
Concept explainers
Write the chemical formula and Lewis structure of the following each of which contains five carbon atoms:
(a) an
(b) an
(c) an
a)
Interpretation:
The chemical formula and Lewis structure of an alkane with five carbon atoms are to be written.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
The completely saturated hydrocarbon is known as an alkane.
The general molecular formula of alkane is
Answer to Problem 1E
Chemical formula:
Lewis structure:
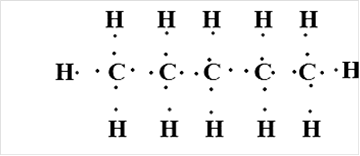
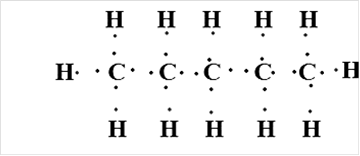
Pentane
Explanation of Solution
The general formula of alkane is
In case of five carbon atoms, the molecular formula of alkane is
- Lewis structures are the diagrams that show the bonding between the atoms of the molecules and existing lone pairs of electrons.
- Bonding electrons are those electrons which are shared between the atoms resulting in the formation of bond.
- Non-bonding electrons are the valence electrons of the atom which are not shared with another atom.
Number of valence electrons in a carbon atom = 4
Number of valence electrons in a hydrogen atom = 1
Total number of valence electrons =
= 32
Thus, Lewis structure of alkane having five carbon atoms is:
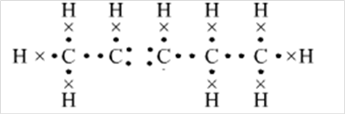
 Or,
Or, 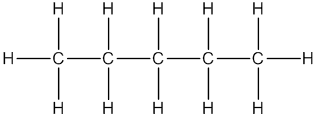
b)
Interpretation:
The chemical formula and Lewis structure of an alkene with five carbon atoms are to be written.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
The unsaturated hydrocarbon with one or more double bond is known as an alkene.
The general molecular formula of alkene is
Answer to Problem 1E
Chemical formula:
Lewis structure:
Explanation of Solution
The general formula of alkene is
In case of five carbon atoms, the molecular formula of alkene is
- Lewis structures are the diagrams that show the bonding between the atoms of the molecules and existing lone pairs of electrons.
- Bonding electrons are those electrons which are shared between the atoms resulting in the formation of bond.
- Non-bonding electrons are the valence electrons of the atom which are not shared with another atom.
Number of valence electrons in a carbon atom = 4
Number of valence electrons in a hydrogen atom = 1
Total number of valence electrons =
= 30
Thus, Lewis structure of alkene having five carbon atoms is:
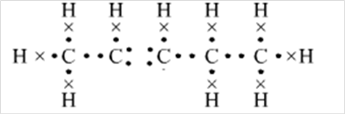 Or,
Or, 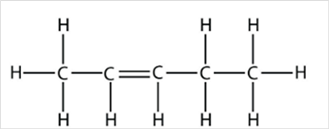
c)
Interpretation:
The chemical formula and Lewis structure of an alkyne with five carbon atoms are to be written.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms.Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
The unsaturated hydrocarbon with one or more triple bond is known as an alkyne.
The general molecular formula of alkyne is
Answer to Problem 1E
Chemical formula:
Lewis structure:
Explanation of Solution
The general formula of alkyne is
In case of five carbon atoms, the molecular formula of alkyne is
- Lewis structures are the diagrams that show the bonding between the atoms of the molecules and existing lone pairs of electrons.
- Bonding electrons are those electrons which are shared between the atoms resulting in the formation of bond.
- Non-bonding electrons are the valence electrons of the atom which are not shared with another atom.
Number of valence electrons in a carbon atom = 4
Number of valence electrons in a hydrogen atom = 1
Total number of valence electrons =
= 28
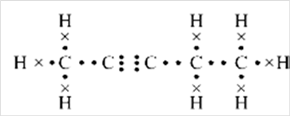
Thus, Lewis structure of alkyne having five carbon atoms is:
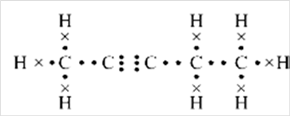 Or,
Or, 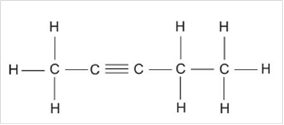
Want to see more full solutions like this?
Chapter 20 Solutions
Chemistry (OER)
Additional Science Textbook Solutions
Human Anatomy & Physiology (2nd Edition)
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
Biology: Life on Earth (11th Edition)
Fundamentals of Anatomy & Physiology (11th Edition)
Campbell Essential Biology (7th Edition)
- For reaction N2(g) + O2(g) --> 2NO(g) Write the rate of the reaction in terms of change of NO.arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardThe reaction of 2-oxacyclopentanone with hydrochloric acid in water (i.e., "excess") produces which of the following carboxylic acids?arrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forwardWhat is the name of the major product formed during the reaction between benzoyl chloride and phenol? benzyl ester O phenyl benzoate ○ cyclopentanoate ○ benzyl phenoate ○ benzenecarboxylic acidarrow_forwardProvide the proper IUPAC or common name for the following compound. Dashes, commas, and spaces must be used correctly.arrow_forward
- Provide the proper IUPAC name (only) for the following compound. Dashes, commas, and spaces must be used correctly. HO. OHarrow_forwardQuestion 2 0/1 pts Provide the proper IUPAC name only for the following compound. Dashes, commas, and spaces must be used correctly. HO CH 3 1-methyl-1-cyclohexanecarboxylic acidarrow_forwardPlease assign all the carbons for C-NMR and hydrogen for H-NMR. Please if I can get that less than hourarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
