
LL ORG CHEM
6th Edition
ISBN: 9781264840083
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 58P
Researchers at Rutgers University have developed biocompatible
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
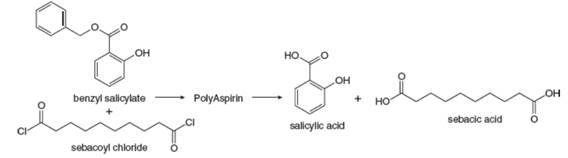
Researchers at Rutgers University have developed biocompatible polymers that degrade into nonsteroidal anti-inflammatory drugs. For example, the reaction of two equivalents of benzyl salicylate and one equivalent of sebacoyl chloride forms a poly(anhydride ester) called PolyAspirin, which hydrolyzes to salicylic acid (an anti-inflammatory agent) and sebacic acid, which is excreted. This technology can perhaps be used for localized drug delivery at specific sites of injury. What is the structure of PolyAspirin?
The plastic known as PETE (polyethyleneterephthalate) is used to make plastic soft drink bottles and containers for salad dressing, shampoos, and dishwashing liquids. PETE is a polymer of terephthalic acid and ethylene glycol. Today, PETE is the most widely recycled of all the plastics. After it is separated from other plastics, PETE can be used in polyester fabric, door mats, and tennis ball containers. In 2015, 1.84×109 lb1.84×109 lb of PETE bottles were recycled in the U.S. The density of PETE is 1.38 g/mLg/mL.
What volume, in liters, of PETE bottles were recycled in 2015 in the U.S.?
Express your answer to three significant figures.
The plastic known as PETE (polyethyleneterephthalate) is used to make plastic soft drink bottles and containers for salad dressing, shampoos, and dishwashing liquids. PETE is a polymer of terephthalic acid and ethylene glycol. Today, PETE is the most widely recycled of all the plastics. After it is separated from other plastics, PETE can be used in polyester fabric, door mats, and tennis ball containers. In 2015, 1.84×109 lb1.84×109 lb of PETE bottles were recycled in the U.S. The density of PETE is 1.38 g/mLg/mL.
Suppose a landfill holds 2.6×107 L2.6×107 L of recycled PETE. If all the PETE bottles recycled in 2015 in the U.S. were placed instead in landfills, how many landfills would be needed?
Express your answer as an integer.
Chapter 28 Solutions
LL ORG CHEM
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Draw the mechanism for the radical polymerization...Ch. 28.2 - Prob. 8PCh. 28.2 - Prob. 9PCh. 28.2 - Prob. 10PCh. 28.3 - Problem 30.12
What polymer is formed by anionic...Ch. 28.5 - Prob. 12PCh. 28.5 - Prob. 13P
Ch. 28.6 - Problem 30.15
What polyamide is formed from each...Ch. 28.6 - Prob. 19PCh. 28.7 - Prob. 20PCh. 28.8 - Prob. 21PCh. 28.9 - Prob. 22PCh. 28.9 - Prob. 23PCh. 28 - Prob. 24PCh. 28 - Prob. 25PCh. 28 - 30.30 Draw each polymer in Problem 30.29 using the...Ch. 28 - Prob. 44PCh. 28 - 30.49 Draw the products of each reaction.
a. e....Ch. 28 - Prob. 56PCh. 28 - 30.56 Compound A is a novel poly (ester amide)...Ch. 28 - 30.57 Researchers at Rutgers University have...Ch. 28 - 30.58 Melmac, a thermosetting polymer formed from...Ch. 28 - 30.59 Although chain branching in radical...Ch. 28 - Prob. 61P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The plastic known as PETE (polyethyleneterephthalate) is used to make plastic soft drink bottles and containers for salad dressing, shampoos, and dishwashing liquids. PETE is a polymer of terephthalic acid and ethylene glycol. Today, PETE is the most widely recycled of all the plastics. After it is separated from other plastics, PETE can be used in polyester fabric, door mats, and tennis ball containers. In 2015, 1.84×109 lb1.84×109 lb of PETE bottles were recycled in the U.S. The density of PETE is 1.38 g/mLg/mL. If 2.74×109 kg2.74×109 kg of PETE bottles were sold in 2015, what percentage of those bottles were recycled? Express your answer to three significant figures.arrow_forwardThe plastic known as PETE (polyethyleneterephthalate) is used to make plastic soft drink bottles and containers for salad dressing, shampoos, and dishwashing liquids. PETE is a polymer of terephthalic acid and ethylene glycol. Today, PETE is the most widely recycled of all the plastics. After it is separated from other plastics, PETE can be used in polyester fabric, door mats, and tennis ball containers. In 2015, 1.84×109 lb1.84×109 lb of PETE bottles were recycled in the U.S. The density of PETE is 1.38 g/mLg/mL. How many kilograms of PETE bottles were recycled in 2015 in the U.S.? Express your answer to three significant figures.arrow_forwardPTT (polytrimethylene terephthalate) is a polyester formed from 1,3-propanediol (HOCH 2CH 2CH 2OH) by the given reaction. PTT is sold under the trade name Sorona by DuPont. 1,3-Propanediol can now be made from a plant source like corn, rather than from petroleum starting materials. This makes PTT a more environmentally friendly polyester than PET. What is the structure of PTT?arrow_forward
- Can you explain condensation polymerization and give an example with structure of one with 2 carboxylic acid groups and 2 OH groups.arrow_forwardIn reference to cloth or fiber, the term acetate usually means cellulose acetate, a semisynthetic polymer made by treating cellulose with acetic anhydride. Cellulose acetate is spun into yarn by dissolving it in acetone or methylene chloride and forcing the solution through spinnerets into warm air, where the solvent evaporates. Predict what usually happens when students wear polyvinyl chloride shoes to the organic laboratory.arrow_forwardIn reference to cloth or fiber, the term acetate usually means cellulose acetate, a semisynthetic polymer made by treating cellulose with acetic anhydride. Cellulose acetate is spun into yarn by dissolving it in acetone or methylene chloride and forcing the solution through spinnerets into warm air, where the solvent evaporates. An organic chemistry student wore a long-sleeved acetate blouse to the laboratory. She was rinsing a warm separatory funnel with acetone when the pressure rose and blew out the stopper. Her right arm was drenched with acetone, but she was unconcerned because acetone is not very toxic. About ten minutes later, the right arm of the student’s blouse disintegrated into a pile of white fluff, leaving her with a ragged short sleeve and the tatters of a cuff remaining around her wrist. Explain how a substance as innocuous as acetone ruined the student’s blouse.arrow_forward
- Give an example of an ester polymer and cite its usearrow_forwardGlyptal® resin makes a strong, solid polymer matrix for electronic parts. Glyptal is made from terephthalic acid and glycerol. Draw the structure of Glyptal, and explain its remarkable strength and rigidity.arrow_forwardBriefly describe, using examples and sketches as appropriate, an experiment that illustrates how self-assembly can be used as a “one pot” synthesis of supramolecular polymer micellesarrow_forward
- 12. A way to produce a highly cross-linked polyester is to use glycerol. Alkyd resins are a polymer of this type. The polymer forms tough coatings when baked onto a surface and is in used in automobile paints and large appliances. Draw the structure of the polymer formed from the condensation of one glycerol and three phthalic acid molecules: H₂C-CH-CH₂ COH OH OH OH + сон Glycerol phthalic acidarrow_forwardDiscuss the manufacture of Sugar from Sugar Canearrow_forwardIllustrate the Synthesis and degradation of polymers ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Molecular spectroscopy; Author: Vidya-mitra;https://www.youtube.com/watch?v=G6HjLIWvCQo;License: Standard YouTube License, CC-BY