
ORGANIC CHEMISTRYPKGDRL+MLCRL MDL
3rd Edition
ISBN: 9781119416746
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 21, Problem 86PP
Interpretation Introduction
Interpretation:
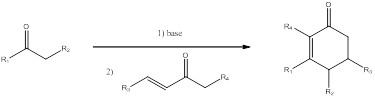
Reagents to be used for synthesis of the given compound using Robinson annulation
Concept introduction:
Robinson annulation is used for synthesizing polycyclic compound, namely six membered ring. This method is used to convert
- Michael addition
- Aldol condensation.
Robinson annulation is comprised of Michael addition which is followed by intamolecular Aldol condensation.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Choose the right answer
8.
What is the major product of the following reaction?
KMnO4
b
a
TOH
OH
OH
C
d
OH
"OH
HO
OH
OH
Choose the right answer
Chapter 21 Solutions
ORGANIC CHEMISTRYPKGDRL+MLCRL MDL
Ch. 21.1 - Prob. 1CCCh. 21.1 - Prob. 2CCCh. 21.1 - Prob. 3CCCh. 21.1 - Prob. 1LTSCh. 21.1 - Prob. 4PTSCh. 21.1 - APPLY the skill
As with aldehydes and ketones, the...Ch. 21.1 - Draw the enolate ion that is formed when each of...Ch. 21.1 - Prob. 7CCCh. 21.2 - Prob. 8CCCh. 21.2 - Prob. 9CC
Ch. 21.2 - Prob. 10CCCh. 21.2 - Prob. 11CCCh. 21.2 - Prob. 12CCCh. 21.2 - Prob. 13CCCh. 21.3 - Prob. 2LTSCh. 21.3 - Prob. 14PTSCh. 21.3 - Prob. 15PTSCh. 21.3 - Prob. 16ATSCh. 21.3 - Prob. 3LTSCh. 21.3 - Prob. 17PTSCh. 21.3 - Prob. 18ATSCh. 21.3 - Prob. 4LTSCh. 21.3 - Prob. 19PTSCh. 21.3 - Prob. 20ATSCh. 21.3 - Prob. 21CCCh. 21.3 - Prob. 22CCCh. 21.3 - Prob. 23CCCh. 21.4 - Prob. 24CCCh. 21.4 - Prob. 25CCCh. 21.4 - Prob. 26CCCh. 21.4 - Prob. 27CCCh. 21.4 - Prob. 28CCCh. 21.5 - Prob. 29CCCh. 21.5 - Prob. 30CCCh. 21.5 - Prob. 5LTSCh. 21.5 - Prob. 31PTSCh. 21.5 - Prob. 32ATSCh. 21.5 - Prob. 6LTSCh. 21.5 - Prob. 33PTSCh. 21.5 - Prob. 34ATSCh. 21.6 - Prob. 35CCCh. 21.6 - Prob. 36CCCh. 21.6 - Prob. 37CCCh. 21.6 - Prob. 7LTSCh. 21.6 - Prob. 38PTSCh. 21.6 - Prob. 39ATSCh. 21.6 - Prob. 40CCCh. 21.6 - Prob. 41CCCh. 21.7 - Prob. 8LTSCh. 21.7 - Prob. 42PTSCh. 21.7 - Prob. 43PTSCh. 21.7 - Prob. 9LTSCh. 21.7 - Prob. 45PTSCh. 21.7 - Prob. 46ATSCh. 21 - Prob. 47PPCh. 21 - Prob. 48PPCh. 21 - Prob. 49PPCh. 21 - Prob. 50PPCh. 21 - Prob. 51PPCh. 21 - Prob. 52PPCh. 21 - Prob. 53PPCh. 21 - Prob. 54PPCh. 21 - Prob. 55PPCh. 21 - Prob. 56PPCh. 21 - Prob. 57PPCh. 21 - Prob. 58PPCh. 21 - Prob. 59PPCh. 21 - Prob. 60PPCh. 21 - Prob. 61PPCh. 21 - Prob. 62PPCh. 21 - Prob. 63PPCh. 21 - Prob. 64PPCh. 21 - Prob. 65PPCh. 21 - Prob. 66PPCh. 21 - Prob. 67PPCh. 21 - Prob. 68PPCh. 21 - Prob. 69PPCh. 21 - Prob. 70PPCh. 21 - Prob. 71PPCh. 21 - Prob. 72PPCh. 21 - Prob. 73PPCh. 21 - Prob. 74PPCh. 21 - Prob. 75PPCh. 21 - Prob. 76PPCh. 21 - Prob. 77PPCh. 21 - Prob. 78PPCh. 21 - Prob. 79PPCh. 21 - Prob. 80PPCh. 21 - Prob. 81PPCh. 21 - Prob. 82PPCh. 21 - Prob. 83PPCh. 21 - Prob. 84PPCh. 21 - Prob. 85PPCh. 21 - Prob. 86PPCh. 21 - Prob. 87PPCh. 21 - Prob. 88PPCh. 21 - Prob. 89IPCh. 21 - Prob. 90IPCh. 21 - Prob. 91IPCh. 21 - Prob. 92IPCh. 21 - Prob. 93IPCh. 21 - Prob. 94IPCh. 21 - Prob. 95IPCh. 21 - Prob. 96IPCh. 21 - Prob. 97IPCh. 21 - Prob. 98IPCh. 21 - Prob. 99IPCh. 21 - Prob. 100IPCh. 21 - Prob. 101IPCh. 21 - Prob. 102IPCh. 21 - Prob. 103IPCh. 21 - Prob. 104IPCh. 21 - Prob. 105IPCh. 21 - Prob. 106IPCh. 21 - Prob. 107IPCh. 21 - Prob. 108IPCh. 21 - Prob. 109IPCh. 21 - Prob. 110IPCh. 21 - Prob. 111IPCh. 21 - Prob. 112IPCh. 21 - Prob. 113IPCh. 21 - Prob. 114IPCh. 21 - Prob. 115IPCh. 21 - Prob. 116CPCh. 21 - Prob. 117CPCh. 21 - Prob. 118CP
Knowledge Booster
Similar questions
- 3. Draw ALL THE POSSBILE PRODUCTS AND THE MECHANISMS WITH ALL RESONANCE STRUCTURES. Explain using the resonance structures why the major product(s) are formed over the minor product(s). H₂SO4, HONO CHarrow_forward7. Provide the product(s), starting material(s) and/or condition(s) required for the No mechanisms required. below reaction HO + H-I CI FO Br2, FeBr3 O I-Oarrow_forward6. Design the most efficient synthesis of the following product starting from phenot Provide the reaction conditions for each step (more than one step is required) and explain the selectivity of each reaction. NO MECHANISMS ARE REQUIRED. OH step(s) CIarrow_forward
- What is the skeletal structure of the product of the following organic reaction?arrow_forwardIf a reaction occurs, what would be the major products? Please include a detailed explanation as well as a drawing showing how the reaction occurs and what the final product is.arrow_forwardWhat is the major organic product of the following nucleophilic acyl substitution reaction of an acid chloride below?arrow_forward
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardIf a reaction occurs, what would be the major products? Please include a detailed explanation as well as a drawing showing how the reaction occurs and what the final product is.arrow_forwardPlease help me answer the following questions using the data I included. 1&2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY