO2CH3 OH a. b. CI Brarrow_forward
- ох 4-tert-butyl oxy cyclohex-1-ene Incorrect, 1 attempt remaining The systematic name of this compound classifies the -OR group as a substituent of the hydrocarbon, which is considered the principal functional group. The ether substituent is named with the suffix 'oxy'. The general format for the systematic name of a hydrocarbon is: [prefix/substituent] + [parent] + [functional group suffix] Substituents are listed in alphabetical order. Molecules with a chiral center will indicate the absolute configuration at the beginning of its name with the R and S notation.arrow_forward5. Compressibility (6 points total). The isothermal compressibility is a measure of how hard/easy it is to compress an object (how squishy is it?) at constant temperature. It is др defined as Br=-()=-(200²)T' (a) You might wonder why there is a negative sign in this formula. What does it mean when this quantity is positive and what does it mean when this quantity is negative? (b) Derive the formula for the isothermal compressibility of an ideal gas (it is very simple!) (c) Explain under what conditions for the ideal gas the compressibility is higher or lower, and why that makes sense.arrow_forward19. (3 pts) in Chapter 7 we will see a reaction of halocyclohexanes that requires that the halogen occupy an axial position with this in mind, would you expect cis-1-bromo-3-methylcyclohexane or trans-1-bromo-3-methylcyclohexane to be more reactive in this reaction? Briefly explain your choice using structures to support your answer. Mere-eries-cecleone) The tran-i-browse-3-methylcyclohexionearrow_forward
- Please help me calculate the undiluted samples ppm concentration. My calculations were 280.11 ppm. Please see if I did my math correctly using the following standard curve. Link: https://mnscu-my.sharepoint.com/:x:/g/personal/vi2163ss_go_minnstate_edu/EVSJL_W0qrxMkUjK2J3xMUEBHDu0UM1vPKQ-bc9HTcYXDQ?e=hVuPC4arrow_forwardProvide an IUPAC name for each of the compounds shown. (Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to commas, dashes, etc.) H₁₂C C(CH3)3 C=C H3C CH3 CH3CH2CH CI CH3 Submit Answer Retry Entire Group 2 more group attempts remaining Previous Nextarrow_forwardArrange the following compounds / ions in increasing nucleophilicity (least to most nucleophilic) CH3NH2 CH3C=C: CH3COO 1 2 3 5 Multiple Choice 1 point 1, 2, 3 2, 1, 3 3, 1, 2 2, 3, 1 The other answers are not correct 0000arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





