
LCPO CHEMISTRY W/MODIFIED MASTERING
8th Edition
ISBN: 9780135214756
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.15P
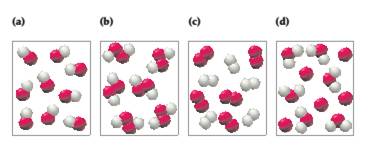
Which of the following drawings represents a pure sample of hydrogen peroxide
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Chemistry Question
Draw the formula of the product obtained by reacting adipic acid 1st with PCl5 and 2nd treatment with NH3.
please help me with my homework
Chapter 2 Solutions
LCPO CHEMISTRY W/MODIFIED MASTERING
Ch. 2 - An element is a shiny, silver-colored solid at...Ch. 2 - Prob. 2.2ACh. 2 - PRACTICE 2.3Compounds A and B are colorless gases...Ch. 2 - APPLY 2.4If the chemical formula of compound A in...Ch. 2 - PRACTICE 2.5The gold foil that Rutherford used in...Ch. 2 - Prob. 2.6ACh. 2 - PRACTICE 2.7The isotope 3475Se is used medically...Ch. 2 - APPLY 2.8Element X is toxic to humans in high...Ch. 2 - PRACTICE 2.9Copper metal has two naturally occur-...Ch. 2 - Prob. 2.10A
Ch. 2 - Prob. 2.11PCh. 2 - APPLY 2.12If 2.26 1022 atoms Of element Y have a...Ch. 2 - Use the data from the mass spectrum of asample of...Ch. 2 - Use the data from the mass spectrum of a sample of...Ch. 2 - Which of the following drawings represents a pure...Ch. 2 - Red and blue spheres representatoms of different...Ch. 2 - Thymine, one of the four basesin deoxyribonucleic...Ch. 2 - Prob. 2.18ACh. 2 - Prob. 2.19PCh. 2 - Which of the following drawings most likely...Ch. 2 - Prob. 2.21PCh. 2 - Prob. 2.22ACh. 2 - Prob. 2.23PCh. 2 - Prob. 2.24ACh. 2 - Prob. 2.25PCh. 2 - Prob. 2.26ACh. 2 - Global climate is affected by variations in (a)...Ch. 2 - Prob. 2.28PCh. 2 - How many protons, neutrons, and electrons arein...Ch. 2 - Which sample of H2O has a higher ratio of 18O/16O...Ch. 2 - The last ice age occurred from 110,000 to11,700...Ch. 2 - For this problem, assume that water consistsonly...Ch. 2 - Prob. 2.33PCh. 2 - Prob. 2.34CPCh. 2 - Prob. 2.35CPCh. 2 - Prob. 2.36CPCh. 2 - Prob. 2.37CPCh. 2 - If yellow spheres represent sulfur atoms and red...Ch. 2 - Prob. 2.39CPCh. 2 - Prob. 2.40CPCh. 2 - Which of the following three drawings represents a...Ch. 2 - In the following drawings, red and blue spheres...Ch. 2 - Prob. 2.43CPCh. 2 - Prob. 2.44CPCh. 2 - Prob. 2.45CPCh. 2 - Prob. 2.46SPCh. 2 - Which element accounts for roughly 75% of the...Ch. 2 - Prob. 2.48SPCh. 2 - Prob. 2.49SPCh. 2 - Prob. 2.50SPCh. 2 - Prob. 2.51SPCh. 2 - Prob. 2.52SPCh. 2 - Prob. 2.53SPCh. 2 - Examine Figure 2.2, A portion of Mendeleev’s...Ch. 2 - Prob. 2.55SPCh. 2 - Prob. 2.56SPCh. 2 - Prob. 2.57SPCh. 2 - Prob. 2.58SPCh. 2 - Prob. 2.59SPCh. 2 - Prob. 2.60SPCh. 2 - Prob. 2.61SPCh. 2 - Prob. 2.62SPCh. 2 - Prob. 2.63SPCh. 2 - 2.60 List several general properties of the...Ch. 2 - Prob. 2.65SPCh. 2 - Prob. 2.66SPCh. 2 - Prob. 2.67SPCh. 2 - At room temperature, a certain element is found to...Ch. 2 - Prob. 2.69SPCh. 2 - At room temperature, a certain element is yellow...Ch. 2 - Prob. 2.71SPCh. 2 - Prob. 2.72SPCh. 2 - Prob. 2.73SPCh. 2 - Prob. 2.74SPCh. 2 - Prob. 2.75SPCh. 2 - How does Dalton’s atomic theory account for the...Ch. 2 - Prob. 2.77SPCh. 2 - A sample of mercury with a mass of 114.0 g was...Ch. 2 - Prob. 2.79SPCh. 2 - In methane, one part hydrogen combine with three...Ch. 2 - In borane, one part hydrogen combine with 3.6...Ch. 2 - Benzene, ethane, and ethylene are just three of a...Ch. 2 - Prob. 2.83SPCh. 2 - Prob. 2.84SPCh. 2 - 2.79 In addition to carbon monoxide (CO) and...Ch. 2 - Prob. 2.86SPCh. 2 - What affects the magnitude of the deflection of...Ch. 2 - Prob. 2.88SPCh. 2 - Prob. 2.89SPCh. 2 - Which of the following charges is NOT possible for...Ch. 2 - What discovery about atomic structure was made...Ch. 2 - Prior to Rutherford’s gold foil experiment, the...Ch. 2 - A period at the end of sentence written with a...Ch. 2 - A 1/4 inch thick lead sheet is used for protection...Ch. 2 - Prob. 2.95SPCh. 2 - What is the difference between an atom’s atomic...Ch. 2 - Prob. 2.97SPCh. 2 - Prob. 2.98SPCh. 2 - Prob. 2.99SPCh. 2 - Prob. 2.100SPCh. 2 - The radioactive isotope cesium-137 was produced in...Ch. 2 - Prob. 2.102SPCh. 2 - Prob. 2.103SPCh. 2 - How many protons, neutrons, and electrons are in...Ch. 2 - Prob. 2.105SPCh. 2 - Prob. 2.106SPCh. 2 - Prob. 2.107SPCh. 2 - Prob. 2.108SPCh. 2 - Prob. 2.109SPCh. 2 - Fluorine occurs naturally as a single isotope. How...Ch. 2 - Hydrogen has three isotopes (1H,2H,and3H) , and...Ch. 2 - The unified atomic mass unit (u) is defined as...Ch. 2 - Prob. 2.113SPCh. 2 - Prob. 2.114SPCh. 2 - Label the following statements as true or false....Ch. 2 - Copper has two naturally occurring isotopes,...Ch. 2 - Sulfur has four naturally occurring isotopes,...Ch. 2 - aturally occurring boron consists of two isotopes:...Ch. 2 - Prob. 2.119SPCh. 2 - Magnesium has three naturally occurring isotopes:...Ch. 2 - A sample of naturally occurring silicon consists...Ch. 2 - Copper metal has two naturally occurring isotopes:...Ch. 2 - Germanium has five naturally occurring...Ch. 2 - What is the mass in grams of each of the following...Ch. 2 - How many moles are in each of the following...Ch. 2 - If the atomic weight of an element is x, what is...Ch. 2 - If the atomic weight of an element is x, what is...Ch. 2 - Prob. 2.128SPCh. 2 - If 4.611021 atoms of element Z have a mass of 0.8...Ch. 2 - Refer to Figure 2.10 showing a schematic...Ch. 2 - Copper has two naturally occurring isotopes,...Ch. 2 - Use the data from the mass spectrum of a sample of...Ch. 2 - Use the data from the mass spectrum of a sample of...Ch. 2 - Prob. 2.134SPCh. 2 - Which of the following bonds are likely to be...Ch. 2 - The symbol CO stands for carbon monoxide, but the...Ch. 2 - Prob. 2.137SPCh. 2 - Prob. 2.138SPCh. 2 - Prob. 2.139SPCh. 2 - Prob. 2.140SPCh. 2 - Prob. 2.141SPCh. 2 - Prob. 2.142SPCh. 2 - Prob. 2.143SPCh. 2 - Prob. 2.144SPCh. 2 - Prob. 2.145SPCh. 2 - Give systematic names for the following binary...Ch. 2 - Give systematic names for the following binary...Ch. 2 - Prob. 2.148SPCh. 2 - Prob. 2.149SPCh. 2 - Prob. 2.150SPCh. 2 - Prob. 2.151SPCh. 2 - Give systematic names for the following compounds:...Ch. 2 - Name the following ions: (a) Ba2+ (b) Cs+ (c) V3+...Ch. 2 - Prob. 2.154SPCh. 2 - Prob. 2.155SPCh. 2 - Prob. 2.156SPCh. 2 - Prob. 2.157SPCh. 2 - Prob. 2.158SPCh. 2 - Prob. 2.159SPCh. 2 - Prob. 2.160SPCh. 2 - Prob. 2.161SPCh. 2 - Prob. 2.162SPCh. 2 - Prob. 2.163SPCh. 2 - Prob. 2.164SPCh. 2 - Prob. 2.165SPCh. 2 - Ammonia (NH3) and hydrazine (N2H4) are both...Ch. 2 - If 3.670 g of nitrogen combines with 0.5275 g of...Ch. 2 - Prior to 1961, the atomic mass unit (amu) was...Ch. 2 - What was the mass ¡n atomic mass units of a 40Ca...Ch. 2 - Prob. 2.170MPCh. 2 - The mass percent of an element in a compound is...Ch. 2 - In an alternate universe, the smallest negatively...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- helparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forward
- pressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward5.arrow_forward
- 6.arrow_forward0/5 alekscgi/x/sl.exe/1o_u-IgNglkr7j8P3jH-IQs_pBaHhvlTCeeBZbufuBYTi0Hz7m7D3ZcSLEFovsXaorzoFtUs | AbtAURtkqzol 1HRAS286, O States of Matter Sketching a described thermodynamic change on a phase diagram The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 3 pressure (atm) + 0- 0 5+ 200 temperature (K) 400 Explanation Check X 0+ F3 F4 F5 F6 F7 S 2025 McGraw Hill LLC All Rights Reserved. Terms of Use Privacy Center Accessibility Q Search LUCR + F8 F9 F10 F11 F12 * % & ( 5 6 7 8 9 Y'S Dele Insert PrtSc + Backsarrow_forward5.arrow_forward
- 9arrow_forwardalekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZS18w-nDB10538ZsAtmorZoFusYj2Xu9b78gZo- O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 3- 200 temperature (K) Explanation Chick Q Sowncharrow_forward0+ aleksog/x/lsl.exe/1ou-lgNgkr7j8P3H-IQs pBaHhviTCeeBZbufuBYTOHz7m7D3ZStEPTBSB3u9bsp3Da pl19qomOXLhvWbH9wmXW5zm O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 Gab The temperature on a sample of pure X held at 0.75 atm and -229. °C is increased until the sample sublimes. The temperature is then held constant and the pressure is decreased by 0.50 atm. On the phase diagram below draw a path that shows this set of changes. F3 pressure (atm) 0- 0 200 Explanation temperature (K) Check F4 F5 ☀+ Q Search Chill Will an 9 ENG F6 F7 F8 F9 8 Delete F10 F11 F12 Insert PrtSc 114 d Ararrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY