![WebAssign for Zumdahl/Zumdahl/DeCoste's Chemistry, 10th Edition [Instant Access], Single-Term](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
WebAssign for Zumdahl/Zumdahl/DeCoste's Chemistry, 10th Edition [Instant Access], Single-Term
10th Edition
ISBN: 9781337909969
Author: Zumdahl; Steven S.
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 95CP
Consider the following complex ion, where A and B represent ligands.
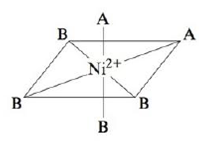
The complex is known to be diamagnetic. Do A and B produce very similar or very different crystal fields? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major organic product(s) of the following reactions. Indicate which of the following mechanisms is in operation: SN1, SN2, E1, or E2.
(c)
(4pts)
Mechanism:
heat
(E1)
CH3OH
+
1.5pts each
_E1 _ (1pt)
Br
CH3OH
(d)
(4pts)
Mechanism:
SN1
(1pt)
(e)
(3pts)
1111 I
H
10
Ill!!
H
LDA
THF (solvent)
Mechanism: E2
(1pt)
NC
(f)
Bri!!!!!
CH3
NaCN
(3pts)
acetone
Mechanism: SN2
(1pt)
(SN1)
-OCH3
OCH3
1.5pts each
2pts for either product
1pt if incorrect
stereochemistry
H
Br
(g)
“,、
(3pts)
H
CH3OH
+21
Mechanism:
SN2
(1pt)
H
CH3
2pts
1pt if incorrect
stereochemistry
H
2pts
1pt if incorrect
stereochemistry
A mixture of butyl acrylate and 4'-chloropropiophenone has been taken for proton NMR analysis. Based on this proton NMR, determine the relative percentage of each compound in the mixture
Chapter 21 Solutions
WebAssign for Zumdahl/Zumdahl/DeCoste's Chemistry, 10th Edition [Instant Access], Single-Term
Ch. 21 - What two first-row transition metals have...Ch. 21 - Prob. 2RQCh. 21 - Prob. 3RQCh. 21 - Prob. 4RQCh. 21 - Prob. 5RQCh. 21 - Prob. 6RQCh. 21 - Prob. 7RQCh. 21 - Prob. 8RQCh. 21 - Prob. 9RQCh. 21 - Prob. 10RQ
Ch. 21 - Prob. 1ALQCh. 21 - Prob. 2ALQCh. 21 - Prob. 3ALQCh. 21 - Prob. 4ALQCh. 21 - Prob. 5QCh. 21 - Four different octahedral chromium coordination...Ch. 21 - Prob. 7QCh. 21 - Prob. 8QCh. 21 - Prob. 9QCh. 21 - Prob. 10QCh. 21 - Prob. 11QCh. 21 - Prob. 13QCh. 21 - Prob. 14QCh. 21 - Which of the following ligands are capable of...Ch. 21 - Prob. 16QCh. 21 - Prob. 17QCh. 21 - What is the maximum number of unpaired d electrons...Ch. 21 - Prob. 19QCh. 21 - Prob. 20QCh. 21 - Prob. 21QCh. 21 - Prob. 22QCh. 21 - Prob. 23ECh. 21 - Prob. 24ECh. 21 - Prob. 25ECh. 21 - Prob. 26ECh. 21 - Prob. 27ECh. 21 - Prob. 28ECh. 21 - Prob. 29ECh. 21 - Prob. 30ECh. 21 - Prob. 31ECh. 21 - When an aqueous solution of KCN is added to a...Ch. 21 - Consider aqueous solutions of the following...Ch. 21 - A coordination compound of cobalt(III) contains...Ch. 21 - Prob. 35ECh. 21 - Prob. 36ECh. 21 - Prob. 37ECh. 21 - Prob. 38ECh. 21 - Prob. 39ECh. 21 - Give formulas for the following complex ions. a....Ch. 21 - Prob. 41ECh. 21 - Prob. 42ECh. 21 - Prob. 43ECh. 21 - Amino acids can act as ligands toward transition...Ch. 21 - Prob. 45ECh. 21 - Prob. 46ECh. 21 - Prob. 47ECh. 21 - Prob. 48ECh. 21 - Prob. 49ECh. 21 - Prob. 50ECh. 21 - Prob. 51ECh. 21 - Prob. 52ECh. 21 - Prob. 53ECh. 21 - Prob. 54ECh. 21 - The CrF64 ion is known to have four unpaired...Ch. 21 - Prob. 56ECh. 21 - Prob. 57ECh. 21 - The complex ion Fe(CN)63 is paramagnetic with one...Ch. 21 - Prob. 59ECh. 21 - Prob. 60ECh. 21 - Prob. 61ECh. 21 - Prob. 62ECh. 21 - The wavelength of absorbed electromagnetic...Ch. 21 - The complex ion NiCL42 has two unpaired electrons,...Ch. 21 - How many unpaired electrons are present in the...Ch. 21 - The complex ion PdCl42is diamagnetic. Propose a...Ch. 21 - Prob. 67ECh. 21 - Prob. 68ECh. 21 - Prob. 69ECh. 21 - Prob. 70ECh. 21 - Silver is sometimes found in nature as large...Ch. 21 - Prob. 72ECh. 21 - Prob. 73AECh. 21 - Prob. 74AECh. 21 - Prob. 75AECh. 21 - The compound cisplatin, Pt(NH3)2Cl2, has been...Ch. 21 - Prob. 77AECh. 21 - Prob. 78AECh. 21 - Prob. 79AECh. 21 - Prob. 80AECh. 21 - Prob. 81AECh. 21 - Name the following coordination compounds. a....Ch. 21 - Prob. 83AECh. 21 - Prob. 84AECh. 21 - Prob. 85AECh. 21 - Prob. 86AECh. 21 - Carbon monoxide is toxic because it binds more...Ch. 21 - Prob. 88AECh. 21 - Prob. 89CWPCh. 21 - Which of the following molecules exhibit(s)...Ch. 21 - Prob. 91CWPCh. 21 - The following table indicates the number of...Ch. 21 - Prob. 93CWPCh. 21 - Which of the following statement(s) is( are) true?...Ch. 21 - Consider the following complex ion, where A and B...Ch. 21 - Consider the pseudo-octahedral complex ion of...Ch. 21 - Prob. 97CPCh. 21 - Prob. 98CPCh. 21 - Chelating ligands often form more stable complex...Ch. 21 - Prob. 100CPCh. 21 - Prob. 101CPCh. 21 - Prob. 102CPCh. 21 - Sketch and explain the most likely crystal field...Ch. 21 - Ethylenediaminetetraacetate (EDTA4 ) is used as a...Ch. 21 - Prob. 105CPCh. 21 - The ferrate ion, FeO42, is such a powerful...Ch. 21 - Ammonia and potassium iodide solutions are added...Ch. 21 - a. In the absorption spectrum of the complex ion...Ch. 21 - There are three salts that contain complex ions of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Q5: Label each chiral carbon in the following molecules as R or S. Make sure the stereocenter to which each of your R/S assignments belong is perfectly clear to the grader. (8pts) R OCH 3 CI H S 2pts for each R/S HO R H !!! I OH CI HN CI R Harrow_forwardCalculate the proton and carbon chemical shifts for this structurearrow_forwardA. B. b. Now consider the two bicyclic molecules A. and B. Note that A. is a dianion and B. is a neutral molecule. One of these molecules is a highly reactive compound first characterized in frozen noble gas matrices, that self-reacts rapidly at temperatures above liquid nitrogen temperature. The other compound was isolated at room temperature in the early 1960s, and is a stable ligand used in organometallic chemistry. Which molecule is the more stable molecule, and why?arrow_forward
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
The Bohr Model of the atom and Atomic Emission Spectra: Atomic Structure tutorial | Crash Chemistry; Author: Crash Chemistry Academy;https://www.youtube.com/watch?v=apuWi_Fbtys;License: Standard YouTube License, CC-BY