
Concept explainers
a)
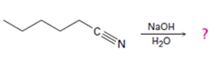
Interpretation:
The products of the reaction along with the mechanism of the reaction are to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and hexanoic acid.
The mechanism of the reaction is given below.
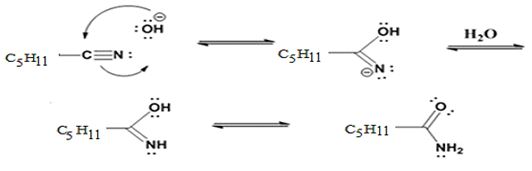
Formation of the amide:
Hydrolysis of the amide:
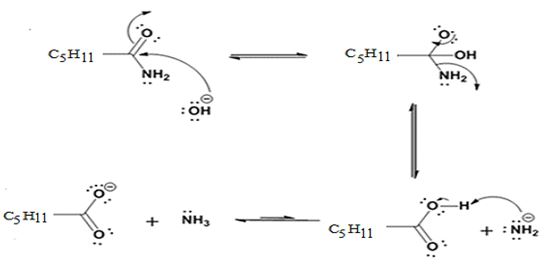
Where C5H11 = -CH2CH2CH2CH2CH3
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in hexanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give hexanamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields hexanoic acid. The deprotonation of the acid by the amide anion leads to the formation of carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and hexanoic acid.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

Where C5H11 = -CH2CH2CH2CH2CH3
b)
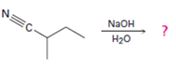
Interpretation:
The products of the reaction along with the mechanism of the reaction are to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and cyclobutanecarboxylate anion.
The mechanism of the reaction is given below.
Formation of the amide:
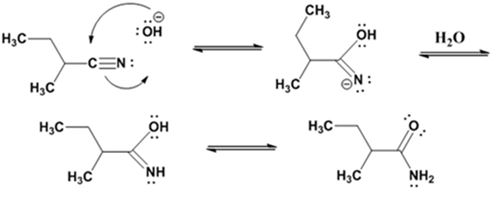
Hydrolysis of the amide:
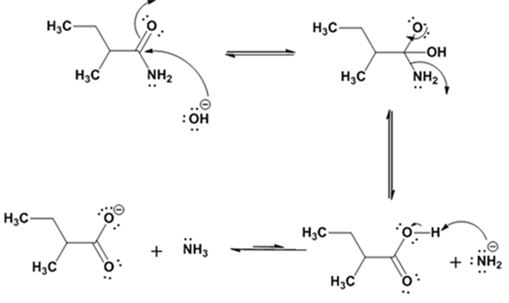
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in 2-methylbutanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give 2-methylbutanamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields butanoic acid. The deprotonation of the acid by the amide anion leads to the formation of carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and cyclobutanecarboxylate anion.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

c)
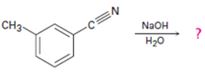
Interpretation:
The product of the reaction is to be given along with the mechanism of the reaction.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine. iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
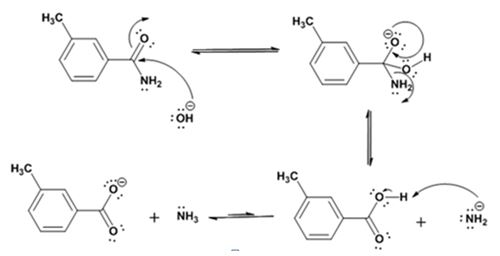
The products of the reaction are ammonia and m-methylbenzamide.
The mechanism of the reaction is given below.
Formation of the amide:
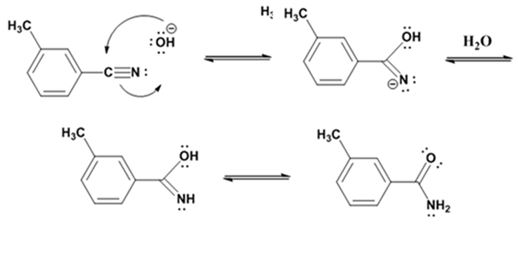
Hydrolysis of the amide:
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in m-methylbenzonitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give 3-methylbenzamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields m-methylbenzoic acid. The deprotonation of the acid by the amide anion leads to the formation of the carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and m-methylbenzamide.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

d)
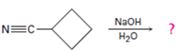
Interpretation:
The products of the reaction along with the mechanism of the reaction is to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and cyclobutanecarboxylate ion.
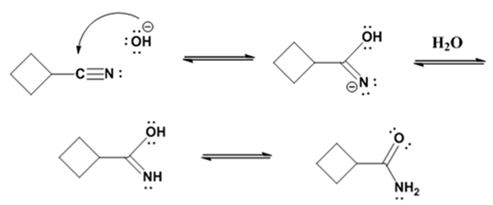
The mechanism of the reaction is given below.
Formation of the amide:
Hydrolysis of the amide:
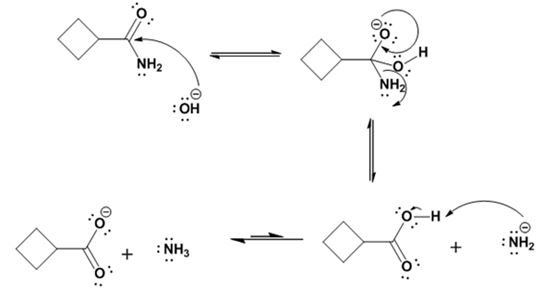
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in cyclobutanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give cyclobutanecarboxylamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields cyclobutane
The products of the reaction are ammonia and cyclobutanecarboxylate ion.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

Want to see more full solutions like this?
Chapter 20 Solutions
Study Guide with Student Solutions Manual for McMurry's Organic Chemistry, 9th
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forward(a) Sketch the 'H NMR of the following chemical including the approximate chemical shifts, the multiplicity (splitting) of all signals and the integration (b) How many signals would you expect in the 13C NMR? CH3arrow_forwardDraw the Show the major and minor product(s) for the following reaction mechanisms for both reactions and show all resonance structures for any Explain why the major product is favoured? intermediates H-Brarrow_forward
- 3. Draw ALL THE POSSBILE PRODUCTS AND THE MECHANISMS WITH ALL RESONANCE STRUCTURES. Explain using the resonance structures why the major product(s) are formed over the minor product(s). H₂SO4, HONO CHarrow_forward7. Provide the product(s), starting material(s) and/or condition(s) required for the No mechanisms required. below reaction HO + H-I CI FO Br2, FeBr3 O I-Oarrow_forward6. Design the most efficient synthesis of the following product starting from phenot Provide the reaction conditions for each step (more than one step is required) and explain the selectivity of each reaction. NO MECHANISMS ARE REQUIRED. OH step(s) CIarrow_forward
- What is the skeletal structure of the product of the following organic reaction?arrow_forwardIf a reaction occurs, what would be the major products? Please include a detailed explanation as well as a drawing showing how the reaction occurs and what the final product is.arrow_forwardWhat is the major organic product of the following nucleophilic acyl substitution reaction of an acid chloride below?arrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


