
ALEKS 360 ACCESS CARD F/GEN. ORG.CHEM
3rd Edition
ISBN: 9781264452545
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 5, Problem 5.60P
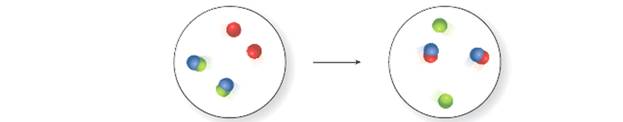
Classify the reaction depicted in the molecular art as combination, decomposition, single replacement, or double replacement.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the condensation reaction between Alanine and histidine write the amididation reaction mechanism using arrows then write the three letter code for the product of the reaction and the one letter code for the product of the reaction.
Write the amididation reaction mechanism of p-aminophenol and acetic acid to produce acetaminophen please use arrows.
Name the following using IUPAC.
Chapter 5 Solutions
ALEKS 360 ACCESS CARD F/GEN. ORG.CHEM
Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Label the reactants and products, and indicate how...Ch. 5.1 - One term in a balanced chemical equation contained...Ch. 5.2 - Write a balanced equation for each reaction....Ch. 5.2 - Write a balanced equation for the following...Ch. 5.2 - Write a balanced equation for the reaction of...Ch. 5.2 - Balance each chemical equation. Al+H2SO4Al2(...Ch. 5.3 - Write a balanced equation for the reaction...Ch. 5.3 - Classify each reaction as a combination or...
Ch. 5.3 - Classify each reaction as a single replacement or...Ch. 5.3 - Classify each reaction as a combination,...Ch. 5.3 - Fill in the needed reactants or products for each...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Prob. 5.16PCh. 5.5 - How many items are contained in one mole of (a)...Ch. 5.5 - How many carbon atoms are contained in each of the...Ch. 5.5 - How many molecules are contained in each of the...Ch. 5.5 - How many moles of water contain each of the...Ch. 5.6 - Calculate the formula weight of each ionic...Ch. 5.6 - The unmistakable odor of a freshly cut cucumber is...Ch. 5.6 - Prob. 5.23PCh. 5.6 - Calculate the number of grams contained in each of...Ch. 5.6 - How many moles are contained in each of the...Ch. 5.6 - How many molecules are contained in two 500.-mg...Ch. 5.7 - Use the balanced equation for the reaction of N2...Ch. 5.7 - Use the balanced equation in Sample Problem 5.15...Ch. 5.8 - Prob. 5.29PCh. 5.8 - Using the balanced equation for the combustion of...Ch. 5.8 - Prob. 5.31PCh. 5.8 - Use the balanced equation, N2+O22NO, to answer the...Ch. 5.9 - Prob. 5.33PCh. 5.9 - Prob. 5.34PCh. 5.9 - The synthetic antiviral drug Tamiflu, currently...Ch. 5.10 - Consider the reaction of hydrogen and nitrogen to...Ch. 5.10 - Using the balanced equation for the reaction of H2...Ch. 5.10 - Using the balanced equation, 3H2(g)+N2(g)2NH3(g),...Ch. 5.10 - Using the balanced equation, N2(g)+O2(g)2NO(g),...Ch. 5.10 - Prob. 5.41PCh. 5.10 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Prob. 5.44PCh. 5 - How many atoms of each element are drawn on each...Ch. 5 - How many atoms of each element are drawn on each...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Prob. 5.48PCh. 5 - Balance each equation. a....Ch. 5 - Balance each equation. a....Ch. 5 - Prob. 5.51PCh. 5 - Prob. 5.52PCh. 5 - Prob. 5.53PCh. 5 - Prob. 5.54PCh. 5 - For the reaction depicted in the molecular art:...Ch. 5 - Prob. 5.56PCh. 5 - Prob. 5.57PCh. 5 - Prob. 5.58PCh. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Prob. 5.67PCh. 5 - Rechargeablenickel-cadmium batteries are used in...Ch. 5 - The reaction of hydrogen (H2) with acetylene...Ch. 5 - Prob. 5.70PCh. 5 - Calculate the formula weight and molar mass of...Ch. 5 - Calculate the formula weight and molar mass of...Ch. 5 - L-Dopa is a drug used to treat Parkinson’s...Ch. 5 - Niacin, vitamin B3, is found in soybeans, which...Ch. 5 - Which quantity has the greater mass? 1 mol of Fe...Ch. 5 - Prob. 5.76PCh. 5 - Mescaline is a hallucinogen in peyote, a cactus...Ch. 5 - Prob. 5.78PCh. 5 - How many grams are contained in 5.00 mol of each...Ch. 5 - How many grams are contained in 0.50 mol of each...Ch. 5 - Prob. 5.81PCh. 5 - How many moles are contained in each number of...Ch. 5 - Prob. 5.83PCh. 5 - Prob. 5.84PCh. 5 - Prob. 5.85PCh. 5 - Prob. 5.86PCh. 5 - Using the balanced equation for the combustion of...Ch. 5 - Sodium metal (Na) reacts violently when added to...Ch. 5 - Prob. 5.89PCh. 5 - Prob. 5.90PCh. 5 - What is the percent yield of B in a reaction that...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - The reaction of methane (CH4) with Cl2forms...Ch. 5 - Methanol (CH4O), which is used as a fuel in...Ch. 5 - Consider the given reaction mixture that contains...Ch. 5 - Consider the reaction of A2 and B2 to form A2B,...Ch. 5 - Prob. 5.97PCh. 5 - Prob. 5.98PCh. 5 - Prob. 5.99PCh. 5 - Prob. 5.100PCh. 5 - The local anesthetic ethyl chloride ( C2H5Cl,...Ch. 5 - The solvent dichloromethane (, molar mass 84.93...Ch. 5 - Answer the following questions about the...Ch. 5 - Answer the following questions about diethyl ether...Ch. 5 - Prob. 5.105PCh. 5 - Prob. 5.106PCh. 5 - Prob. 5.107PCh. 5 - Prob. 5.108PCh. 5 - Prob. 5.109PCh. 5 - Prob. 5.110PCh. 5 - DDT, a pesticide that kills disease-carrying...Ch. 5 - Prob. 5.112PCh. 5 - TCDD, also called dioxin...Ch. 5 - Prob. 5.114CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
11. In the early 1800s, French naturalist Jean Baptiste Lamarck suggested that the best explanation for the rel...
Campbell Biology: Concepts & Connections (9th Edition)
Why do scientists think that all forms of life on earth have a common origin?
Genetics: From Genes to Genomes
Give the IUPAC name for each compound.
Organic Chemistry
How does the removal of hydrogen atoms from nutrient molecules result in a loss of energy from the nutrient mol...
SEELEY'S ANATOMY+PHYSIOLOGY
Some people compare DNA to a blueprint stored in the office of a construction company. Explain how this analogy...
Biology: Concepts and Investigations
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forwardName the following molecules with IUpacarrow_forward
- What is the molecular orbital for cyclopropenyl anion and is it aromatic, antiaromatic or nonaromatic?arrow_forwardUsing the chart describe the change from cystine to tyrosine and its impact on the protein. Using the chart describe the change from histidine to aspartic acid and its impact on the protein.arrow_forwardHow to get the predicted product of this reaction belowarrow_forward
- Please help me fill out the chart then using the chart describe the change from cystine to tyrosine and its impact on the protein. Then using the chart describe the change from histidine to aspartic acid.arrow_forwardWrite the Esterification reaction mechanism for acetic acid, and one propanol to make propanol ethanoate (molecule that gives peas its odor in flavor)arrow_forwardProvide solutionsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY