
Principles of Instrumental Analysis
7th Edition
ISBN: 9781337468039
Author: Skoog
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Question
Chapter 3, Problem 3.20QAP
Interpretation Introduction
Interpretation:
The given circuit is to be proved as a subtracting circuit when its four resistances are equal.
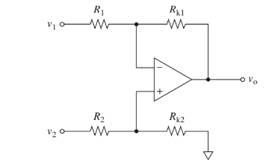
Concept introduction:
A difference amplifier configuration is here with two input signals applied at inverting and non-inverting terminals
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Steps and explanation please
Steps and explanation please
can you please draw out and list step-by-step the synthetic strategy for this rxn? thank you sm in advance
Chapter 3 Solutions
Principles of Instrumental Analysis
Ch. 3 - Prob. 3.1QAPCh. 3 - Prob. 3.2QAPCh. 3 - Prob. 3.3QAPCh. 3 - Prob. 3.4QAPCh. 3 - Prob. 3.5QAPCh. 3 - Prob. 3.6QAPCh. 3 - Prob. 3.7QAPCh. 3 - Prob. 3.8QAPCh. 3 - Prob. 3.9QAPCh. 3 - Prob. 3.10QAP
Ch. 3 - Prob. 3.11QAPCh. 3 - Prob. 3.12QAPCh. 3 - Prob. 3.13QAPCh. 3 - Prob. 3.14QAPCh. 3 - Prob. 3.15QAPCh. 3 - Prob. 3.16QAPCh. 3 - Prob. 3.17QAPCh. 3 - Prob. 3.18QAPCh. 3 - Prob. 3.19QAPCh. 3 - Prob. 3.20QAPCh. 3 - Prob. 3.21QAPCh. 3 - Prob. 3.22QAPCh. 3 - Prob. 3.23QAPCh. 3 - Prob. 3.24QAPCh. 3 - Prob. 3.25QAP
Knowledge Booster
Similar questions
- Steps and explanations pleasearrow_forwardUse diagram to answer the following: 1.Is the overall rxn endo- or exothermic. Explain briefly your answer____________________2. How many steps in this mechanism?_____________3. Which is the rate determining step? Explain briefly your answer____________________4. Identify (circle and label) the reactants,the products and intermediate (Is a Cation, Anion, or a Radical?) Please explain and provide full understanding.arrow_forwardDraw the entire mechanism and add Curved Arrows to show clearly how electrons areredistributed in the process. Please explain and provide steps clearly.arrow_forward
- Match the denticity to the ligand. Water monodentate ✓ C₂O2 bidentate H₂NCH₂NHCH2NH2 bidentate x EDTA hexadentate Question 12 Partially correct Mark 2 out of 2 Flag question Provide the required information for the coordination compound shown below: Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2✔ Geometry: linear Oxidation state of transition metal ion: +3 x in 12 correct out of 2 question Provide the required information for the coordination compound shown below. Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2 Geometry: linear 0 Oxidation state of transition metal ion: +3Xarrow_forwardCan you explain step by step behind what the synthetic strategy would be?arrow_forwardPlease explain step by step in detail the reasoning behind this problem/approach/and answer. thank you!arrow_forward
- 2. Predict the product(s) that forms and explain why it forms. Assume that any necessary catalytic acid is present. .OH HO H₂N OHarrow_forwardconsider the rate of the reaction below to be r. Whats the rate after each reaction? Br + NaCN CN + NaBr a. Double the concentration of alkyl bromide b. Halve the concentration of the electrophile & triple concentration of cyanide c. Halve the concentration of alkyl chloridearrow_forwardPredict the organic reactant that is involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning