
For four situations for an ideal gas, the table gives the energy transferred to or front the gas as heat Q and either the work W done by the gas or the work Won done on the gas, all in joules. Rank the four situations in terms of the temperature change of the gas, most positive first.
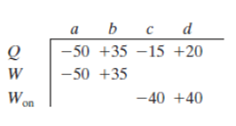
To rank:
The four situations in terms of temperature change of the gas, most positive first.
Answer to Problem 1Q
Solution:
The ranking of four situations in terms of temperature change of the gas is
Explanation of Solution
1) Concept:
We can find the change in the internal energy using the first law of thermodynamics. Then using the relation between change in internal energy and change in temperature, we can rank the four situations in terms of temperature change of the gas.
2) Formulae:
3) Given:
The table containing four situations for an ideal gas
4) Calculations:
The change in the internal energy is given by
So, change in the internal energy of an ideal gas depends solely on the change in temperature. According to the first law of thermodynamics,
where, heat energy Q is taken positive if it is added to the system and work done W is taken as positive if it is done by the gas.
a) In situation a,
Hence,
b) In situation b,
Hence,
c) In situation c,
Hence,
d) In situation d,
Hence,
Therefore, the ranking of four situations in terms of temperature change of the gas is,
Conclusion:
The change in the internal energy of an ideal gas solely depends on the change in temperature.
Want to see more full solutions like this?
Chapter 19 Solutions
Fundamentals Of Physics
Additional Science Textbook Solutions
Human Anatomy & Physiology (Marieb, Human Anatomy & Physiology) Standalone Book
Introductory Chemistry (6th Edition)
College Physics: A Strategic Approach (3rd Edition)
Microbiology: An Introduction
Human Anatomy & Physiology (2nd Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
- pls help on all asked questions kindlyarrow_forward19. Mount Everest, Earth's highest mountain above sea level, has a peak of 8849 m above sea level. Assume that sea level defines the height of Earth's surface. (re = 6.38 × 106 m, ME = 5.98 × 1024 kg, G = 6.67 × 10 -11 Nm²/kg²) a. Calculate the strength of Earth's gravitational field at a point at the peak of Mount Everest. b. What is the ratio of the strength of Earth's gravitational field at a point 644416m below the surface of the Earth to a point at the top of Mount Everest? C. A tourist watching the sunrise on top of Mount Everest observes a satellite orbiting Earth at an altitude 3580 km above his position. Determine the speed of the satellite.arrow_forwardpls help on allarrow_forward
- pls help on allarrow_forward6. As the distance between two charges decreases, the magnitude of the electric potential energy of the two-charge system: a) Always increases b) Always decreases c) Increases if the charges have the same sign, decreases if they have the opposite signs d) Increases if the charges have the opposite sign, decreases if they have the same sign 7. To analyze the motion of an elastic collision between two charged particles we use conservation of & a) Energy, Velocity b) Momentum, Force c) Mass, Momentum d) Energy, Momentum e) Kinetic Energy, Potential Energyarrow_forwardpls help on all asked questions kindlyarrow_forward
- pls help on all asked questions kindlyarrow_forward17. Two charges, one of charge +2.5 × 10-5 C and the other of charge +3.7 × 10-6 C, are 25.0 cm apart. The +2.5 × 10−5 C charge is to the left of the +3.7 × 10−6 C charge. a. Draw a diagram showing the point charges and label a point Y that is 20.0 cm to the left of the +3.7 × 10-6 C charge, on the line connecting the charges. (Field lines do not need to be drawn.) b. Calculate the net electric field at point Y.arrow_forward3arrow_forward

 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





