
ALEKS 360 for Silberberg Chemistry: The Molecular Nature of Matter and Change
9th Edition
ISBN: 9781260477313
Author: Martin Silberberg, Patricia Amateis
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23, Problem 23.94P
Interpretation Introduction
Interpretation:
The colour of the weaker field ligand
Concept introduction:
The fact that colour depends on the ligand allows us to create a spectrochemical series, which ranks the ability of a ligand to split
The relative magnitude of
The spectrochemical series is
Relationships on an artist’s colour wheel in which complementary colors are wedges opposite each other.
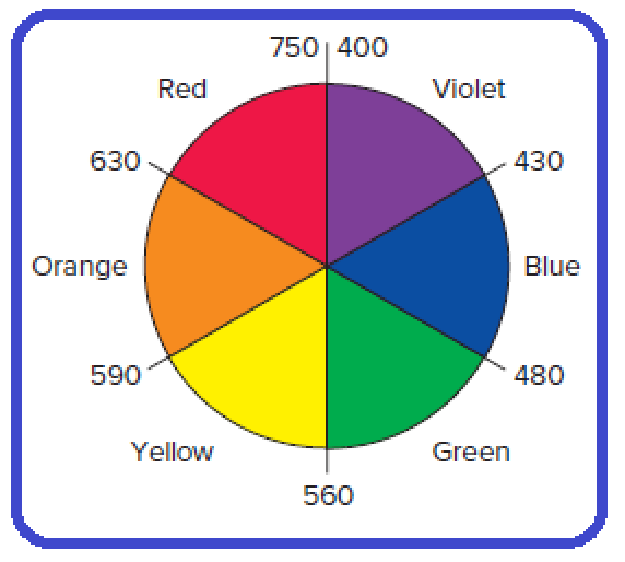
Figure 1
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structure of the pound in the provided
CO
as a 300-1200
37(2), 11 ( 110, and 2.5 (20
Please help me with # 4 and 5. Thanks in advance!
A small artisanal cheesemaker is testing the acidity of their milk
before it coagulates. During fermentation, bacteria produce lactic
acid (K₁ = 1.4 x 104), a weak acid that helps to curdle the milk and
develop flavor. The cheesemaker has measured that the developing
mixture contains lactic acid at an initial concentration of 0.025 M.
Your task is to calculate the pH of this mixture and determine whether
it meets the required acidity for proper cheese development. To
achieve the best flavor, texture and reduce/control microbial growth,
the pH range needs to be between pH 4.6 and 5.0.
Assumptions:
Lactic acid is a monoprotic acid
H
H
:0:0:
H-C-C
H
:0:
O-H
Figure 1: Lewis Structure for Lactic Acid
For simplicity, you can use the generic formula HA to represent the acid
You can assume lactic acid dissociation is in water as milk is mostly water.
Temperature is 25°C
1. Write the K, expression for the dissociation of lactic acid in the space provided. Do not forget to
include state symbols.…
Chapter 23 Solutions
ALEKS 360 for Silberberg Chemistry: The Molecular Nature of Matter and Change
Ch. 23.1 - Write partial electron configurations (no noble...Ch. 23.1 - Find the charge of each of the following ions,...Ch. 23.2 - Prob. 23.2AFPCh. 23.2 - Prob. 23.2BFPCh. 23.3 - Give the coordination number and the charge of the...Ch. 23.3 - Write the formula of the coordination compound...Ch. 23.3 - Prob. 23.4AFPCh. 23.3 - Prob. 23.4BFPCh. 23.3 - Prob. 23.5AFPCh. 23.3 - Prob. 23.5BFP
Ch. 23.4 - Prob. 23.6AFPCh. 23.4 - Prob. 23.6BFPCh. 23.4 - Prob. 23.7AFPCh. 23.4 - Prob. 23.7BFPCh. 23 - Prob. 23.1PCh. 23 - Write the general electron configuration of a...Ch. 23 - Prob. 23.3PCh. 23 - Prob. 23.4PCh. 23 - Prob. 23.5PCh. 23 - Prob. 23.6PCh. 23 - (a) What is the range in electronegativity across...Ch. 23 - (a) Explain the major difference between the...Ch. 23 - (a) What behavior distinguishes paramagnetic and...Ch. 23 - Prob. 23.10PCh. 23 - Using the periodic table to locate each element,...Ch. 23 - Using the periodic table to locate each element,...Ch. 23 - Using the periodic table to locate each element,...Ch. 23 - Prob. 23.14PCh. 23 - Prob. 23.15PCh. 23 - What is the highest oxidation state for (a) Ta;...Ch. 23 - What is the highest oxidation state for (a) Nb;...Ch. 23 - Prob. 23.18PCh. 23 - Prob. 23.19PCh. 23 - Prob. 23.20PCh. 23 - Prob. 23.21PCh. 23 - Prob. 23.22PCh. 23 - Prob. 23.23PCh. 23 - Prob. 23.24PCh. 23 - Prob. 23.25PCh. 23 - Prob. 23.26PCh. 23 - What atomic property of the lanthanides leads to...Ch. 23 - Prob. 23.28PCh. 23 - Prob. 23.29PCh. 23 - Give the electron configuration of (a) La; (b)...Ch. 23 - Prob. 23.31PCh. 23 - Only a few lanthanides show an oxidation state...Ch. 23 - Prob. 23.33PCh. 23 - Prob. 23.34PCh. 23 - Describe the makeup of a complex ion, including...Ch. 23 - Prob. 23.36PCh. 23 - Prob. 23.37PCh. 23 - Prob. 23.38PCh. 23 - Prob. 23.39PCh. 23 - Prob. 23.40PCh. 23 - Prob. 23.41PCh. 23 - Prob. 23.42PCh. 23 - Prob. 23.43PCh. 23 - Prob. 23.44PCh. 23 - Prob. 23.45PCh. 23 - Prob. 23.46PCh. 23 - What are the charge and coordination number of the...Ch. 23 - What are the charge and coordination number of the...Ch. 23 - Prob. 23.49PCh. 23 - Give systematic names for the following...Ch. 23 - What are the charge and coordination number of the...Ch. 23 - What are the charge and coordination number of the...Ch. 23 - Prob. 23.53PCh. 23 - Prob. 23.54PCh. 23 - Prob. 23.55PCh. 23 - Prob. 23.56PCh. 23 - Prob. 23.57PCh. 23 - Prob. 23.58PCh. 23 - Prob. 23.59PCh. 23 - Prob. 23.60PCh. 23 - Prob. 23.61PCh. 23 - Prob. 23.62PCh. 23 - For any of the following that can exist as...Ch. 23 - Prob. 23.64PCh. 23 - Prob. 23.65PCh. 23 - Prob. 23.66PCh. 23 - Chromium(III), like cobalt(III), has a...Ch. 23 - When MCl4(NH3)2 is dissolved in water and treated...Ch. 23 - Prob. 23.69PCh. 23 -
What is a coordinate covalent bond?
Is such a...Ch. 23 - Prob. 23.71PCh. 23 - Prob. 23.72PCh. 23 - Prob. 23.73PCh. 23 - In terms of the theory of color absorption,...Ch. 23 - Prob. 23.75PCh. 23 - Prob. 23.76PCh. 23 - Prob. 23.77PCh. 23 - How do the relative magnitudes of Epairing and Δ...Ch. 23 - Prob. 23.79PCh. 23 - Give the number of d electrons (n of dn) for the...Ch. 23 - Give the number of d electrons (n of dn) for the...Ch. 23 - Prob. 23.82PCh. 23 - How many d electrons (n of dn) are in the central...Ch. 23 - Prob. 23.84PCh. 23 - Prob. 23.85PCh. 23 - Prob. 23.86PCh. 23 - Prob. 23.87PCh. 23 - Prob. 23.88PCh. 23 - Prob. 23.89PCh. 23 - Prob. 23.90PCh. 23 - Prob. 23.91PCh. 23 - Prob. 23.92PCh. 23 - Prob. 23.93PCh. 23 - Prob. 23.94PCh. 23 - Prob. 23.95PCh. 23 - Prob. 23.96PCh. 23 - Prob. 23.97PCh. 23 - Prob. 23.98PCh. 23 - When neptunium (Np) and plutonium (Pu) were...Ch. 23 - Prob. 23.100PCh. 23 - Prob. 23.101PCh. 23 - For the compound [Co(en)2Cl2]Cl, give:
The...Ch. 23 - Prob. 23.103PCh. 23 - Prob. 23.104PCh. 23 - Prob. 23.105PCh. 23 - Prob. 23.106PCh. 23 - Prob. 23.107PCh. 23 - Prob. 23.108PCh. 23 - Prob. 23.109PCh. 23 - Prob. 23.110PCh. 23 - Prob. 23.111PCh. 23 - The actinides Pa, U, and Np form a series of...Ch. 23 - Prob. 23.113PCh. 23 - Prob. 23.114PCh. 23 - Prob. 23.115PCh. 23 - Prob. 23.116PCh. 23 - Prob. 23.117PCh. 23 - Prob. 23.118PCh. 23 - Prob. 23.119PCh. 23 - Prob. 23.120PCh. 23 - Prob. 23.121PCh. 23 - Prob. 23.122P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. :0: :0 H. 0:0 :0: :6: S: :0: Select to Edit Arrows ::0 Select to Edit Arrows H :0: H :CI: Rotation Select to Edit Arrows H. < :0: :0: :0: S:arrow_forward3:48 PM Fri Apr 4 K Problem 4 of 10 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Mg. :0: Select to Add Arrows :0: :Br: Mg :0: :0: Select to Add Arrows Mg. Br: :0: 0:0- Br -190 H 0:0 Select to Add Arrows Select to Add Arrows neutralizing workup H CH3arrow_forwardIarrow_forward
- Draw the Markovnikov product of the hydrobromination of this alkene. Note for advanced students: draw only one product, and don't worry about showing any stereochemistry. Drawing dash and wedge bonds has been disabled for this problem. + Explanation Check 1 X E 4 1 1 1 1 1 HBr Click and drag to start drawing a structure. 80 LE #3 @ 2 $4 0 I அ2 % 85 F * K M ? BH 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center & 6 27 FG F10 8 9 R T Y U D F G H P J K L Z X C V B N M Q W A S H option command H command optiarrow_forwardBe sure to use wedge and dash bonds to show the stereochemistry of the products when it's important, for example to distinguish between two different major products. Predict the major products of the following reaction. Explanation Q F1 A Check F2 @ 2 # 3 + X 80 F3 W E S D $ 4 I O H. H₂ 2 R Pt % 05 LL ee F6 F5 T <6 G Click and drag to start drawing a structure. 27 & A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Acce Y U H DII 8 9 F10 4 J K L Z X C V B N M T H option command F11 P H commandarrow_forwardCurved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the arrows to draw the intermediate and product in this reaction or mechanistic step(s). Include all lone pairs and charges as appropriate. Ignore stereochemistry. Ignore inorganic byproducts. H :0: CH3 O: OH Q CH3OH2+ Draw Intermediate protonation CH3OH CH3OH nucleophilic addition H Draw Intermediate deprotonation :0: H3C CH3OH2* protonation H 0: H CH3 H.arrow_forward
- Predicting the reactants or products of hemiacetal and acetal formation uentify the missing organic reactants in the following reaction: H+ X+Y OH H+ за Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H2O) are not shown. In the drawing area below, draw the skeletal ("line") structures of the missing organic reactants X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Explanation Check Click and drag to start drawing a structure. ? olo 18 Ar © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forwardcan someone please answer thisarrow_forwardPlease, please help me figure out the the moles, molarity and Ksp column. Step by step details because I've came up with about three different number and have no idea what I'm doing wrong.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY