
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
2nd Edition
ISBN: 9780077633707
Author: Janice Smith
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.103AP
Isobutyl cyanoacrylate is used in medical glues to close wounds, thus avoiding the need for sutures.
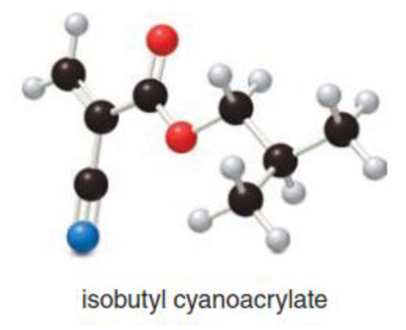
- a. Convert the ball-and-stick model to a Lewis structure with all bonds and lone pairs drawn in.
- b. Determine the shape around each carbon atom.
- c. Label all polar bonds.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Considering the position of the elements in the periodic table and their relative electronegativities and bond polarities, which bond is longest?
a. carbon - Oxygen triple bond
b. carbon - Oxygen single bond
c. carbon - Carbon single bond
d. carbon - Carbon double bond
e. carbon - Nitrogen triple bond
Which bond is the strongest?
a. carbon - Nitrogen triple bond
b. carbon - Nitrogen double bond
c. carbon - Hydrogen bond
d. carbon - Carbon triple bond
e. carbon - Carbon single bond
A. CHF
i. Best Lewis Structure
B. HNO (H is connected to one of the O's)
i. Best Lewis Structure
ii. Electron geometry on the C atom
ii. Electron geometry on the N atom
iii. Approximate bond angles about the C atom
iii. Approximate bond angles around the N atom
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the C atom.
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the N atom.
vi. Is the molecule polar or nonpolar?
vi. Is the molecule polar or nonpolar?
or ion:
1. Draw the complete Lewis structure of the molecule or ion.
2. Determine the total number of electron groups (clouds) surrounding the
central atom.
3. Determine the electron geometry of the groups around the central atom
(linear, trigonal planar, etc.)
4. Determine the molecular shape of the molecule (linear, bent, trigonal
planar, etc.)
5. Determine the molecular polarity (polar vs, nonpolar)
6. SCN-, thiocyanate ion (carbon central atom)
Lewis structure
Total number of electron groups around
the central atom
Electron geometry
Molecular shape
Molecular polarity
Chapter 3 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
Ch. 3.1 - Predict whether the bonds in the following species...Ch. 3.2 - Write the ion symbol for an atom with the given...Ch. 3.2 - Prob. 3.4PCh. 3.2 - Prob. 3.5PCh. 3.2 - How many electrons and protons are contained in...Ch. 3.2 - Prob. 3.7PCh. 3.3 - Write the formula for the ionic compound formed...Ch. 3.3 - Prob. 3.9PCh. 3.4 - Prob. 3.10PCh. 3.4 - Give the symbol for each ion. a. stannous b....
Ch. 3.4 - Name each ionic compound. a. NaF b. MgO c. SrBr2...Ch. 3.4 - Name each ionic compound. a. CrCl3 b. PbS c. SnF4...Ch. 3.4 - Prob. 3.14PCh. 3.5 - List four physical properties of ionic compounds.Ch. 3.6 - Write the formula for the compound formed when K+...Ch. 3.6 - Prob. 3.17PCh. 3.6 - Name each compound. a. Na2CO3 b. Ca(OH)2 c....Ch. 3.6 - Prob. 3.19PCh. 3.7 - Use electron-dot symbols to show how a hydrogen...Ch. 3.7 - Prob. 3.21PCh. 3.8 - Draw a Lewis structure for each covalent molecule....Ch. 3.8 - Prob. 3.23PCh. 3.8 - Prob. 3.24PCh. 3.9 - Prob. 3.25PCh. 3.9 - Prob. 3.26PCh. 3.10 - Prob. 3.27PCh. 3.11 - Prob. 3.28PCh. 3.11 - Prob. 3.29PCh. 3.11 - Show the direction of the dipole in each bond....Ch. 3.12 - Prob. 3.31PCh. 3.12 - Prob. 3.32PCh. 3 - Which formulas represent ionic compounds and which...Ch. 3 - Which pairs of elements are likely to form ionic...Ch. 3 - Prob. 3.35UKCCh. 3 - Prob. 3.36UKCCh. 3 - Prob. 3.37UKCCh. 3 - Prob. 3.38UKCCh. 3 - Prob. 3.39UKCCh. 3 - Prob. 3.40UKCCh. 3 - Prob. 3.41UKCCh. 3 - Prob. 3.42UKCCh. 3 - Prob. 3.43UKCCh. 3 - Prob. 3.44UKCCh. 3 - Prob. 3.45UKCCh. 3 - Prob. 3.46UKCCh. 3 - (a) Translate each ball-and-stick model to a Lewis...Ch. 3 - Prob. 3.48UKCCh. 3 - Prob. 3.49APCh. 3 - How many protons and electrons are present in each...Ch. 3 - Prob. 3.51APCh. 3 - Prob. 3.52APCh. 3 - Prob. 3.53APCh. 3 - Give the ion symbol for each ion. a. barium ion b....Ch. 3 - Prob. 3.65APCh. 3 - Write the formula for the ionic compound formed...Ch. 3 - Prob. 3.67APCh. 3 - Prob. 3.68APCh. 3 - Name each ionic compound. a. Na2O b. BaS c. PbS2...Ch. 3 - Name each ionic compound. a. KF b. ZnCl2 c. Cu2S...Ch. 3 - Prob. 3.71APCh. 3 - Write formulas to illustrate the difference...Ch. 3 - Prob. 3.73APCh. 3 - Name each ionic compound. a. (NH4)2SO4 b. NaH2PO4...Ch. 3 - Prob. 3.75APCh. 3 - Prob. 3.76APCh. 3 - Prob. 3.77APCh. 3 - Label each statement as true or false. Correct any...Ch. 3 - Prob. 3.79APCh. 3 - Prob. 3.80APCh. 3 - Prob. 3.81APCh. 3 - Prob. 3.82APCh. 3 - Convert the 3-D model of oxalic acid into a Lewis...Ch. 3 - Convert the 3-D model of the general anesthetic...Ch. 3 - Prob. 3.85APCh. 3 - Prob. 3.86APCh. 3 - Prob. 3.87APCh. 3 - Prob. 3.88APCh. 3 - Prob. 3.89APCh. 3 - Prob. 3.90APCh. 3 - Prob. 3.91APCh. 3 - Prob. 3.92APCh. 3 - Prob. 3.93APCh. 3 - Prob. 3.94APCh. 3 - Rank the atoms in each group in order of...Ch. 3 - Prob. 3.96APCh. 3 - Prob. 3.97APCh. 3 - Prob. 3.98APCh. 3 - Prob. 3.99APCh. 3 - Which bond in each pair is more polarthat is, has...Ch. 3 - Prob. 3.101APCh. 3 - Prob. 3.102APCh. 3 - Isobutyl cyanoacrylate is used in medical glues to...Ch. 3 - Prob. 3.104APCh. 3 - Prob. 3.105CPCh. 3 - Prob. 3.106CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the most polar bond in the molecule?arrow_forwardAnswer the following questions about the molecule OCS. a. How many valence electrons does OCS contain? b. Draw a valid Lewis structure. c. Label all polar bonds. d. What is the shape around the C atom? e. Is OCS a polar molecule? Explain.arrow_forward1. Draw the best Lewis dot structure for the anion CCl, in the correct molecular geometry (Include formal charges and lone pair electrons, and use dashed and solid wedge bonds if necessary) 2. How many electron groups are present around the central atom and what is the electron group geometry? 3. What is the molecular geometry and ideal bond angles? 4. Is the molecule polar or honpolar? If it is polar, draw a dipole moment arrow next to your structure to indicate the directionality of the dipole moment. Answers: Edit View Insert Formuat Tools Table 12pt Paragraph BIUA 2 of Bons O words IMG4Sg IMG 3449 og IMG 344 jpg IMG 3447 jp Dearrow_forward
- Answer each of the following questions correctly. Choose the correct answer. 1. It is a measure of how equally the electrons in a bond are distributed between the two atoms involved in a covalent bond. a. Polarity b. Octet rule c. Ionization energy d. Electron affinity 2. The shape of bonding molecular orbital shows that the greatest electron density is in the region. a. Between the two nuclei b. Close to the more atom electronegative c. Close to the bigger atom d. Uniformly around the two nuclei 3. In which compound is the bond that has the most ionic character found? a. HCl b. Kl c. MgS d. NOarrow_forwardI only need help with the highlighted questions please :)arrow_forwardDraw a Lewis structure for BF3 that obeys the octet rule if possible and answer the following questions based on your drawing. 1. For the central boron atom: - The number of lone pairs = ? - The number of single bonds = ? - The number of double bonds = ? 2. The central boron atom is a. obeys the octet rule. b. has an incomplete octet. c. has an expanded octet.arrow_forward
- Have each member of your group select one of the molecules and complete steps a–d. Have members present their results to the rest of the group, explaining the reasoning they used to determine their answers. CS2 NCl3 CF4 CH2F2 a. Draw the Lewis structure.b. Determine the molecular geometry and draw it accurately.c. Indicate the polarity of any polar bonds within the structure.d. Classify the molecule as polar or nonpolar.arrow_forwardFor each molecule below, a. Draw the lewis structure, using wedges and dashes to indicate the VSEPR shape as necessary. b. Draw the bond dipole for all polar bonds. c. Determine if the overall molecule is polar or nonpolar. first molecule: H₂ second molecule: CO₂ third molecule: H₂Oarrow_forwardWhich of the following statements is INCORRECT? a. Polar covalent bonds can be present in a nonpolar molecule.b. Polar covalent bond is present if the electronegativity difference between atoms is equal or less than 0.4.c. Polarity of bond and molecular geometry are the two factors that affect the polarity of molecules. d. Polar bonds forms when electrons are unequally shared by two atoms in a compound.arrow_forward
- Draw the Lewis Dot structure for N2 (on paper, not on Canvas) then answer the questions. a. How many total valence electrons are in N2? Express your answer as a whole number. b. How many single bonds are in N2? Express your answer as a whole number. c. How many double bonds are in N2? Express your answer as a whole number. d. How many triple bonds are in N2? Express your answer as a whole number.arrow_forwardThe element sulphur has atomic number 16. Sulphur forms a molecular compound with chloride with a molecular formula SCl2. The atomic number of chlorine is 17. a. Write down the electronic configuration of a sulphur atom and a chlorine atom. b. Draw a Lewis structure of an SCl2 molecule, by showing the electrons in the outer shells of the atoms. c. State how many bond pairs and lone pairs of electrons are arranged around the sulphur atoms in SCl2 molecule. d. Sketch the shape and state the molecular geometry of SCl2 molecule. e. Explain the hybridisation of the central atom in SCl2 molecule. f.Explain the intermolecular forces exists in SCl2 molecule.arrow_forwardQuestion 48 48. Which of the following statements are true? 1. 11. III. IV. The electrons in each molecule tend to orient themselves around the most electronegative element. Each molecular drawing follows the localized electron model. Both HF and CO₂ are linear molecules and therefore polar. The bond angles of NH3 are slightly less than 109.5° because the lone pair compresses the angles between the bonding pairs. a. I, III, IV b. I, II, IV c. I, II, III d. II, IV e. All of the above statements are correct. C B esc E Q @ 2 W # 3 E $ 4 * R % 5 T < 6 Y & 7 8 Uarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY