
Concept explainers
(a)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(a)
Explanation of Solution
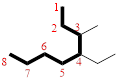
In the given compound, the longest carbon chain (highlighted with bold lines) contains EIGHT carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is OCTANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘4-ethyl-3-methyl-’
Therefore, the systematic name of the given compounds is ‘4-ethyl-3-methyloctane’.
(b)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(b)
Explanation of Solution
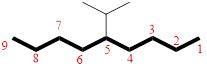
In the given compound, the longest carbon chain (highlighted with bold lines) contains NINE carbons and while numbering the parent chain, substituents should get the least possible number. The parent name is NONANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘5-isopropyl’
Therefore, the systematic name of the given compounds is ‘5-isopropylnonane’.
(c)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(c)
Explanation of Solution
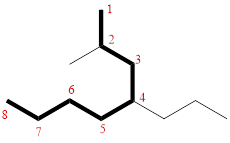
In the given compound, the longest carbon chain (highlighted with bold lines) contains EIGHT carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is OCTANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘2-methyl-4-propyl’
Therefore, the systematic name of the given compounds is ‘2-methyl-4-propyloctane’.
(d)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(d)
Explanation of Solution
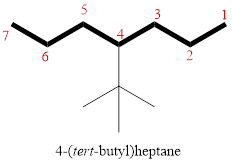
In the given compound, the longest carbon chain (highlighted with bold lines) contains SIX carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is HEXANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘4-(tert-butyl)’
Therefore, the systematic name of the given compounds is ‘4-(tert-butyl)heptane’.
(e)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(e)
Explanation of Solution
In the given compound, the longest carbon chain (highlighted with bold lines) contains ELEVEN carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is UNDECANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘5-(sec-butyl)-4-ethyl-2-methyl’
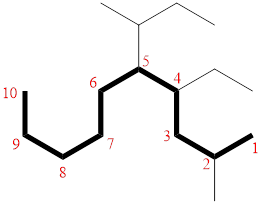
Therefore, the systematic name of the given compounds is ‘5-(sec-butyl)-4-ethyl-2-methyldecane’.
(f)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(f)
Explanation of Solution
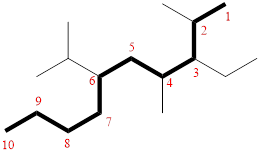
In the given compound, the longest carbon chain (highlighted with bold lines) contains TEN carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is DECANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘3-ethyl-6-isopropyl-2,4-dimethyl’
Therefore, the systematic name of the given compounds is ‘3-ethyl-6-isopropyl-2,4-dimethyl decane’.
(g)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(g)
Explanation of Solution
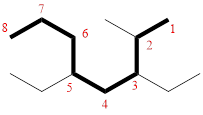
In the given compound, the longest carbon chain (highlighted with bold lines) contains EIGHT carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is OCTANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘3,5-diethyl-2-methyl’
Therefore, the systematic name of the given compounds is ‘3,5-diethyl-2-methyloctane’.
(h)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(h)
Explanation of Solution
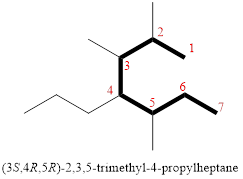
In the given compound, the longest carbon chain (highlighted with bold lines) contains SEVEN carbons and while numbering the parent chain, substituents should get the least possible number.
The parent name is HEPTANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘2,3,5-trimethyl-4-propyl’
Therefore, the systematic name of the given compounds is ‘2,3,5-trimethyl-4-propylheptane’.
(i)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Nomenclature of organic compounds:
The naming of the organic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
- The longer number of Carbon chain of a compound is identified this is called parent of the compound.
- In the cyclic compounds the number of carbon involving in ring formation is called parent of the compound.
- The compound have more than one parent chains means the larger number of substitutions present in the chain is consider as a parent chain.
- The names of all substituents are arranged by alphabets to starts with lowest numbering.
- In the complex substituent having compounds the substituent name is assigned by a name each of them based on numbers going away from the parent.
(i)
Explanation of Solution
In the given compound, the number of carbon involving in ring formation is called parent ring
(Highlighted with bold lines) contains SIX carbons and while numbering the parent ring, substituents should get the least possible number. The parent name is CYCLOHEXANE. The substituents are arranged in alphabetical order followed by the parent name.
Then as usual, the substituents are arranged in alphabetical order as ‘2,3,5,6-tetramethyl-1-propyl-’
Therefore, the systematic name of the given compounds is ‘2,3,5,6-tetramethyl-1-propyl cyclohexane’.
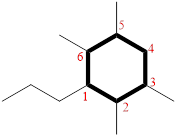
(j)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Bicyclic compounds: Compounds that contain two fused rings called bicyclic compounds.
Nomenclature of bicyclic compounds:
The organic compound naming is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Identify and the parent: The term ‘Bicylo-’ is introduced in the name of the parent. Count the number of carbons excluding the bridge heads. In the compound below, each of the three paths has two carbons. These three numbers are ordered from largest to smallest, as [2.2.2] and placed in the middle of the parent
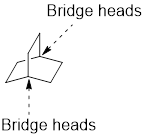
Identify and name substituents: If substituent is present, the parent must be numbered properly in order to assign the locants to the substituent. To number the parent, start at one of the bridgeheads and begin numbering along the longest path, then go to the second longest path, and finally go along the shortest path.
Arrange the substituents alphabetically.
In the complex substituent in compounds, the substituent name is assigned by a name each of them based on numbers going away from the parent.
(j)
Explanation of Solution
The cyclic system has ten carbons and the parent name is DECANE. The term ‘Bicylo-’ is introduced in the name of the parent. On Counting the number of carbons excluding the bridge heads three numbers like (4, 4, and 0) are ordered from largest to smallest, as [4.4.0] and placed in the middle of the parent as bicyclo[4.4.0]decane.
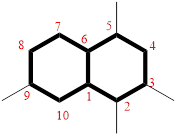
Then as usual, the substituents are arranged in alphabetical order as ‘2,3,5,9-tetramethyl’
Therefore, the systematic name of the given hydrocarbon core is 2,3,5,9-tetramethylbicyclo[4.4.0]decane.
(k)
Interpretation:
The systematic name of given compounds has to be provided.
Concept Introduction:
Bicyclic compounds: Compounds that contain two fused rings called bicyclic compounds.
Nomenclature of bicyclic compounds:
The organic compound naming is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Identify and the parent: The term ‘Bicylo-’ is introduced in the name of the parent. Count the number of carbons excluding the bridge heads. In the compound below, each of the three paths has two carbons. These three numbers are ordered from largest to smallest, as [2.2.2] and placed in the middle of the parent

Identify and name substituents: If substituent is present, the parent must be numbered properly in order to assign the locants to the substituent. To number the parent, start at one of the bridgeheads and begin numbering along the longest path, then go to the second longest path, and finally go along the shortest path.
Arrange the substituents alphabetically.
In the complex substituent in compounds, the substituent name is assigned by a name each of them based on numbers going away from the parent.
(k)
Explanation of Solution
The cyclic system has ten carbons and the parent name is OCTANE. The term ‘Bicylo-’ is introduced in the name of the parent. On Counting the number of carbons excluding the bridge heads three numbers like (2, 2, and 2) are ordered from largest to smallest, as [2.2.2] and placed in the middle of the parent as bicyclo[2.2.2]octane.
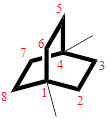
Then as usual, the substituents are arranged in alphabetical order as ‘1,4-dimethyl’
Therefore, the systematic name of the given hydrocarbon core is 1,4-dimethylbicyclo[2.2.2]octane.
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry, 3e WileyPLUS Registration Card + Loose-leaf Print Companion
- 10. Write out the mechanism (intermediate/transition state) for this reaction; indicate stereochemistry in product. H3C CH₂OH CH3 SN1 Harrow_forwardWrite "most" under the member of each trio which is most stable. Write "least under the member of each trio which is least stable. b) Draw a Fischer projection of a pair of enantiomers with three chiral carbons. Which of these two would you expect to be more soluble in water? Why? 1-butanol 1-heptanol Which of these two would you expect to have the higher boiling point? Why? hexyl methyl ether 1-heptanolarrow_forwardWrite "most" under the most acidic compound. Write "least" under the least acidic compound. OH NO₂ OCH3 Br 9. Compound X, C50H84F2, reacts with excess H2/Pd to give a C50H88F2 compound. How many rings are in X? How many double bonds are in X? Show your work.arrow_forward
- 4. State whether these two are: a) the same molecule b) c) d) different compounds that are not isomers constitutional isomers diastereomers e) enantiomers CH3 CH₁₂ H OH HO H H OH HO H CH, CH₂ 5. a) How many stereocenters does this compound have? b) How many stereoisomers are possible for this compound? CH₂ OH CHCHarrow_forwardCalculating the pH at equivalence of a titration A chemist titrates 210.0 mL of a 0.1003 M hydrobromic acid (HBr) solution with 0.7550M KOH solution at 25 °C. Calculate the pH at equivalence. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of KOH solution added. pH = ] ☑ o0o 18 Ararrow_forwardDo you do chemistry assignmentsarrow_forward
- Using the conditions of spontaneity to deduce the signs of AH and AS Use the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy AS. Note: if you have not been given enough information to decide a sign, select the "unknown" option. reaction observations conclusions A This reaction is always spontaneous, but proceeds slower at temperatures above 120. °C. ΔΗ is (pick one) AS is (pick one) ΔΗ is (pick one) B This reaction is spontaneous except above 117. °C. AS is (pick one) ΔΗ is (pick one) This reaction is slower below 20. °C than C above. AS is |(pick one) ? 18 Ar 1arrow_forwardCalculating the pH at equivalence of a titration Try Again Your answer is incorrect. 0/5 a A chemist titrates 70.0 mL of a 0.7089 M hydrocyanic acid (HCN) solution with 0.4574M KOH solution at 25 °C. Calculate the pH at equivalence. The pK of hydrocyanic acid is 9.21. Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of KOH solution added. pH = 11.43] G 00. 18 Ar B•arrow_forwardBiological Macromolecules Naming and drawing the products of aldose oxidation and reduction aw a Fischer projection of the molecule that would produce L-ribonic acid if it were subjected to mildly oxidizing reaction conditions. Click and drag to start drawing a structure. X AP ‡ 1/5 Naor Explanation Check McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibilarrow_forward
- ● Biological Macromolecules Identifying the parts of a disaccharide Take a look at this molecule, and then answer the questions in the table below it. CH2OH O H H H OH OH OH H H CH2OH H O OH H OH H H H H OH Is this a reducing sugar? Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. Explanation Check O yes X O no ○ yes O no Uarrow_forwardThe aim of the lab is to measure the sodium content from tomato sauce using the Mohr titration method. There are two groups being: Regular Tomato sauce & Salt Reduced tomato sauce QUESTION: State how you would prepare both Regular & Salt reduced tomato sauce samples for chemical analysis using the Mohr titration methodarrow_forwardUsing the conditions of spontaneity to deduce the signs of AH and AS Use the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy AS. Note: if you have not been given enough information to decide a sign, select the "unknown" option. reaction observations conclusions A The reverse of this reaction is always spontaneous but proceeds faster at temperatures above -48. °C. ΔΗ is (pick one) ✓ AS is (pick one) B This reaction is spontaneous except below 114. °C but proceeds at a slower rate below 135. °C. ΔΗ is (pick one) AS is (pick one) ΔΗ is C This reaction is exothermic and proceeds faster at temperatures above -43. °C. (pick one) AS is (pick one) v Х 5 ? 18 Ararrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





