
Chemistry Principles And Practice
3rd Edition
ISBN: 9781305295803
Author: David Reger; Scott Ball; Daniel Goode
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 22, Problem 65QE
Interpretation Introduction
Interpretation:
The monomer of the given polyacrylonytrile has to be drawn.
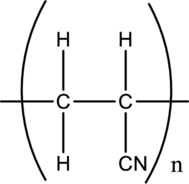
Concept introduction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major organic product(s) of the following reactions. Indicate which of the following mechanisms is in operation: SN1, SN2, E1, or E2.
(c)
(4pts)
Mechanism:
heat
(E1)
CH3OH
+
1.5pts each
_E1 _ (1pt)
Br
CH3OH
(d)
(4pts)
Mechanism:
SN1
(1pt)
(e)
(3pts)
1111 I
H
10
Ill!!
H
LDA
THF (solvent)
Mechanism: E2
(1pt)
NC
(f)
Bri!!!!!
CH3
NaCN
(3pts)
acetone
Mechanism: SN2
(1pt)
(SN1)
-OCH3
OCH3
1.5pts each
2pts for either product
1pt if incorrect
stereochemistry
H
Br
(g)
“,、
(3pts)
H
CH3OH
+21
Mechanism:
SN2
(1pt)
H
CH3
2pts
1pt if incorrect
stereochemistry
H
2pts
1pt if incorrect
stereochemistry
A mixture of butyl acrylate and 4'-chloropropiophenone has been taken for proton NMR analysis. Based on this proton NMR, determine the relative percentage of each compound in the mixture
Chapter 22 Solutions
Chemistry Principles And Practice
Ch. 22 - Prob. 1QECh. 22 -
Explain what is special about the element carbon...Ch. 22 - Prob. 3QECh. 22 - Prob. 4QECh. 22 - Prob. 5QECh. 22 - Prob. 6QECh. 22 - Prob. 7QECh. 22 - Prob. 8QECh. 22 - Prob. 9QECh. 22 - Prob. 10QE
Ch. 22 - Prob. 11QECh. 22 - Prob. 12QECh. 22 - Prob. 13QECh. 22 - Prob. 14QECh. 22 - Prob. 15QECh. 22 - Prob. 16QECh. 22 - Prob. 17QECh. 22 - Prob. 18QECh. 22 - Prob. 19QECh. 22 - Prob. 20QECh. 22 - Prob. 21QECh. 22 - Prob. 22QECh. 22 - Prob. 23QECh. 22 - Prob. 24QECh. 22 - Prob. 25QECh. 22 - Prob. 26QECh. 22 - Prob. 27QECh. 22 - Prob. 28QECh. 22 - Prob. 29QECh. 22 - Prob. 30QECh. 22 - Prob. 31QECh. 22 - Prob. 32QECh. 22 - Prob. 33QECh. 22 - Prob. 34QECh. 22 - Prob. 35QECh. 22 - Prob. 36QECh. 22 - Prob. 37QECh. 22 - Prob. 38QECh. 22 - Prob. 39QECh. 22 - Prob. 40QECh. 22 - Prob. 41QECh. 22 - Prob. 42QECh. 22 - Prob. 43QECh. 22 - Prob. 44QECh. 22 - Prob. 45QECh. 22 - Prob. 46QECh. 22 - Prob. 47QECh. 22 - Prob. 48QECh. 22 - Prob. 49QECh. 22 - Prob. 50QECh. 22 - Prob. 51QECh. 22 - Prob. 52QECh. 22 - Prob. 53QECh. 22 - Prob. 54QECh. 22 - Name the following compounds. (a) FCH2CH2CH2OH (b)...Ch. 22 - Prob. 56QECh. 22 - Prob. 57QECh. 22 - Prob. 58QECh. 22 - Prob. 59QECh. 22 - Prob. 60QECh. 22 - Prob. 61QECh. 22 - Prob. 62QECh. 22 - Prob. 63QECh. 22 - Prob. 64QECh. 22 - Prob. 65QECh. 22 - Prob. 66QECh. 22 - Prob. 67QECh. 22 - Prob. 68QECh. 22 - Prob. 69QECh. 22 - Prob. 70QECh. 22 - Prob. 71QECh. 22 - Prob. 72QECh. 22 - Prob. 73QECh. 22 - Prob. 74QECh. 22 - Prob. 75QECh. 22 - Prob. 76QECh. 22 - Prob. 77QECh. 22 - Prob. 78QECh. 22 - Prob. 79QECh. 22 - Prob. 80QECh. 22 - Prob. 81QECh. 22 - Prob. 82QECh. 22 - Prob. 83QECh. 22 - Prob. 84QECh. 22 - Prob. 85QECh. 22 - Prob. 86QECh. 22 - Prob. 87QECh. 22 - Prob. 88QE
Knowledge Booster
Similar questions
- Q5: Label each chiral carbon in the following molecules as R or S. Make sure the stereocenter to which each of your R/S assignments belong is perfectly clear to the grader. (8pts) R OCH 3 CI H S 2pts for each R/S HO R H !!! I OH CI HN CI R Harrow_forwardCalculate the proton and carbon chemical shifts for this structurearrow_forwardA. B. b. Now consider the two bicyclic molecules A. and B. Note that A. is a dianion and B. is a neutral molecule. One of these molecules is a highly reactive compound first characterized in frozen noble gas matrices, that self-reacts rapidly at temperatures above liquid nitrogen temperature. The other compound was isolated at room temperature in the early 1960s, and is a stable ligand used in organometallic chemistry. Which molecule is the more stable molecule, and why?arrow_forward
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning