
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
13th Edition
ISBN: 9780137444298
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 18.8, Problem 18.66PP
Interpretation Introduction
To determine:Draw the condensed formula for the a-keto acid produced from the following transamination.
a.
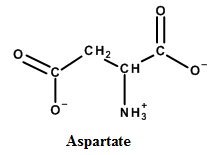 |
b.
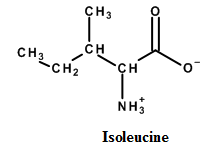 |
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part I.
Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff:
Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone
and
(3,3-dimethyl-2-butanone) 2,3-dimethyl-1,3-butadiene. Give reasonable mechanism
the formation of the products
For
Show the mechanism for these reactions
Draw the stepwise mechanism
Chapter 18 Solutions
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
Ch. 18.1 - Prob. 18.1PPCh. 18.1 - Prob. 18.2PPCh. 18.1 - Prob. 18.3PPCh. 18.1 - Prob. 18.4PPCh. 18.1 - Prob. 18.5PPCh. 18.1 - Prob. 18.6PPCh. 18.2 - Prob. 18.7PPCh. 18.2 - Prob. 18.8PPCh. 18.2 - Prob. 18.9PPCh. 18.2 - Prob. 18.10PP
Ch. 18.2 - Prob. 18.11PPCh. 18.2 - Prob. 18.12PPCh. 18.3 - Prob. 18.13PPCh. 18.3 - Prob. 18.14PPCh. 18.3 - Prob. 18.15PPCh. 18.3 - Prob. 18.16PPCh. 18.3 - Prob. 18.17PPCh. 18.3 - Prob. 18.18PPCh. 18.4 - Prob. 18.19PPCh. 18.4 - Prob. 18.20PPCh. 18.4 - Prob. 18.21PPCh. 18.4 - Prob. 18.22PPCh. 18.4 - Prob. 18.23PPCh. 18.4 - Prob. 18.24PPCh. 18.4 - Prob. 18.25PPCh. 18.4 - Prob. 18.26PPCh. 18.4 - Prob. 18.27PPCh. 18.4 - Prob. 18.28PPCh. 18.4 - Prob. 18.29PPCh. 18.4 - Prob. 18.30PPCh. 18.4 - Prob. 18.31PPCh. 18.4 - Prob. 18.32PPCh. 18.4 - Prob. 18.33PPCh. 18.4 - Prob. 18.34PPCh. 18.5 - Prob. 18.35PPCh. 18.5 - Prob. 18.36PPCh. 18.5 - Prob. 18.37PPCh. 18.5 - Prob. 18.38PPCh. 18.5 - Prob. 18.39PPCh. 18.5 - Prob. 18.40PPCh. 18.5 - Prob. 18.41PPCh. 18.5 - Prob. 18.42PPCh. 18.6 - Prob. 18.43PPCh. 18.6 - Prob. 18.44PPCh. 18.6 - Prob. 18.45PPCh. 18.6 - Prob. 18.46PPCh. 18.6 - Prob. 18.47PPCh. 18.6 - Prob. 18.48PPCh. 18.6 - Prob. 18.49PPCh. 18.6 - Prob. 18.50PPCh. 18.6 - Prob. 18.51PPCh. 18.6 - Prob. 18.52PPCh. 18.6 - Prob. 18.53PPCh. 18.6 - Prob. 18.54PPCh. 18.6 - Prob. 18.55PPCh. 18.6 - Prob. 18.56PPCh. 18.7 - Prob. 18.57PPCh. 18.7 - Prob. 18.58PPCh. 18.7 - Prob. 18.59PPCh. 18.7 - Prob. 18.60PPCh. 18.7 - Prob. 18.61PPCh. 18.7 - Prob. 18.62PPCh. 18.7 - Prob. 18.63PPCh. 18.7 - Prob. 18.64PPCh. 18.8 - 18.65 Draw the condensed structural formula for...Ch. 18.8 - 18.66 Draw the condensed structural formula for...Ch. 18.8 - 18.67 Why does the body convert NH4+ to urea?
Ch. 18.8 - Prob. 18.68PPCh. 18.8 - Prob. 18.69PPCh. 18.8 - Prob. 18.70PPCh. 18.8 - Draw the condensed structural formulas for the...Ch. 18.8 - Prob. 18.72PPCh. 18 - Prob. 18.73UTCCh. 18 - Prob. 18.74UTCCh. 18 - Prob. 18.75UTCCh. 18 - Prob. 18.76UTCCh. 18 - Prob. 18.77APPCh. 18 - Prob. 18.78APPCh. 18 - Prob. 18.79APPCh. 18 - Prob. 18.80APPCh. 18 - Prob. 18.81APPCh. 18 - Prob. 18.82APPCh. 18 - Prob. 18.83APPCh. 18 - Prob. 18.84APPCh. 18 - Prob. 18.85APPCh. 18 - Prob. 18.86APPCh. 18 - Prob. 18.87APPCh. 18 - Prob. 18.88APPCh. 18 - Prob. 18.89APPCh. 18 - Prob. 18.90APPCh. 18 - Prob. 18.91APPCh. 18 - Prob. 18.92APPCh. 18 - Prob. 18.93CPCh. 18 - Prob. 18.94CPCh. 18 - Prob. 18.95CPCh. 18 - Prob. 18.96CPCh. 18 - Prob. 18.97CPCh. 18 - Prob. 18.98CPCh. 18 - Prob. 33CICh. 18 - CI.34 In response to signals from the nervous...Ch. 18 - Prob. 35CICh. 18 - Prob. 36CICh. 18 - Prob. 37CICh. 18 - Prob. 38CICh. 18 - Prob. 39CICh. 18 - Thalassemia is an inherited genetic mutation that...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a structural formula of the principal product formed when benzonitrile is treated with each reagent. (a) H₂O (one equivalent), H₂SO₄, heat (b) H₂O (excess), H₂SO₄, heat (c) NaOH, H₂O, heat (d) LiAlH4, then H₂Oarrow_forwardDraw the stepwise mechanism for the reactionsarrow_forwardDraw stepwise mechanismarrow_forward
- Part I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: a) Give the major reason for the exposure of benzophenone al isopropyl alcohol (w/acid) to direct sunlight of pina colone Mechanism For b) Pinacol (2,3-dimethy 1, 1-3-butanediol) on treatment w/ acid gives a mixture (3,3-dimethyl-2-butanone) and 2, 3-dimethyl-1,3-butadiene. Give reasonable the formation of the productsarrow_forwardwhat are the Iupac names for each structurearrow_forwardWhat are the IUPAC Names of all the compounds in the picture?arrow_forward
- 1) a) Give the dominant Intermolecular Force (IMF) in a sample of each of the following compounds. Please show your work. (8) SF2, CH,OH, C₂H₂ b) Based on your answers given above, list the compounds in order of their Boiling Point from low to high. (8)arrow_forward19.78 Write the products of the following sequences of reactions. Refer to your reaction road- maps to see how the combined reactions allow you to "navigate" between the different functional groups. Note that you will need your old Chapters 6-11 and Chapters 15-18 roadmaps along with your new Chapter 19 roadmap for these. (a) 1. BHS 2. H₂O₂ 3. H₂CrO4 4. SOCI₂ (b) 1. Cl₂/hv 2. KOLBU 3. H₂O, catalytic H₂SO4 4. H₂CrO4 Reaction Roadmap An alkene 5. EtOH 6.0.5 Equiv. NaOEt/EtOH 7. Mild H₂O An alkane 1.0 2. (CH3)₂S 3. H₂CrO (d) (c) 4. Excess EtOH, catalytic H₂SO OH 4. Mild H₂O* 5.0.5 Equiv. NaOEt/EtOH An alkene 6. Mild H₂O* A carboxylic acid 7. Mild H₂O* 1. SOC₁₂ 2. EtOH 3.0.5 Equiv. NaOEt/E:OH 5.1.0 Equiv. NaOEt 6. NH₂ (e) 1. 0.5 Equiv. NaOEt/EtOH 2. Mild H₂O* Br (f) i H An aldehyde 1. Catalytic NaOE/EtOH 2. H₂O*, heat 3. (CH,CH₂)₂Culi 4. Mild H₂O* 5.1.0 Equiv. LDA Br An ester 4. NaOH, H₂O 5. Mild H₂O* 6. Heat 7. MgBr 8. Mild H₂O* 7. Mild H₂O+arrow_forwardLi+ is a hard acid. With this in mind, which if the following compounds should be most soluble in water? Group of answer choices LiBr LiI LiF LiClarrow_forward
- Q4: Write organic product(s) of the following reactions and show the curved-arrow mechanism of the reactions. Br MeOH OSO2CH3 MeOHarrow_forwardProvide the correct IUPAC name for the compound shown here. Reset cis- 5- trans- ☑ 4-6- 2- 1- 3- di iso tert- tri cyclo sec- oct but hept prop hex pent yl yne ene anearrow_forwardQ6: Predict the major product(s) for the following reactions. Note the mechanism (SN1, SN2, E1 or E2) the reaction proceeds through. If no reaction takes place, indicate why. Pay attention to stereochemistry. NaCN DMF Br σ Ilm... Br H Br H H NaCN CH3OH KOtBu tBuOH NaBr H₂O LDA Et2O (CH3)2CHOH KCN DMSO NaOH H₂O, A LDA LDA Systemarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY