
Concept explainers
(a)
Interpretation:
Using crystal field theory, the energy level diagram of d-orbitals of the complex ion
Concept Introduction:
There are five d-orbitals in a metal ion. They have similar energy levels that they are degenerated. Under the influence of ligands during complex formation, the degeneracy in d-orbitals is destroyed that they are split into two sets of orbitals - one set having lower energy and the another set is higher in energy. Crystal field splitting refers to the difference in energy levels between these two sets of d-orbitals.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of octahedral complex, the
(a)
Answer to Problem 22.58QP
The energy level diagram of d-orbitals of the complex ion

Two unpaired electrons are present in this complex ion.
Explanation of Solution
The electronic configuration of

(b)
Interpretation:
Using crystal field theory, the energy level diagram of d-orbitals of the low-spin complex ion
Concept Introduction:
There are five d-orbitals in a metal ion. They have similar energy levels that they are degenerated. Under the influence of ligands during complex formation, the degeneracy in d-orbitals is destroyed that they are split into two sets of orbitals - one set having lower energy and the another set is higher in energy. Crystal field splitting refers to the difference in energy levels between these two sets of d-orbitals.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of octahedral complex, the
(b)
Answer to Problem 22.58QP
The energy level diagram of d-orbitals of the low-spin complex ion
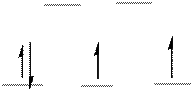
The low-spin complex ion
Explanation of Solution
Atomic number of Osmium is
The electronic configuration of

From the above arrangement we could say there are two unpaired electrons in the complex ion
(c)
Interpretation:
Using crystal field theory, the energy level diagram of d-orbitals of the high-spin complex ion
Concept Introduction:
There are five d-orbitals in a metal ion. They have similar energy levels that they are degenerated. Under the influence of ligands during complex formation, the degeneracy in d-orbitals is destroyed that they are split into two sets of orbitals - one set having lower energy and the another set is higher in energy. Crystal field splitting refers to the difference in energy levels between these two sets of d-orbitals.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of octahedral complex, the
(c)
Answer to Problem 22.58QP
The energy level diagram of d-orbitals of the high-spin complex ion
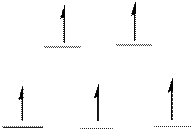
There are five unpaired electrons in the complex ion
Explanation of Solution
Atomic number of Manganese is
The electronic configuration of

From the above arrangement we could say there are five unpaired electrons in the complex ion
Want to see more full solutions like this?
Chapter 22 Solutions
General Chemistry
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forwardIf the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forward
- Given the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forwardIn the battery:Pt | H2 (g) | H+ (aq) | Fe2+ (aq) | FeIndicate the cathode and anode.arrow_forward
- Write the equations that occur when the electrode Pb (s) | PbI2 (s) | KI (ac) in a galvanic cell. a) It functions as a positive electrode b) It functions as a negative electrode c) What is the ion with respect to which this electrode is reversible?arrow_forwardState the formula to find the electromotive force of a battery as a function of the potential of the anode and the cathode.arrow_forwardWhy are normal electrode potentials also called relative electrode potentials?arrow_forward
- Easily differentiate between electrochemical potential and Galvani potential.arrow_forwardConstruct a molecular orbital diagram for carbon monoxide. Identify the relevant point group,include all of the appropriate symmetry labels and pictures, and fill in the electrons. Make sure toaccount for the difference in electronegativity between C and O. Hint: CO is substantiallyisoelectronic to N2. (PLEASE DRAW THE ENTIRE MO DIAGRAM!!!)arrow_forwardplease help with hwarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





