
Chemistry: Structures and Properties, Books a la Carte Plus MasteringChemistry with eText -- Access Card Package
1st Edition
ISBN: 9780321974617
Author: Nivaldo J. Tro
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 22, Problem 92E
Interpretation Introduction
Interpretation:
The structure of Saran
Concept introduction:
A polymer is a substance with very high molecular mass in which each constituent unit is arranged in a regular manner with the help of covalent bonds.
The simplest unit of a polymer is known as the “monomer”. The reaction through which the monomers get converted into larger unit of a polymer is called as “
Given:
Saran polymer is made up of two monomers- vinylidene chloride and vinyl chloride as follows:
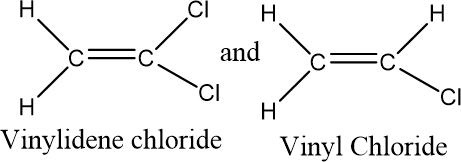
The monomers are added alternately to form the Saran polymer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please sirrr soollveee these parts pleaseeee and thank youuuuu
Please sirrr soollveee these parts pleaseeee and thank youuuuu, don't solve it by AI plleeaasseee
Please sirrr soollveee these parts pleaseeee and thank youuuuu
Chapter 22 Solutions
Chemistry: Structures and Properties, Books a la Carte Plus MasteringChemistry with eText -- Access Card Package
Ch. 22 - Prob. 1SAQCh. 22 - Prob. 2SAQCh. 22 - Prob. 3SAQCh. 22 - Prob. 4SAQCh. 22 - Prob. 5SAQCh. 22 - Prob. 6SAQCh. 22 - Prob. 7SAQCh. 22 - Prob. 8SAQCh. 22 - Prob. 9SAQCh. 22 - Prob. 10SAQ
Ch. 22 - Prob. 1ECh. 22 - Prob. 2ECh. 22 - Prob. 3ECh. 22 - Prob. 4ECh. 22 - Prob. 5ECh. 22 - Prob. 6ECh. 22 - Prob. 7ECh. 22 - What are the main classifications of hydrocarbons?...Ch. 22 - Prob. 9ECh. 22 - What are structural isomers? How do the properties...Ch. 22 - Prob. 11ECh. 22 - Define each term related to optical isomerism:...Ch. 22 - Prob. 13ECh. 22 - Prob. 14ECh. 22 - Explain geometric isomerism in alkenes. How do the...Ch. 22 - Prob. 16ECh. 22 - Prob. 17ECh. 22 - Prob. 18ECh. 22 - Prob. 19ECh. 22 - Prob. 20ECh. 22 - Prob. 21ECh. 22 - Prob. 22ECh. 22 - Prob. 23ECh. 22 - Prob. 24ECh. 22 - Prob. 25ECh. 22 - Prob. 26ECh. 22 - Prob. 27ECh. 22 - Prob. 28ECh. 22 - Prob. 29ECh. 22 - Prob. 30ECh. 22 - Prob. 31ECh. 22 - Prob. 32ECh. 22 - Prob. 33ECh. 22 - How do an addition polymer and a condensation...Ch. 22 - Prob. 35ECh. 22 - Prob. 36ECh. 22 - Prob. 37ECh. 22 - Prob. 38ECh. 22 - Prob. 39ECh. 22 - Prob. 40ECh. 22 - Prob. 41ECh. 22 - Prob. 42ECh. 22 - Name each alkane.Ch. 22 - Prob. 44ECh. 22 - Prob. 45ECh. 22 - Draw a structure for each alkane. a....Ch. 22 - Prob. 47ECh. 22 - Complete and balance each hydrocarbon combustion...Ch. 22 - Prob. 49ECh. 22 - Prob. 50ECh. 22 - Prob. 51ECh. 22 - Prob. 52ECh. 22 - Name each alkene.Ch. 22 - Prob. 54ECh. 22 - Prob. 55ECh. 22 - Name each alkyne.Ch. 22 - Draw the correct structure for each compound. a....Ch. 22 - Prob. 58ECh. 22 - Prob. 59ECh. 22 - Prob. 60ECh. 22 - Prob. 61ECh. 22 - Prob. 62ECh. 22 - Prob. 63ECh. 22 - Prob. 64ECh. 22 - Prob. 65ECh. 22 - Prob. 66ECh. 22 - Prob. 67ECh. 22 - Prob. 68ECh. 22 - Prob. 69ECh. 22 - Prob. 70ECh. 22 - Prob. 71ECh. 22 - Prob. 72ECh. 22 - Prob. 73ECh. 22 - Prob. 74ECh. 22 - Prob. 75ECh. 22 - Prob. 76ECh. 22 - Prob. 77ECh. 22 - Prob. 78ECh. 22 - Prob. 79ECh. 22 - Prob. 80ECh. 22 - Prob. 81ECh. 22 - Prob. 82ECh. 22 - Prob. 83ECh. 22 - Prob. 84ECh. 22 - Prob. 85ECh. 22 - Prob. 86ECh. 22 - Prob. 87ECh. 22 - Prob. 88ECh. 22 - Prob. 89ECh. 22 - Prob. 90ECh. 22 - Prob. 91ECh. 22 - Prob. 92ECh. 22 - Prob. 93ECh. 22 - Prob. 94ECh. 22 - Identify each organic compound as an alkane,...Ch. 22 - Identify each organic compound as an alkane,...Ch. 22 - Prob. 97ECh. 22 - Prob. 98ECh. 22 - Prob. 99ECh. 22 - Determine whether each pair of structures are...Ch. 22 - Prob. 101ECh. 22 - Prob. 102ECh. 22 - Classify each organic reaction as combustion,...Ch. 22 - Prob. 104ECh. 22 - Prob. 105ECh. 22 - Prob. 106ECh. 22 - Prob. 107ECh. 22 - Prob. 108ECh. 22 - Prob. 109ECh. 22 - Prob. 110ECh. 22 - Prob. 111ECh. 22 - Prob. 112ECh. 22 - Prob. 113ECh. 22 - Prob. 114ECh. 22 - Prob. 115ECh. 22 - Prob. 116ECh. 22 - Prob. 117ECh. 22 - Prob. 118ECh. 22 - Prob. 119ECh. 22 - Prob. 120ECh. 22 - Draw the structure and name a compound with the...Ch. 22 - Prob. 122E
Knowledge Booster
Similar questions
- 4. Read paragraph 4.15 from your textbook, use your calculated lattice energy values for CuO, CuCO3 and Cu(OH)2 an explain thermal decomposition reaction of malachite: Cu2CO3(OH)2 →2CuO + H2O + CO2 (3 points)arrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forwardIII O Organic Chemistry Using wedges and dashes in skeletal structures Draw a skeletal ("line") structure for each of the molecules below. Be sure your structures show the important difference between the molecules. key O O O O O CHON Cl jiii iiiiiiii You can drag the slider to rotate the molecules. Explanation Check Click and drag to start drawing a structure. Q Search X G ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Use F 3 W C 3/5arrow_forward
- 3. Use Kapustinskii's equation and data from Table 4.10 in your textbook to calculate lattice energies of Cu(OH)2 and CuCO3 (4 points)arrow_forward2. Copper (II) oxide crystalizes in monoclinic unit cell (included below; blue spheres 2+ represent Cu²+, red - O²-). Use Kapustinski's equation (4.5) to calculate lattice energy for CuO. You will need some data from Resource section of your textbook (p.901). (4 points) CuOarrow_forwardWhat is the IUPAC name of the following compound? OH (2S, 4R)-4-chloropentan-2-ol O (2R, 4R)-4-chloropentan-2-ol O (2R, 4S)-4-chloropentan-2-ol O(2S, 4S)-4-chloropentan-2-olarrow_forward
- Use the reaction coordinate diagram to answer the below questions. Type your answers into the answer box for each question. (Watch your spelling) Energy A B C D Reaction coordinate E A) Is the reaction step going from D to F endothermic or exothermic? A F G B) Does point D represent a reactant, product, intermediate or transition state? A/ C) Which step (step 1 or step 2) is the rate determining step? Aarrow_forward1. Using radii from Resource section 1 (p.901) and Born-Lande equation, calculate the lattice energy for PbS, which crystallizes in the NaCl structure. Then, use the Born-Haber cycle to obtain the value of lattice energy for PbS. You will need the following data following data: AH Pb(g) = 196 kJ/mol; AHƒ PbS = −98 kJ/mol; electron affinities for S(g)→S¯(g) is -201 kJ/mol; S¯(g) (g) is 640kJ/mol. Ionization energies for Pb are listed in Resource section 2, p.903. Remember that enthalpies of formation are calculated beginning with the elements in their standard states (S8 for sulfur). The formation of S2, AHF: S2 (g) = 535 kJ/mol. Compare the two values, and explain the difference. (8 points)arrow_forwardIn the answer box, type the number of maximum stereoisomers possible for the following compound. A H H COH OH = H C Br H.C OH CHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning