
Concept explainers
Complete and balance the equations below, and classify them as precipitation, acid-base, gas-forming, or
(a) NiCO3 + H2SO4 →.
(b) Co(OH)2 + HBr →
(c) AgCH3CO2 + NaCI →
(d) NiO + CO →.
(a)
Interpretation:
- The given reaction has to be classified as precipitation or acid-base or gas forming reaction and the reaction has to be balanced.
- State of the product and reactant should be shown.
Concept introduction:
Precipitation reaction: The formation of the product is insoluble when the ions combine in the solution is called precipitation reaction.
Acid - base reaction: Formation of the salt from the cation from the base and anion from the acid and formation of water is also the product.
Gas forming reaction: The reaction of acid and metal carbonates which produce carbonic acid. The carbonic acid decomposes which gives water and carbon dioxide.
Oxidation - reduction reaction: The electrons are transferred to one to other is called oxidation reduction reaction.
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 56PS
The given reaction is gas forming reaction and the balanced equation is shown below.
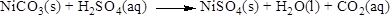
Explanation of Solution
The product of the reaction, balancing of the reaction and the type of the reaction is shown below, the given reaction is gas forming reaction and the balanced equation is shown below
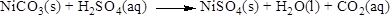
The given compound is nickel carbonate and sulfuric acid. In this reaction nickel carbonate reaction with sulfuric acid to give nickel sulfate and carbon dioxide and water.
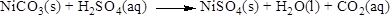
Balance the equation,
The reaction is already balanced. Therefore the balanced equation is given below.
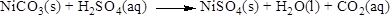
(b)
Interpretation:
- The given reaction has to be classified as precipitation or acid-base or gas forming reaction and the reaction has to be balanced.
- State of the product and reactant should be shown.
Concept introduction:
Precipitation reaction: The formation of the product is insoluble when the ions combine in the solution is called precipitation reaction.
Acid - base reaction: Formation of the salt from the cation from the base and anion from the acid and formation of water is also the product.
Gas forming reaction: The reaction of acid and metal carbonates which produce carbonic acid. The carbonic acid decomposes which gives water and carbon dioxide.
Oxidation - reduction reaction: The electrons are transferred to one to other is called oxidation reduction reaction.
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 56PS
The given reaction is acid - base reaction and the state of the reaction is shown below.

Explanation of Solution
The given reaction is acid - base reaction and the state of the reaction is shown below

The given compound is Cobalt(II) hydroxide and hydrogen bromide. In this reaction Cobalt(II) hydroxide reaction with hydrogen bromide to give cobalt(II) bromide and formation of water is also the product.
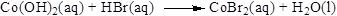
Balance the equation,
Balance the bromine atom in the given equation, when balancing the equation, we should not alter the subscripts and we can change coefficients. There are two bromine atoms in the right side and one bromine atoms in the left side. Therefore two molecule of hydrogen bromide is added to left side of reaction. Therefore the balanced equation is given below.
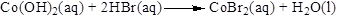
Balance the hydrogen atom in the given equation. There are four hydrogen atoms in the left side and two hydrogen atoms in the right side. Therefore two molecule of water is added to right side of reaction. Therefore the balanced equation is given below.

(c)
Interpretation:
- The given reaction has to be classified as precipitation or acid-base or gas forming reaction and the reaction has to be balanced.
- State of the product and reactant should be shown.
Concept introduction:
Precipitation reaction: The formation of the product is insoluble when the ions combine in the solution is called precipitation reaction.
Acid - base reaction: Formation of the salt from the cation from the base and anion from the acid and formation of water is also the product.
Gas forming reaction: The reaction of acid and metal carbonates which produce carbonic acid. The carbonic acid decomposes which gives water and carbon dioxide.
Oxidation - reduction reaction: The electrons are transferred to one to other is called oxidation reduction reaction.
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 56PS
The given reaction is precipitation reaction and the state of the reaction is shown below

Explanation of Solution
The given reaction is precipitation reaction and the state of the reaction is shown below

The given compound is silver acetate and sodium chloride. In this reaction silver acetate reaction with sodium chloride to give sodium acetate and silver chloride. Almost all the salts of

Balance the equation,
The reaction is already balanced. Therefore the balanced equation is given below.

(d)
Interpretation:
- The given reaction has to be classified as precipitation or acid-base or gas forming reaction and the reaction has to be balanced.
- State of the product and reactant should be shown.
Concept introduction:
Precipitation reaction: The formation of the product is insoluble when the ions combine in the solution is called precipitation reaction.
Acid - base reaction: Formation of the salt from the cation from the base and anion from the acid and formation of water is also the product.
Gas forming reaction: The reaction of acid and metal carbonates which produce carbonic acid. The carbonic acid decomposes which gives water and carbon dioxide.
Oxidation - reduction reaction: The electrons are transferred to one to other is called oxidation reduction reaction.
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 56PS
The given reaction is oxidation and reduction reaction and the state of the reaction is shown below.

Explanation of Solution
The given reaction is oxidation and reduction reaction and the state of the reaction is shown below

The given compound is nickel oxide and carbon monoxide. In this reaction nickel oxide reaction with carbon monoxide to give nickel and carbon dioxide gas. Here the oxidation state of nickel is

Balance the equation,
The reaction is already balanced. Therefore the balanced equation is given below.

Want to see more full solutions like this?
Chapter 3 Solutions
CHEMISTRY+CHEM...HYBRID ED.(LL)>CUSTOM<
- Please label this COZY spectraarrow_forwardPlease label this HNMRarrow_forwardConsider the following gas chromatographs of Compound A, Compound B, and a mixture of Compounds A and B. Inject A B mixture Area= 9 Area = 5 Area = 3 Area Inject . མི། Inject J2 What is the percentage of Compound B in the the mixture?arrow_forward
- Rank these according to stability. CH3 H3C CH3 1 CH3 H3C 1 most stable, 3 least stable O 1 most stable, 2 least stable 2 most stable, 1 least stable O2 most stable, 3 least stable O3 most stable, 2 least stable O3 most stable, 1 least stable CH3 2 CH3 CH3 H₂C CH3 3 CH3 CHarrow_forwardConsider this IR and NMR: INFRARED SPECTRUM TRANSMITTANCE 0.8- 0.6 0.4 0.2 3000 10 9 8 00 HSP-00-541 7 CO 6 2000 Wavenumber (cm-1) сл 5 ppm 4 M Which compound gave rise to these spectra? N 1000 1 0arrow_forwardConsider this reaction (molecular weights are under each compound): HC=CH + 2 HCI --> C2H4Cl 2 MW = 26 36.5 99 If 4.4 g of HC=CH are reacted with 110 mL of a 2.3 M HCI solution, and 6.0 g of product are actually produced, what is the percent yield?arrow_forward
- What is the name of the major product of this reaction? OH CH3 H₂SO4, heat 1-methylcyclohexene O2-methyl-1-cyclohexene O 3-mthylcyclohexene 1-methyl-2-cyclohexenearrow_forwardWe added a brown solution of Br2 to one of our products, and the brown color disappeared. This indicated that our product wasarrow_forwardRank the following according to reactivity toward nitration: a) benzene b) bromobenzene c) nitrobenzene d) phenol Od) greatest, c) least Od) greatest, b) least Od) greatest, a) least a) greatest, b) least a) greatest, c) least Oa) greatest, d) least Ob) greatest, a) least O b) greatest, c) least Ob) greatest, d) least O c) greatest, a) least O c) greatest, b) least O c) greatest, d) leastarrow_forward
- O-Nitrophenol was distilled over with the steam in our experiment while the other isomer did not. This is due to: O intramolecular hydrogen bonding in the ortho isomer O intermolecular hydrogen bonding in the the ortho isomer O the ortho isomer has a lower density O the ortho isomer has a lower molecular weightarrow_forwardK 44% Problem 68 of 15 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. :6: :: :CI: CI CI: :0:0 Select to Add Arrows Select to Add Arrows H H Cl CI: CI CI: Select to Add Arrows Select to Add Arrows H :CI: Alarrow_forwardI I H :0: Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. 0:0 :0: CI ΑΙ :CI: :CI: :0: CI Select to Add Arrows Select to Add Arrows cl. :0: Cl © ハ CI:: CI H CO Select to Add Arrows Select to Add Arrows 10: AI ::arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





