
Concept explainers
(a)
Interpretation: For the given
Concept introduction:
Amines are the hydrocabons which have nitrogen
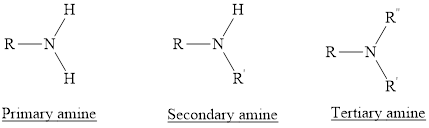
(1) Primary Amines: The nitrogen atoms is substituted by one alkyl group and two hydrogen atoms are also attached to nitrogen atom is called as primary amine. For example,
(2) Secondary Amines: The nitrogen atoms is substituted by two alkyl groups and one hydrogen atom is also attached to nitrogen atom is called as secondary amine.
(3) Tertiary Amines: The nitrogen atoms is substituted by three alkyl groups and no hydrogen atom is attached to nitrogen atom is called as tertiary amine.
(a)
Answer to Problem 47PS
The molecular formula of the given amine is
Explanation of Solution
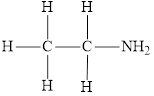
The molecular formula of the given amine can be written by using its given name ethylamine.
The ethyl group
Therefore molecular formula will have on ethyl group and two hydrogen atoms attached to nitrogen atom, that is
The structural formula for this amine is drawn as follows,

(b)
Interpretation: For the given amines structural formula has to be drawn and also their molecular formula has to be given.
Concept introduction:
Amines are the hydrocabons which have nitrogen
(1) Primary Amines: The nitrogen atoms is substituted by one alkyl group and two hydrogen atoms are also attached to nitrogen atom is called as primary amine. For example,
(2) Secondary Amines: The nitrogen atoms is substituted by two alkyl groups and one hydrogen atom is also attached to nitrogen atom is called as secondary amine.
(3) Tertiary Amines: The nitrogen atoms is substituted by three alkyl groups and no hydrogen atom is attached to nitrogen atom is called as tertiary amine.

(b)
Answer to Problem 47PS
The molecular formula of the given amine is
Explanation of Solution
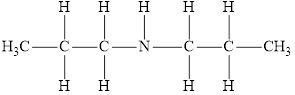
The molecular formula of the given amine can be written by using its given name dipropylamine.
The two propyl groups
It is secondary amine as two alkyl groups are attached to nitrogen atom.
Therefore molecular formula will have two propyl groups and one hydrogen atom attached to nitrogen atom, that is
The structural formula for this amine is drawn as follows,

(c)
Interpretation: For the given amines structural formula has to be drawn and also their molecular formula has to be given.
Concept introduction:
Amines are the hydrocabons which have nitrogen
(1) Primary Amines: The nitrogen atoms is substituted by one alkyl group and two hydrogen atoms are also attached to nitrogen atom is called as primary amine. For example,
(2) Secondary Amines: The nitrogen atoms is substituted by two alkyl groups and one hydrogen atom is also attached to nitrogen atom is called as secondary amine.
(3) Tertiary Amines: The nitrogen atoms is substituted by three alkyl groups and no hydrogen atom is attached to nitrogen atom is called as tertiary amine.

(c)
Answer to Problem 47PS
The molecular formula of the given amine is
Explanation of Solution
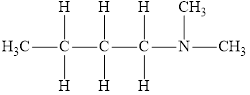
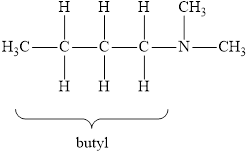
The molecular formula of the given amine can be written by using its given name butyl dimethylamine.
The two methyl groups
It is tertiary amine as three alkyl groups are attached to nitrogen atom.
Therefore molecular formula will have two methyl groups, one butyl group and no hydrogen atom attached to nitrogen atom, that is
The structural formula for this amine is drawn as follows,
(d)
Interpretation: For the given amines structural formula has to be drawn and also their molecular formula has to be given.
Concept introduction:
Amines are the hydrocabons which have nitrogen
(1) Primary Amines: The nitrogen atoms is substituted by one alkyl group and two hydrogen atoms are also attached to nitrogen atom is called as primary amine. For example,
(2) Secondary Amines: The nitrogen atoms is substituted by two alkyl groups and one hydrogen atom is also attached to nitrogen atom is called as secondary amine.
(3) Tertiary Amines: The nitrogen atoms is substituted by three alkyl groups and no hydrogen atom is attached to nitrogen atom is called as tertiary amine.

(d)
Answer to Problem 47PS
The molecular formula of the given amine is
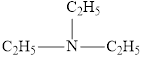
Explanation of Solution
The molecular formula of the given amine can be written by using its given name triethylamine.
The three ethyl groups
It is tertiary amine as three alkyl groups are attached to nitrogen atom.
Therefore molecular formula will have three ethyl groups, no hydrogen atom attached to nitrogen atom, that is
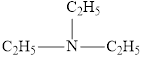
Want to see more full solutions like this?
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Write the systematic name of each organic molecule: structure HO-C-CH2-CH3 O -OH CH3-CH2-CH2-CH2-CH2-C-OH CH3 CH3-CH-CH2-C-OH Explanation Check S namearrow_forwardtheres 2 productsarrow_forwardDraw the major product of this solvolysis reaction. Ignore any inorganic byproducts. + CH3CH2OH Drawing Q Atoms, Bonds and Rings OCH2CH3 || OEt Charges OH 00-> | Undo Reset | Br Remove Done Drag To Pan +arrow_forward
- Draw the major product of this SN1 reaction. Ignore any inorganic byproducts. CH3CO2Na CH3CO2H Drawing + Br Q Atoms, Bonds and Rings OAC Charges OH ОАс Na ဂ Br Undo Reset Remove Done Drag To Pan +arrow_forwardOrganic Functional Groups entifying positions labeled with Greek letters in acids and derivatives 1/5 ssible, replace an H atom on the a carbon of the molecule in the drawing area with a ce an H atom on the ẞ carbon with a hydroxyl group substituent. ne of the substituents can't be added for any reason, just don't add it. If neither substi er the drawing area. O H OH Oneither substituent can be added. Check D 1 Accessibility ado na witharrow_forwardDifferentiate between electrophilic and nucleophilic groups. Give examples.arrow_forward
- An aldehyde/ketone plus an alcohol gives a hemiacetal, and an excess of alcohol gives an acetal. The reaction is an equilibrium; in aldehydes, it's shifted to the right and in ketones, to the left. Explain.arrow_forwardDraw a Haworth projection or a common cyclic form of this monosaccharide: H- -OH H- OH H- -OH CH₂OHarrow_forwardAnswer the question in the first photoarrow_forward
- Ggggffg2258555426855 please don't use AI Calculate the positions at which the probability of a particle in a one-dimensional box is maximum if the particle is in the fifth energy level and in the eighth energy level.arrow_forwardExplain the concepts of hemiacetal and acetal.arrow_forwardBriefly describe a nucleophilic addition.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
