
(a)
To determine: The experiment to confirm the formation of
(a)
Answer to Problem 1DE
Solution: The reaction of the given product with
Explanation of Solution
The reaction of the given coordination compound with the solution of
Whereas, if the compound formed is
This will confirm the formation of desired product.
The reaction of the given product with
(b)
To determine: If the cobalt present in the given reaction is in
(b)
Answer to Problem 1DE
Solution: The oxidation state of cobalt is
Explanation of Solution
The oxidation state of cobalt in
Substitute the values of oxidation state of ethylene diammine, chlorine and the total charge on the molecule in the above equation,
Therefore, the oxidation state of cobalt in
The electronic configuration of
The ethylene diammine present in the given complex is a strong field ligand which causes pairing of electrons in metal ions.
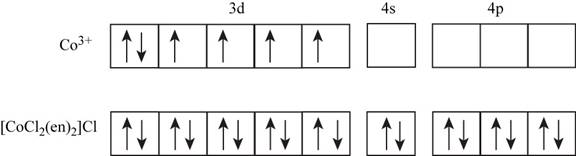
Figure 1
Since, there is no unpaired electron present in the given complex. Therefore, the spin state of the cobalt is zero in the given complex.
The oxidation state of cobalt is
(c)
To determine: The existence of geometrical isomerism in the complex
(c)
Answer to Problem 1DE
Solution: The geometrical isomerism exists in the given complex and the isomers are of different colors which confirm their presence in the mixture.
Explanation of Solution
The geometrical isomerism is the isomerism in which the structure and molecular formula of the molecule is same but the orientation of groups around the metal atoms changes.
The molecule
The geometrical isomers of the
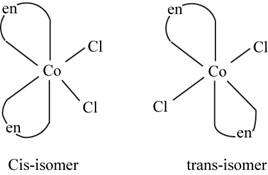
Figure 2
The isomers of
The geometrical isomerism exists in the given complex and the isomers are of different colors which confirm their presence in the mixture.
(d)
To determine: The way to know the isomer which is formed as a product in the given reaction out of the two isomers.
(d)
Answer to Problem 1DE
Solution: The color of trans isomer is green and of cis isomer is violet which gives the confirmation of the geometry of product.
Explanation of Solution
The color of cis isomer of
The color of trans isomer is green and of cis isomer is violet which gives the confirmation of the geometry of product.
Want to see more full solutions like this?
Chapter 23 Solutions
Laboratory Experiments For Chemistry: The Central Science, Si Edition
- So, the first image is what I'm trying to understand regarding my approach. The second image illustrates my teacher's method, and the third image includes my notes on the concepts behind these types of problems.arrow_forwardHAND DRAWarrow_forwardDraw a mental model for calcium chloride mixed with sodium phosphatearrow_forward
- here is my question (problem number 20) please explain to me thanks!arrow_forwardThe bromination of anisole is an extremely fast reaction. Complete the resonance structures of the intermediate arenium cation for the reaction (Part 1), and then answer the question that follows (Part 2).arrow_forwardDrawing of 3-fluro-2methylphenolarrow_forward
- Which compound(s) will be fully deprotonated (>99%) by reaction with one molar equivalent of sodium hydroxide? I, II, III I, || I, III I only II, III SH | H3C-C=C-H || III NH2arrow_forwardWill NBS (and heat or light) work for this reaction, or do we have to use Br2?arrow_forwardHAND DRAWarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





