
Tutorials in Introductory Physics
1st Edition
ISBN: 9780130970695
Author: Peter S. Shaffer, Lillian C. McDermott
Publisher: Addison Wesley
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 25.5, Problem 1aTH
Monochromatic light from a distant point source is incident on two slits. The resulting graph of intensity versus θ is shown below. Point Y is the center of the screen; points X and Z are minima.
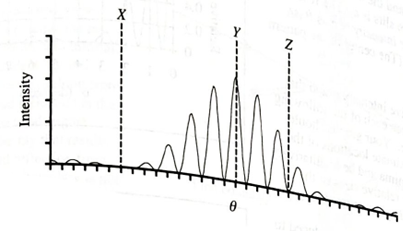
- If one of the slits in the mask were covered, would the intensity at each of the following points increase, decrease, or stay the same? Explain your reasoning in each case.
- Point X.
- Point Y.
- Point Z.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
No chatgpt pls will upvote
No chatgpt pls will upvote
No chatgpt pls will upvote
Chapter 25 Solutions
Tutorials in Introductory Physics
Ch. 25.1 - The top view diagram at right illustrates two...Ch. 25.1 - The top view diagram at right illustrates two...Ch. 25.1 - Label each nodal line and line of maximum...Ch. 25.1 - Prob. 2bTHCh. 25.1 - How do the angles a and ß compare? Explain.Ch. 25.1 - Prob. 3bTHCh. 25.1 - Prob. 3cTHCh. 25.1 - The enlarged diagram at right illustrates the...Ch. 25.1 - For what values of D (in terms of ) will there be:...Ch. 25.1 - Use your answers from parts d and e to write...
Ch. 25.1 - Determine the angles for which there will be nodal...Ch. 25.1 - Consider the following incorrect statement...Ch. 25.2 - In the space above the photograph at right,...Ch. 25.2 - The screen is 2.2m from the slits, and the...Ch. 25.2 - Suppose that the width of the right slit were...Ch. 25.2 - The graph of intensity versus angle at right...Ch. 25.3 - The photograph at right illustrates the pattern...Ch. 25.3 - The photograph at right illustrates the pattern...Ch. 25.3 - Consider the original doubleslit pattern from...Ch. 25.3 - Consider the original doubleslit pattern from...Ch. 25.3 - Consider the original doubleslit pattern from...Ch. 25.3 - Prob. 3aTHCh. 25.3 - Monochromatic light from a distant point source...Ch. 25.4 - Light from a distant point source is incident on a...Ch. 25.4 - The graph at right shows the intensity on a...Ch. 25.4 - The graph at right shows the intensity on a...Ch. 25.4 - There is a systematic way of determining the...Ch. 25.4 - There is a systematic way of determining the...Ch. 25.4 - There is a systematic way of determining the...Ch. 25.5 - Monochromatic light from a distant point source is...Ch. 25.5 - Monochromatic light from a distant point source is...Ch. 25.5 - Light from a laser (=633nm) is incident on two...Ch. 25.5 - Monochromatic light from a distant point source is...Ch. 25.5 - Monochromatic light from a distant point source is...Ch. 25.5 - Monochromatic light from a distant point source is...Ch. 25.6 - Recall the situation from tutorial, in which light...Ch. 25.6 - Recall the situation from tutorial, in which light...Ch. 25.6 - A plate of glass (n=1.5) is placed over a flat...Ch. 25.6 - A plate of glass (n=1.5) is placed over a flat...Ch. 25.6 - A plate of glass (n=1.5) is placed over a flat...Ch. 25.7 - Identical beams of light are incident on three...Ch. 25.7 - Prob. 1bTHCh. 25.7 - Unpolarized light of intensity I0 incident on a...Ch. 25.7 - Unpolarized light of intensity I0 incident on a...Ch. 25.7 - Unpolarized light of intensity I0 incident on a...Ch. 25.7 - Unpolarized light of intensity I0 incident on a...Ch. 25.7 - Unpolarized red light is incident on two...Ch. 25.7 - Unpolarized red light is incident on two...Ch. 25.7 - Unpolarized red light is incident on two...Ch. 25.7 - Unpolarized red light is incident on two...
Additional Science Textbook Solutions
Find more solutions based on key concepts
16. Mars has an atmosphere composed almost entirely of carbon dioxide, with an average temperature of –63°C. Wh...
College Physics: A Strategic Approach (3rd Edition)
3. What are serous membranes, and what are their functions?
Human Anatomy & Physiology (2nd Edition)
Analyzing ecological footprints reveals that (A) Earth's carrying capacity would increase if per capita meat co...
Campbell Biology (11th Edition)
In pea plants, plant height, seed shape, and seed color are governed by three independently assorting genes. Th...
Genetic Analysis: An Integrated Approach (3rd Edition)
What two body structures contain flexible elastic cartilage?
Anatomy & Physiology (6th Edition)
Give a molecular orbital description for each of the following: a. 1,3-pentadiene b. 1,4-pentadiene c. 1,3,5-he...
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- You are standing a distance x = 1.75 m away from this mirror. The object you are looking at is y = 0.29 m from the mirror. The angle of incidence is θ = 30°. What is the exact distance from you to the image?arrow_forwardFor each of the actions depicted below, a magnet and/or metal loop moves with velocity v→ (v→ is constant and has the same magnitude in all parts). Determine whether a current is induced in the metal loop. If so, indicate the direction of the current in the loop, either clockwise or counterclockwise when seen from the right of the loop. The axis of the magnet is lined up with the center of the loop. For the action depicted in (Figure 5), indicate the direction of the induced current in the loop (clockwise, counterclockwise or zero, when seen from the right of the loop). I know that the current is clockwise, I just dont understand why. Please fully explain why it's clockwise, Thank youarrow_forwardA planar double pendulum consists of two point masses \[m_1 = 1.00~\mathrm{kg}, \qquad m_2 = 1.00~\mathrm{kg}\]connected by massless, rigid rods of lengths \[L_1 = 1.00~\mathrm{m}, \qquad L_2 = 1.20~\mathrm{m}.\]The upper rod is hinged to a fixed pivot; gravity acts vertically downward with\[g = 9.81~\mathrm{m\,s^{-2}}.\]Define the generalized coordinates \(\theta_1,\theta_2\) as the angles each rod makes with thedownward vertical (positive anticlockwise, measured in radians unless stated otherwise).At \(t=0\) the system is released from rest with \[\theta_1(0)=120^{\circ}, \qquad\theta_2(0)=-10^{\circ}, \qquad\dot{\theta}_1(0)=\dot{\theta}_2(0)=0 .\]Using the exact nonlinear equations of motion (no small-angle or planar-pendulumapproximations) and assuming the rods never stretch or slip, determine the angle\(\theta_2\) at the instant\[t = 10.0~\mathrm{s}.\]Give the result in degrees, in the interval \((-180^{\circ},180^{\circ}]\).arrow_forward
- What are the expected readings of the ammeter and voltmeter for the circuit in the figure below? (R = 5.60 Ω, ΔV = 6.30 V) ammeter I =arrow_forwardsimple diagram to illustrate the setup for each law- coulombs law and biot savart lawarrow_forwardA circular coil with 100 turns and a radius of 0.05 m is placed in a magnetic field that changes at auniform rate from 0.2 T to 0.8 T in 0.1 seconds. The plane of the coil is perpendicular to the field.• Calculate the induced electric field in the coil.• Calculate the current density in the coil given its conductivity σ.arrow_forward
- An L-C circuit has an inductance of 0.410 H and a capacitance of 0.250 nF . During the current oscillations, the maximum current in the inductor is 1.80 A . What is the maximum energy Emax stored in the capacitor at any time during the current oscillations? How many times per second does the capacitor contain the amount of energy found in part A? Please show all steps.arrow_forwardA long, straight wire carries a current of 10 A along what we’ll define to the be x-axis. A square loopin the x-y plane with side length 0.1 m is placed near the wire such that its closest side is parallel tothe wire and 0.05 m away.• Calculate the magnetic flux through the loop using Ampere’s law.arrow_forwardDescribe the motion of a charged particle entering a uniform magnetic field at an angle to the fieldlines. Include a diagram showing the velocity vector, magnetic field lines, and the path of the particle.arrow_forward
- Discuss the differences between the Biot-Savart law and Coulomb’s law in terms of their applicationsand the physical quantities they describe.arrow_forwardExplain why Ampere’s law can be used to find the magnetic field inside a solenoid but not outside.arrow_forward3. An Atwood machine consists of two masses, mA and m B, which are connected by an inelastic cord of negligible mass that passes over a pulley. If the pulley has radius RO and moment of inertia I about its axle, determine the acceleration of the masses mA and m B, and compare to the situation where the moment of inertia of the pulley is ignored. Ignore friction at the axle O. Use angular momentum and torque in this solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning Stars and Galaxies (MindTap Course List)PhysicsISBN:9781337399944Author:Michael A. SeedsPublisher:Cengage Learning
Stars and Galaxies (MindTap Course List)PhysicsISBN:9781337399944Author:Michael A. SeedsPublisher:Cengage Learning

Glencoe Physics: Principles and Problems, Student...
Physics
ISBN:9780078807213
Author:Paul W. Zitzewitz
Publisher:Glencoe/McGraw-Hill

University Physics Volume 3
Physics
ISBN:9781938168185
Author:William Moebs, Jeff Sanny
Publisher:OpenStax

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Foundations of Astronomy (MindTap Course List)
Physics
ISBN:9781337399920
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning

Stars and Galaxies (MindTap Course List)
Physics
ISBN:9781337399944
Author:Michael A. Seeds
Publisher:Cengage Learning
Domestic Electric Circuits; Author: PrepOnGo Class 10 & 12;https://www.youtube.com/watch?v=2ZvWaloQ3nk;License: Standard YouTube License, CC-BY