
Concept explainers
(a)
Interpretation:
Condensed structural formula for the given set of compounds has to be written.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of functional group if any in the molecule. It can be an
Root word represents the longest continuous carbon skeleton of the organic molecule.
The structure of a molecule can be drawn by analyzing the presence of prefix, suffix and root word in the given IUPAC name.
Condensed structural formula is representation of the organic compound. In this the lengthy carbon chain is shown only with the carbon atoms (along with the hydrogen) without any bonds. The additional branches are shown with explicit bonds.
To Write: The condensed structural formula for the given compounds.
(a)
Answer to Problem 23.39QP
The condensed structural formula for 2,5-dimethyloctane is,
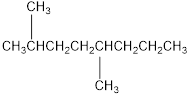
Explanation of Solution
From the given name of the hydrocarbon, the parent carbon is identified as octane. Octane has eight carbon chain. In the given name it is given as two methyl groups are substituted in second and fifth carbon atom resulting in the skeleton of the hydrocarbon.
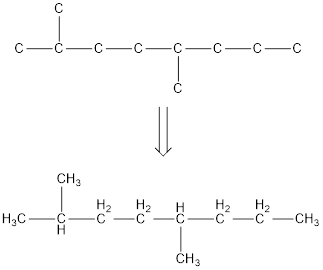
Remove the single bonds in the lengthy chain to obtain the condensed structural formula.
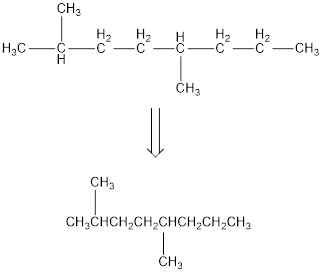
(b)
Interpretation:
Condensed structural formula for the given set of compounds has to be written.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of functional group if any in the molecule. It can be an alkene, alkyne, alcohol, carboxylic acid, alcohol etc.
Root word represents the longest continuous carbon skeleton of the organic molecule.
The structure of a molecule can be drawn by analyzing the presence of prefix, suffix and root word in the given IUPAC name.
Condensed structural formula is representation of the organic compound. In this the lengthy carbon chain is shown only with the carbon atoms (along with the hydrogen) without any bonds. The additional branches are shown with explicit bonds.
To Write: The condensed structural formula for the given compounds.
(b)
Answer to Problem 23.39QP
The condensed structural formula for 4-ethyl-2-methylheptane is,
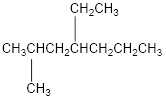
Explanation of Solution
From the given name of the hydrocarbon, the parent carbon is identified as heptane. Heptane has seven carbon chain. In the given name, it is given as a methyl group and ethyl group are substituted in fourth and second carbon atom respectively, resulting in the skeleton of the hydrocarbon.
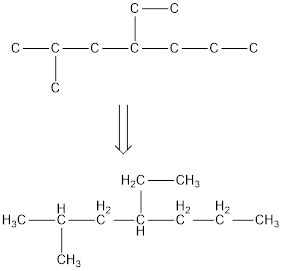
Remove the single bonds in the lengthy chain to obtain the condensed structural formula.
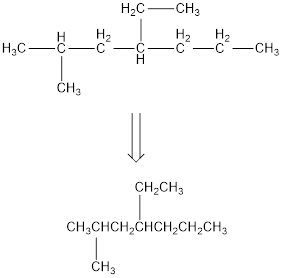
(c)
Interpretation:
Condensed structural formula for the given set of compounds has to be written.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of functional group if any in the molecule. It can be an alkene, alkyne, alcohol, carboxylic acid, alcohol etc.
Root word represents the longest continuous carbon skeleton of the organic molecule.
The structure of a molecule can be drawn by analyzing the presence of prefix, suffix and root word in the given IUPAC name.
Condensed structural formula is representation of the organic compound. In this the lengthy carbon chain is shown only with the carbon atoms (along with the hydrogen) without any bonds. The additional branches are shown with explicit bonds.
To Write: The condensed structural formula for the given compounds.
(c)
Answer to Problem 23.39QP
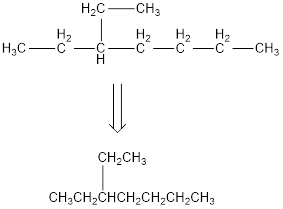
The condensed structural formula for 3-ethylheptane is,
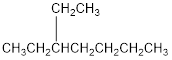
Explanation of Solution
From the given name of the hydrocarbon, the parent carbon is identified as heptane. Heptane has seven carbon chain. In the given name it is given as an ethyl group is substituted in third carbon atom resulting in the skeleton of the hydrocarbon.
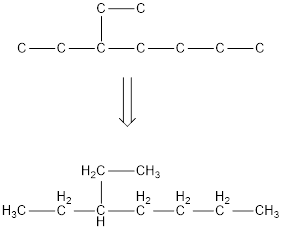
Remove the single bonds in the lengthy chain to obtain the condensed structural formula.
(d)
Interpretation:
Condensed structural formula for the given set of compounds has to be written.
Concept Introduction:
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry). IUPAC name consists of three parts in major namely Prefix suffix and root word.
Prefix represents the substituent present in the molecule and its position in the root name.
Suffix denotes the presence of functional group if any in the molecule. It can be an alkene, alkyne, alcohol, carboxylic acid, alcohol etc.
Root word represents the longest continuous carbon skeleton of the organic molecule.
The structure of a molecule can be drawn by analyzing the presence of prefix, suffix and root word in the given IUPAC name.
Condensed structural formula is representation of the organic compound. In this the lengthy carbon chain is shown only with the carbon atoms (along with the hydrogen) without any bonds. The additional branches are shown with explicit bonds.
To Write: The condensed structural formula for the given compounds.
(d)
Answer to Problem 23.39QP
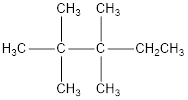
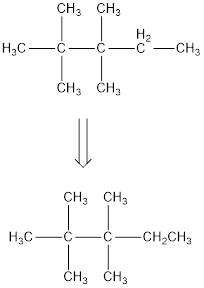
The condensed structural formula for 2,2,3,3-tetramethylpentane is,
Explanation of Solution
From the given name of the hydrocarbon, the parent carbon is identified as pentane. Pentane has five carbon chain. In the given name it is given as two methyl groups are substituted in second carbon and two methyl groups are substituted in third carbon atom resulting in the skeleton of the hydrocarbon.
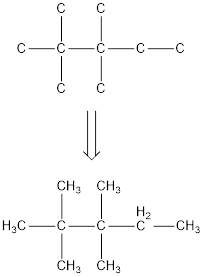
Remove the single bonds in the lengthy chain to obtain the condensed structural formula.
Want to see more full solutions like this?
Chapter 23 Solutions
Bundle: General Chemistry, Loose-Leaf Version, 11th + LabSkills PreLabs v2 for Organic Chemistry (powered by OWLv2), 4 terms (24 months) Printed ... for Ebbing/Gammon's General Chemistry, 11th
- Please predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forwardBriefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forwardElectrochemistry. Briefly describe the Gibbs model and the Gibbs absorption equation.arrow_forward
- Briefly state the electrocapillary equation for ideally polarized electrodes.arrow_forwardWhat is surface excess according to the Gibbs model?arrow_forwardUsing Benzene as starting materid show how each of the Following molecules Contel Ve syntheswed CHI 9. b -50311 с CHY 503H Ночто d. อ •NOV e 11-0-650 NO2arrow_forward
- The molecule PYRIDINE, 6th electrons and is therefore aromatre and is Assigned the Following structure contering Since aromatk moleculoy undergo electrophilic anomatic substitution, Pyridine shodd undergo The Following reaction + HNO3 12504 a. write all of the possible Mononitration Products that could Result From this reaction 18. Bared upon the reaction mechanison determime which of these producty would be the major Product of the hegetionarrow_forwarda. Explain Why electron withdrawing groups tend to be meta-Directors. Your answer Should lyclude all apropriate. Resonance contributing Structures fo. Explain why -ll is an outho -tura drccton even though chlorine has a very High Electronegativityarrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward

 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





