
(a)
Interpretation: The products of the reactions in which diazonium salt is treated with the given reagents have to be drawn.
Concept Introduction:
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
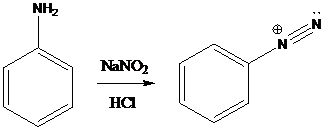
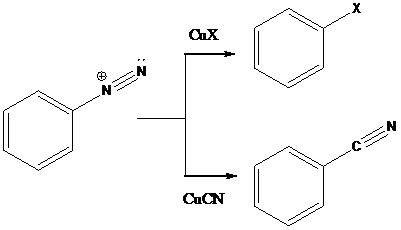
Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.
Schiemann reaction is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
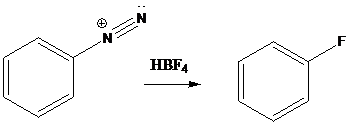
To find: Get the product of the reaction in which diazonium salt is treated with the given reagent (a)
Put the reaction of m-bromoaniline with NaNO2 and HCl
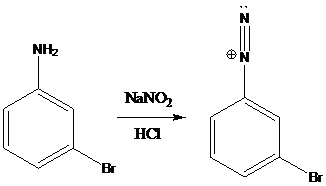
(b)
Interpretation: The products of the reactions in which diazonium salt is treated with the given reagents have to be drawn.
Concept Introduction:
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
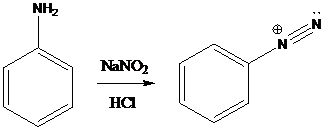
Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

Schiemann reaction is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
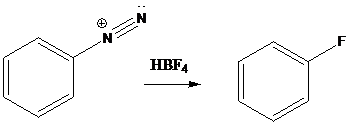
To find: Get the product of the reaction in which diazonium salt is treated with the given reagent (b)
Put the reaction of m-bromoaniline with NaNO2 and HCl
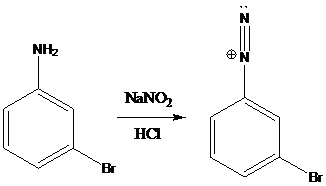
(c)
Interpretation: The products of the reactions in which diazonium salt is treated with the given reagents have to be drawn.
Concept Introduction:
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
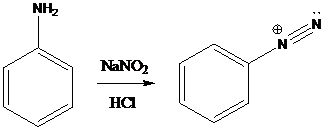
Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

Schiemann reaction is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
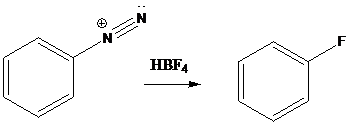
To find: Get the product of the reaction in which diazonium salt is treated with the given reagent
Put the reaction of m-bromoaniline with NaNO2 and HCl
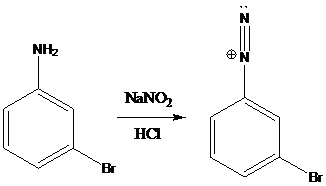
(d)
Interpretation: The products of the reactions in which diazonium salt is treated with the given reagents have to be drawn.
Concept Introduction:
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
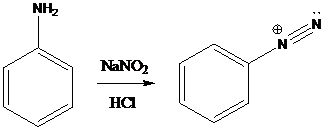
Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

Schiemann reaction is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
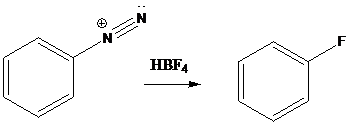
To find: Get the product of the reaction in which diazonium salt is treated with the given reagent
Put the reaction of m-bromoaniline with NaNO2 and HCl
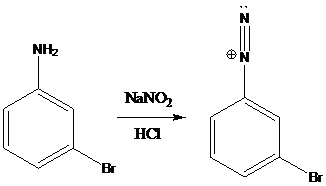
(e)
Interpretation: The products of the reactions in which diazonium salt is treated with the given reagents have to be drawn.
Concept Introduction:
A compound containing an amino group is treated with sodium nitrite and HCl leading to the formation of diazonium salt.
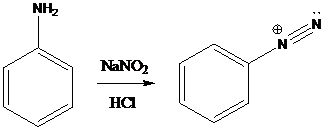
Sandmeyer reactions use copper salts as the reagents. Here, aryldiazonium salt is converted into aryl halides or aryl cyanides by using copper halides or copper cyanides.

Schiemann reaction is the reaction in which diazonium salt is converted into fluoro group using fluoroboric acid (HBF4).
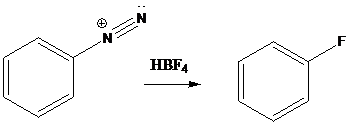
To find: Get the product of the reaction in which diazonium salt is treated with the given reagent (e)
Put the reaction of m-bromoaniline with NaNO2 and HCl
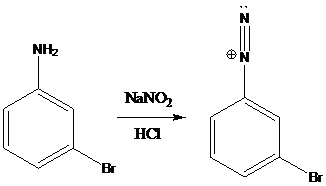
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
EBK ORGANIC CHEMISTRY-STUD.SOLNS.MAN+SG
- If we have the equation 3 ln0 = 2t. What is the value of t?arrow_forward1 N2H4 (l) + 3 O2(g) > 2 NO2 (g) + 2 H2O (g) If 75.0 kg of hydrazine are reacted with 75.0 kg of oxygen, which is the limiting reactant?arrow_forwardPQ-10. What is the major product of this reaction? (A) (C) 930 Me HO O=S=O O-8-CF, C 어 Me H+ OH 270 O 0-5-0 O=S=O O-S-CF CF3 2arrow_forward
- Predict the major organic product(s) of the following reactions. Include stereochemistry when necessary. Write NR if no reaction, try to explain.arrow_forwardQ2: Explain why epoxides that react in an SN1 manner will not show any stereochemical inversion in the product. Q3: Rationalize why Alcohol B will react under the indicated reaction conditions, but Alcohol A will not. A ☑ OH B OH PBr3 R-Brarrow_forwardQ1: Predict the major organic product(s) of the following reactions. Include stereochemistry when necessary. Write NR if no reaction, try to explain. 1.) LDA, THF 2.) СОН CI OH H2SO4, heat OH m...... OH 1.) PCC, CH2Cl2 2.) CH3CH2MgBr, THF 3.) H3O+ 4.) TsCl, pyr 5.) tBuOK, tBuOH 1.) SOCI 2, CHCI 3 2.) CH3CH2ONA, DMF OH 1.) HBr 2.) Mg, THF 3.) H₂CO, THE 4.) H3O+ OH NaH, THFarrow_forward
- Problem 6-29 Identify the functional groups in the following molecules, and show the polarity of each: (a) CH3CH2C=N CH, CH, COCH (c) CH3CCH2COCH3 NH2 (e) OCH3 (b) (d) O Problem 6-30 Identify the following reactions as additions, eliminations, substitutions, or rearrangements: (a) CH3CH2Br + NaCN CH3CH2CN ( + NaBr) Acid -OH (+ H2O) catalyst (b) + (c) Heat NO2 Light + 02N-NO2 (+ HNO2) (d)arrow_forwardPredict the organic product of Y that is formed in the reaction below, and draw the skeletal ("line") structures of the missing organic product. Please include all steps & drawings & explanations.arrow_forwardPlease choose the best reagents to complete the following reactionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





