
Interpretation:
Partial columns has to be filled in the table given in problem statement.
Concept Introduction:
Each and every element present in the Periodic table has a unique name. Some of the elements are named considering their
Chemical names are represented as atomic symbols. In the symbols, the mass number and atomic number are shown. The complete
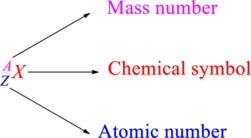
Atomic number is the total number of protons present in the atom of an element. Mass number is the total number of protons and neutrons present in nucleus of an atom.
Explanation of Solution
For entry 1:
Number of protons is given as 31. This means that the atomic number of the species is 31. The element with atomic number 31 is found to be gallium with the symbol of
Mass number is the sum of number of protons and number of neutrons. From the mass number given as 70, the number of neutrons can be calculated as shown below.
Number of electrons is given as 28. The charge on the species can be calculated as shown below.
The symbol for the species is given as
For entry 2:
Number of electrons is given as 42. Charge on the species is given as
Number of protons is found as 45. This means that the atomic number of the species is 45. The element with atomic number 45 is found to be rhodium with the symbol of
Mass number is the sum of number of protons and number of neutrons. From the mass number given as 103, the number of neutrons can be calculated as shown below.
The symbol for the species is given as
For entry 3:
Atomic number is given as 49. This means that the number of protons in the species is 49. The element with atomic number 49 is found to be indium with the symbol of
The charge on the species is given as
Mass number is the sum of number of protons and number of neutrons. Number of neutrons is given as 65. From this mass number can be calculated as shown below.
The symbol for the species is given as
For entry 4:
Symbol is given as
Mass number is the sum of number of protons and number of neutrons. From the mass number given as 40 in the symbol, the number of neutrons can be calculated as shown below.
Charge is given as
Complete table can be given as shown below.
| Symbol | ||||
| Atomic number | ||||
| Mass number | ||||
| Charge | ||||
| Number of protons | ||||
| Number of electrons | ||||
| Number of neutrons |
Want to see more full solutions like this?
Chapter 2 Solutions
Chemistry Principles And Practice
- Can I get helpp drawing my arrowsarrow_forwardWhich of the m/z values corresponds to the base peak in the mass spectrum shown? 100 80 A. 45 B. 44 C. 29 D. 15 Intensity 20 0 10 20 30 40 B- m/z -8 50 E. 30 Which of the m/z values correspond to the molecular ion for the compound shown? A. 18 B. 82 OH C. 100 D. 102 E. 103arrow_forwardCan someone help me with drawing my arrows.arrow_forward
- I'm having trouble with converting lewis diagrams into VSEPR diagrams. I currently have this example of C2BrCl3 which I want to turn into a lewis structure, but I'm not sure what steps I need to do in order to do so. I have the table written down, however, there's two central atoms so what would I do? There seems to be 4 electron domains on the carbon atom and no lone pairs so it would seem like this shape would be tetrahedral. Here's what I have now. Thanks!arrow_forwardWe discussed the solid phase resin using in peptide synthesis. Provide a mechanism, for its formation. DRAW THE MECHANISM.arrow_forwardPlease help. Every time I've asked an expert in the past, it's been wrong :(arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





