
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
8th Edition
ISBN: 9781305582453
Author: Brown
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2.6, Problem CQ
Interpretation Introduction
Interpretation:
The hybridization of the nitrogen atom within the ring and the nitrogen atom protruding from the ring has to be given.
Concept Introduction:
Hybridization:
When two atomic orbitals of equal energy mix together, it gives rise to two new hybridized orbitals of degenerated energy. This process is called Hybridization. Depending on the type of orbitals involved in mixing, hybridization is classified as
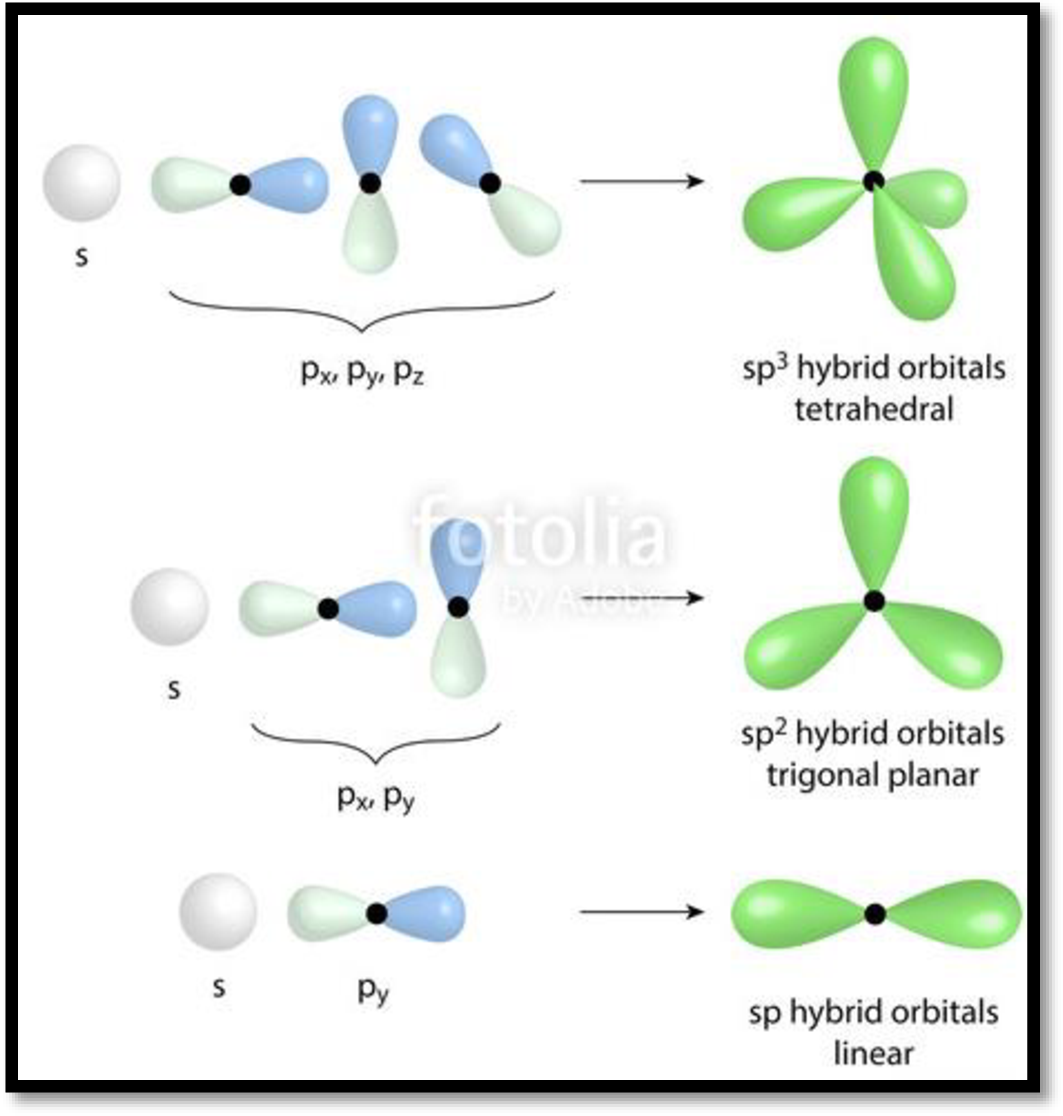
Figure 1
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For a certain gas-phase reaction at constant pressure, the equilibrium constant Kp is observed to double
when the temperature increases from 300 K to 400 K.
Calculate the enthalpy change of the reaction, Ah, using this information.
Hydrogen bonding in water plays a key role in its physical properties. Assume that the energy required to
break a hydrogen bond is approximately 8 kJ/mol. Consider a simplified two-state model where a "formed"
hydrogen bond is in the ground state and a "broken" bond is in the excited state.
Using this model:
•
Calculate the fraction of broken hydrogen bonds at T = 300 K, and also at T = 273 K and T = 373 K.
•
At what temperature would approximately 50% of the hydrogen bonds be broken?
•
What does your result imply about the accuracy or limitations of the two-state model in describing
hydrogen bonding in water?
Finally, applying your understanding:
•
Would you expect it to be easier or harder to vaporize water at higher temperatures? Why?
If you were to hang wet laundry outside, would it dry more quickly on a warm summer day or on a
cold winter day, assuming humidity is constant?
(3 pts) Use the Kapustinskii equation to calculate the lattice enthalpy for MgBr2 anddiscuss any differences between this result and that from #4.
Chapter 2 Solutions
EP ORGANIC CHEMISTRY-OWL V2 ACCESS
Ch. 2.2 - Do the line-angle formulas in each pair represent...Ch. 2.2 - Draw line-angle formulas for the three...Ch. 2.3 - Write IUPAC names for these alkanes.Ch. 2.4 - Combine the proper prefix, infix, and suffix and...Ch. 2.4 - Write the molecular formula, IUPAC name, and...Ch. 2.4 - Write molecular formulas for each bicycloalkane,...Ch. 2.4 - Prob. 2.7PCh. 2.5 - For 1,2-dichloroethane: (a) Draw Newman...Ch. 2.5 - Following is a chair conformation of cyclohexane...Ch. 2.5 - Draw the alternative chair conformation for the...
Ch. 2.5 - Draw a chair conformation of...Ch. 2.6 - Which cycloalkanes show cis, trans isomerism? For...Ch. 2.6 - Following is a planar hexagon representation for...Ch. 2.6 - Here is one cis,trans isomer of...Ch. 2.6 - Prob. AQCh. 2.6 - Prob. BQCh. 2.6 - Prob. CQCh. 2.7 - Arrange the alkanes in each set in order of...Ch. 2 - Write a line-angle formula for each condensed...Ch. 2 - Write the molecular formula of each alkane.Ch. 2 - Using parentheses and subscripts, provide an even...Ch. 2 - Which statements are true about constitutional...Ch. 2 - Prob. 2.20PCh. 2 - Each member of the following set of compounds is...Ch. 2 - Each of the following compounds is an amine...Ch. 2 - Each of the following compounds is either an...Ch. 2 - Draw structural formulas and write IUPAC names for...Ch. 2 - Draw structural formulas for all of the following....Ch. 2 - Write IUPAC names for these alkanes and...Ch. 2 - Write structural formulas and line-angle formulas...Ch. 2 - Explain why each is an incorrect IUPAC name and...Ch. 2 - For each IUPAC name, draw the corresponding...Ch. 2 - Write the IUPAC name for each compound.Ch. 2 - Prob. 2.31PCh. 2 - Torsional strain resulting from eclipsed CH bonds...Ch. 2 - How many different staggered conformations are...Ch. 2 - Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw a...Ch. 2 - Consider 1-bromo-2-methylpropane and draw the...Ch. 2 - trans-1,4-Di-tert-butylcyclohexane exists in a...Ch. 2 - From studies of the dipole moment of...Ch. 2 - Prob. 2.38PCh. 2 - Following are the alternative chair conformations...Ch. 2 - Prob. 2.40PCh. 2 - Prob. 2.41PCh. 2 - Draw line-angle formulas for the cis and trans...Ch. 2 - Name and draw structural formulas for all...Ch. 2 - Using a planar pentagon representation for the...Ch. 2 - Gibbs free energy differences between...Ch. 2 - Prob. 2.46PCh. 2 - Calculate the difference in Gibbs free energy in...Ch. 2 - Draw the alternative chair conformations for the...Ch. 2 - Use your answers from Problem 2.48 to complete the...Ch. 2 - There are four cis,trans isomers of...Ch. 2 - Draw alternative chair conformations for each...Ch. 2 - 1,2,3,4,5,6-Hexachlorocyclohexane shows cis,trans...Ch. 2 - Prob. 2.53PCh. 2 - What generalization can you make about the...Ch. 2 - What unbranched alkane has about the same boiling...Ch. 2 - Complete and balance the following combustion...Ch. 2 - Following are heats of combustion per mole for...Ch. 2 - Following are structural formulas and heats of...Ch. 2 - Without consulting tables, arrange these compounds...Ch. 2 - Which would you predict to have the larger (more...Ch. 2 - Following are structural formulas for 1,4-dioxane...Ch. 2 - Following is a planar hexagon representation of...Ch. 2 - On the left is a stereorepresentation of glucose...Ch. 2 - Prob. 2.64PCh. 2 - Prob. 2.65P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (3 pts) Silver metal adopts a fcc unit cell structure and has an atomic radius of 144 pm. Fromthis information, calculate the density of silver. Show all work.arrow_forward4. (3 pts) From the information below, determine the lattice enthalpy for MgBr2. Show all work. AH/(kJ mol-¹) Sublimation of Mg(s) +148 lonization of Mg(g) +2187 to Mg2+(g) Vaporization of Br₂(1) +31 Dissociation of Br,(g) +193 Electron gain by Br(g) -331 Formation of MgBr₂(s) -524arrow_forward1. (4 pts-2 pts each part) Consider the crystal structures of NaCl, ZnS, and CsCl (not necessarily shown in this order). a. For one of the three compounds, justify that the unit cell is consistent with stoichiometry of the compound. b. In each of the crystal structures, the cations reside in certain holes in the anions' packing structures. For each compound, what type of holes are occupied by the cations and explain why those particular types of holes are preferred.arrow_forward
- (2 pts) What do you expect to happen in a Na2O crystal if a Cl− ion replaces one of the O2−ions in the lattice?arrow_forward(2 pts) WSe2 is an ionic compound semiconductor that can be made to be p-type or n-type.What must happen to the chemical composition for it to be p-type? What must happen tothe chemical composition for it to be n-type?arrow_forward8. (2 pts) Silicon semiconductors have a bandgap of 1.11 eV. What is the longest photon wavelength that can promote an electron from the valence band to the conduction band in a silicon-based photovoltaic solar cell? Show all work. E = hv = hc/λ h = 6.626 x 10-34 Js c = 3.00 x 108 m/s 1 eV 1.602 x 10-19 Jarrow_forward
- A solution containing 100.0 mL of 0.155 M EDTA buffered to pH 10.00 was titrated with 100.0 mL of 0.0152 M Hg(ClO4)2 in a cell: calomel electrode (saturated)//titration solution/Hg(l) Given the formation constant of Hg(EDTA)2-, logKf= 21.5, and alphaY4-=0.30, find out the cell voltage E. Hg2+(aq) + 2e- = Hg(l) E0= 0.852 V E' (calomel electrode, saturated KCl) = 0.241 Varrow_forwardFrom the following reduction potentials I2 (s) + 2e- = 2I- (aq) E0= 0.535 V I2 (aq) + 2e- = 2I- (aq) E0= 0.620 V I3- (aq) + 2e- = 3I- (aq) E0= 0.535 V a) Calculate the equilibrium constant for I2 (aq) + I- (aq) = I3- (aq). b) Calculate the equilibrium constant for I2 (s) + I- (aq) = I3- (aq). c) Calculate the solubility of I2 (s) in water.arrow_forward2. (3 pts) Consider the unit cell for the spinel compound, CrFe204. How many total particles are in the unit cell? Also, show how the number of particles and their positions are consistent with the CrFe204 stoichiometry - this may or may not be reflected by the particle colors in the diagram. (HINT: In the diagram, the blue particle is in an interior position while each red particle is either in a corner or face position.)arrow_forward
- From the following potentials, calculate the activity of Cl- in saturated KCl. E0 (calomel electrode)= 0.268 V E (calomel electrode, saturated KCl)= 0.241 Varrow_forwardCalculate the voltage of each of the following cells. a) Fe(s)/Fe2+ (1.55 x 10-2 M)//Cu2+ (6.55 x 10-3 M)/Cu(s) b) Pt, H2 (0.255 bar)/HCl (4.55 x 10-4 M), AgCl (sat'd)/Ag Fe2+ +2e- = Fe E0= -0.44 V Cu2+ + 2e- = Cu E0= 0.337 V Ag+ + e- = Ag E0= 0.799 V AgCl(s) + e- = Ag(s) + Cl- E0= 0.222 V 2H+ + 2e- = H2 E0= 0.000 Varrow_forwardA solution contains 0.097 M Ce3+, 1.55x10-3 M Ce4+, 1.55x10-3 M Mn2+, 0.097 M MnO4-, and 1.00 M HClO4 (F= 9.649 x 104 C/mol). a) Write a balanced net reaction that can occur between species in this solution. b) Calculate deltaG0 and K for the reaction. c) Calculate E and deltaG for the conditions given. Ce4+ + e- = Ce3+ E0= 1.70 V MnO4- + 8H+ + 5e- = Mn2+ + 4H2O E0= 1.507 Varrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole