
Solutions Manual For Chemistry: Structure And Properties
2nd Edition
ISBN: 9780134460697
Author: Tro
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 18, Problem 83E
Interpretation Introduction
(b)
To determine:
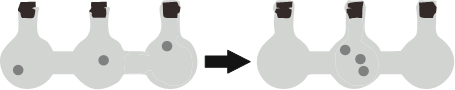
The sign of ∆Ssys for process of water is boiling, water freezing.
Interpretation Introduction
(c)
To determine:
The sign of ∆Ssys for process of water is boiling, water freezing with respect to
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An einstein is the amount of energy needed to dissociate 1 mole of a substance. If we have 0.58 moles, do we need 0.58 einsteins to dissociate that substance?
If the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.Data: Energy of each photon: 0.7835x10-18 J.
If the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.
Chapter 18 Solutions
Solutions Manual For Chemistry: Structure And Properties
Ch. 18 - What is the first law of thermodynamics, and how...Ch. 18 - What is nature’s heat tax, and how does it relate...Ch. 18 - What is a perpetual motion machine? Can such a...Ch. 18 - Is it more efficient to heat your home with a...Ch. 18 - What is a spontaneous process? Provide an example.Ch. 18 - Explain the difference between the spontaneity of...Ch. 18 - What is the precise definition of entropy? What is...Ch. 18 - Why does the entropy of a gas increase when it...Ch. 18 - Explain the difference between macrostates and...Ch. 18 - Based on its fundamental definition, explain why...
Ch. 18 - State the second law of thermodynamics. How does...Ch. 18 - What happens to the entropy of a sample of matter...Ch. 18 - Explain why water spontaneously freezes to form...Ch. 18 - Why do exothermic processes tend to be spontaneous...Ch. 18 - What is the significance of the change in Gibbs...Ch. 18 - Predict the spontaneity of a reaction (and the...Ch. 18 - State the third law of thermodynamics and explain...Ch. 18 - Why is the standard entropy of a substance in the...Ch. 18 - How does the standard entropy of a substance...Ch. 18 - How can you calculate the standard entropy change...Ch. 18 - Describe the three different methods to calculate...Ch. 18 - Why is free energy “free”?Ch. 18 - Explain the difference between G and G .Ch. 18 - Why does water spilled on the floor evaporate even...Ch. 18 - How do you calculate the change ¡n free energy for...Ch. 18 - How does the value of G for a reaction relate to...Ch. 18 - Prob. 27ECh. 18 - Prob. 28ECh. 18 - Prob. 29ECh. 18 - Prob. 30ECh. 18 - Calculate the change in entropy that occurs in the...Ch. 18 - Prob. 32ECh. 18 - Calculate the change ¡n entropy that occurs in the...Ch. 18 - Prob. 34ECh. 18 - Without doing any calculations, determine the sign...Ch. 18 - Prob. 36ECh. 18 - Without doing any calculations, determine the sign...Ch. 18 - Prob. 38ECh. 18 - Calculate Ssurr at the indicated temperature for...Ch. 18 - Prob. 40ECh. 18 - Given the values of Hrxn , Srxn and T, determine...Ch. 18 - Prob. 42ECh. 18 - Prob. 43ECh. 18 - Prob. 44ECh. 18 - Calculate the free energy change for the reaction...Ch. 18 - Prob. 46ECh. 18 - Prob. 47ECh. 18 - Predict the conditions (high temperature, low...Ch. 18 - How does the molar entropy of a substance change...Ch. 18 - What is the molar entropy of a pure crystal at 0...Ch. 18 - For each pair of substances, choose the one that...Ch. 18 - For each pair of substances, choose the one that...Ch. 18 - Rank each set of substances in order of increasing...Ch. 18 - Prob. 54ECh. 18 - Use data from Appendix IIB to calculate Srxn for...Ch. 18 - Use data from Appendix IIB to calculate Srxn for...Ch. 18 - Find S for the formation of CH2Cl2(g) from its...Ch. 18 - Prob. 58ECh. 18 - Methanol burns in oxygen to form carbon dioxide...Ch. 18 - In photosynthesis, plants form glucose (C6H12O6)...Ch. 18 - For each reaction, calculate Hrxn , Srxn and Grxn...Ch. 18 - For each reaction calculate Hrxn , Srxn and Grxn...Ch. 18 - Use standard free energies of formation to...Ch. 18 - Use standard free energies of formation to...Ch. 18 - Consider the reaction: 2NO(g)+O2(g)2NO2(g)...Ch. 18 - Prob. 66ECh. 18 - Determine G for the reaction:...Ch. 18 - Prob. 68ECh. 18 - Consider the sublimation of iodine at 25.0°C:...Ch. 18 - Consider the evaporation of methanol at 25.0°C....Ch. 18 - Consider the reaction: CH3OH(g)CO(g)+2H2(g)...Ch. 18 - Consider the reaction: CO2(g)+CCl4(g)2COCl2(g)...Ch. 18 - Use data from Appendix IIB to calculate the...Ch. 18 - Prob. 74ECh. 18 - Prob. 75ECh. 18 - Prob. 76ECh. 18 - Prob. 77ECh. 18 - Prob. 78ECh. 18 - Consider the reaction: H2(g)+I2(g)2HI(g) The...Ch. 18 - Consider the reaction: 2N0(g) — O(g) 2N02(g) The...Ch. 18 - The change in enthalpy (Hrxn) for a reaction is...Ch. 18 - Prob. 82ECh. 18 - Prob. 83ECh. 18 - Prob. 84ECh. 18 - Our atmosphere is composed primarily of nitrogen...Ch. 18 - Prob. 86ECh. 18 - Ethene (C2H4) can be halogenated by the reaction:...Ch. 18 - H2 reacts with the halogens (X2) according to the...Ch. 18 - Consider this reaction occurring at 298 K:...Ch. 18 - Consider this reaction occurring at 298 K:...Ch. 18 - Prob. 91ECh. 18 - Prob. 92ECh. 18 - These reactions are important in catalytic...Ch. 18 - Prob. 94ECh. 18 - All the oxides of nitrogen have positive values of...Ch. 18 - Prob. 96ECh. 18 - Consider the reaction X2(g)2X(g) . When a vessel...Ch. 18 - Prob. 98ECh. 18 - Indicate and explain the sign of Suniv for each...Ch. 18 - The Haber process is very important for...Ch. 18 - A metal salt with the formula MCl2 crystallizes...Ch. 18 - The solubility of AgCI(s) in water at 25°C is...Ch. 18 - Review the subsection in this chapter entitled...Ch. 18 - Calculate the entropy of each state and rank the...Ch. 18 - Suppose we redefine the standard state as P=2atm ....Ch. 18 - The G for the freezing of H2O(l) at 10°C is 210...Ch. 18 - Consider the reaction that occurs during the Haber...Ch. 18 - The salt ammonium nitrate can follow three modes...Ch. 18 - Given the tabulated data, calculate Svap for each...Ch. 18 - Prob. 110ECh. 18 - Prob. 111ECh. 18 - Consider the changes in the distribution of nine...Ch. 18 - Prob. 113ECh. 18 - Prob. 114ECh. 18 - Prob. 115ECh. 18 - The reaction A(g)B(g) has an equilibrium constant...Ch. 18 - Prob. 117ECh. 18 - Prob. 118ECh. 18 - Prob. 119ECh. 18 - Have each group member look up Hf and S for one...Ch. 18 - Calculate G at 25°C for the reaction in the...Ch. 18 - Prob. 122ECh. 18 - Which reaction Is most likely to have a positive...Ch. 18 - Prob. 2SAQCh. 18 - Arrange the gases—F2, Ar, and CH3F—in order of...Ch. 18 - Prob. 4SAQCh. 18 - Prob. 5SAQCh. 18 - For a certain reaction Hrxn=255kJ and Srxn=211J/K...Ch. 18 - Prob. 7SAQCh. 18 - s8. Use standard free energies of formation to...Ch. 18 - Prob. 9SAQCh. 18 - For the following reaction, Grxn=9.4kJ at 25 °C....Ch. 18 - Prob. 11SAQCh. 18 - Prob. 12SAQCh. 18 - Prob. 13SAQCh. 18 - Prob. 14SAQCh. 18 - Prob. 15SAQCh. 18 - Prob. 16SAQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The quantum yield of the photochemical decay of HI is 2. Calculating the moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. Calculate the number of Einsteins absorbed per mole knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. How many moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forward
- If the energy absorbed per mole of photons is 450 kJ, the number of Einsteins absorbed per 1 mole.arrow_forwardWhen propionic aldehyde in vapor form at 200 mmHg and 30°C is irradiated with radiation of wavelength 302 nm, the quantum yield with respect to the formation of CO is 0.54. If the intensity of the incident radiation is 1.5x10-3 W, find the rate of formation of CO.arrow_forwardDraw mechanismarrow_forward
- Does Avogadro's number have units?arrow_forwardExplain why the total E in an Einstein depends on the frequency or wavelength of the light.arrow_forwardIf the dissociation energy of one mole of O2 is 5.17 eV, determine the wavelength that must be used to dissociate it with electromagnetic radiation. Indicate how many Einstein's of this radiation are needed to dissociate 1 liter of O2 at 25°C and 1 atm of pressure.Data: 1 eV = 96485 kJ mol-1; R = 0.082 atm L K-1; c = 2.998x108 m s-1; h = 6.626x10-34 J s; NA = 6.022x 1023 mol-1arrow_forward
- Indicate the number of Einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy (wavelength 475 nm).arrow_forwardIndicate the number of einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy?arrow_forwardA unit used in photochemistry is the einstein. If 400 kJ mol-1 of energy has been absorbed, how many einsteins is this equivalent to?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY