
PHY F/SCIENTIST MOD MASTERING 24 MO
17th Edition
ISBN: 9780137319497
Author: Knight
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 56EAP
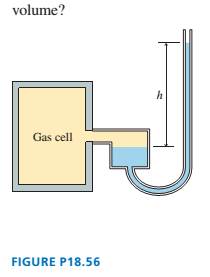
The mercury manometer shown in FIGURE P18.56 is attached
to a gas cell. The mercury height h is 120 mm when the cell is
placed in an ice-water mixture. The mercury height drops to
30 mm when the device is carried into an industrial freezer.
What is the freezer temperature?
Hint: The right tube of the manometer is much narrower than the
left tube. What reasonable assumption can you make about the gas
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Eddie Hall is the current world record holder in the deadlift, a powerlifting maneuver in which a weighted barbell is lifted from the ground to waist height, then dropped. The figure below
shows a side view of the initial and final positions of the deadlift.
a
0 = 55.0°
Fift
h22.5 cm
i
hy = 88.0 cm
b
i
solve for (_) N
Two boxes of fruit on a frictionless horizontal surface are connected by a light string as in the figure below, where m₁ = 11 kg and m₂ = 25 kg. A force of F = 80 N is applied to the 25-kg
box.
mq
m1
Applies
T
Peaches
i
(a) Determine the acceleration of each box and the tension in the string.
acceleration of m₁
acceleration of m₂
tension in the string
m/s²
m/s²
N
(b) Repeat the problem for the case where the coefficient of kinetic friction between each box and the surface is 0.10.
acceleration of m₁
acceleration of m₂
tension in the string
m/s²
m/s2
N
Chapter 18 Solutions
PHY F/SCIENTIST MOD MASTERING 24 MO
Ch. 18 - Prob. 1CQCh. 18 - Prob. 2CQCh. 18 - Prob. 3CQCh. 18 - Prob. 4CQCh. 18 - Prob. 5CQCh. 18 - Prob. 6CQCh. 18 - Prob. 7CQCh. 18 - Prob. 8CQCh. 18 - Prob. 9CQCh. 18 - A gas undergoes the process shown in FIGURE...
Ch. 18 - Prob. 11CQCh. 18 - Prob. 12CQCh. 18 - Prob. 1EAPCh. 18 - Prob. 2EAPCh. 18 - What is the diameter of a copper sphere that has...Ch. 18 - Prob. 4EAPCh. 18 - Prob. 5EAPCh. 18 - How many atoms are in a 2.0 cm × 2.0 cm × 2.0 cm...Ch. 18 - Prob. 7EAPCh. 18 - An element in its solid phase has mass density...Ch. 18 - .0 mol of gold is shaped into a sphere. What is...Ch. 18 - What volume of aluminum has the same number of...Ch. 18 - Prob. 11EAPCh. 18 - Prob. 12EAPCh. 18 - Prob. 13EAPCh. 18 - A concrete bridge is built of 325-cm-long concrete...Ch. 18 - A surveyor has a steel measuring tape that is...Ch. 18 - Two students each build a piece of scientific...Ch. 18 - Prob. 17EAPCh. 18 -
18. What is the temperature in °F and the...Ch. 18 - Prob. 19EAPCh. 18 - .0 mol of gas at a temperature of -120°C fills a...Ch. 18 - Prob. 21EAPCh. 18 - Prob. 22EAPCh. 18 - Prob. 23EAPCh. 18 - Prob. 24EAPCh. 18 - Prob. 25EAPCh. 18 - Prob. 26EAPCh. 18 - Prob. 27EAPCh. 18 - Prob. 28EAPCh. 18 - A rigid, hollow sphere is submerged in boiling...Ch. 18 -
30. A rigid container holds hydrogen gas at a...Ch. 18 - Prob. 31EAPCh. 18 - Prob. 32EAPCh. 18 - Prob. 33EAPCh. 18 - Prob. 34EAPCh. 18 - Prob. 35EAPCh. 18 - Prob. 36EAPCh. 18 - Prob. 37EAPCh. 18 - .0050 mol of gas undergoes the process 1 2 3...Ch. 18 - Prob. 39EAPCh. 18 - Prob. 40EAPCh. 18 - Prob. 41EAPCh. 18 - Prob. 42EAPCh. 18 - Prob. 43EAPCh. 18 - A 15°C, 2.0-cm-diameter aluminum bar just barely...Ch. 18 - Prob. 45EAPCh. 18 - Prob. 46EAPCh. 18 - Prob. 47EAPCh. 18 - Prob. 48EAPCh. 18 - Prob. 49EAPCh. 18 - The 3.0-m-long pipe in FIGURE P18.50 is closed at...Ch. 18 - Prob. 51EAPCh. 18 - An electric generating plant boils water to...Ch. 18 - Prob. 53EAPCh. 18 - The air temperature and pressure in a laboratory...Ch. 18 - Prob. 55EAPCh. 18 - The mercury manometer shown in FIGURE P18.56 is...Ch. 18 - Prob. 57EAPCh. 18 - The 50 kg circular piston shown in FIGURE P18.58...Ch. 18 - Prob. 59EAPCh. 18 - .0 g of helium gas follows the process 1? 2 ?3...Ch. 18 - Prob. 61EAPCh. 18 - 62. FIGURE P18.62 shows two different processes...Ch. 18 - Prob. 63EAPCh. 18 - Prob. 64EAPCh. 18 - Prob. 65EAPCh. 18 - Prob. 66EAPCh. 18 - Prob. 67EAPCh. 18 - Prob. 68EAPCh. 18 - Prob. 69EAPCh. 18 - Prob. 70EAPCh. 18 - Prob. 71EAPCh. 18 - The cylinder in FIGURE CP18.72 has a moveable...Ch. 18 - Containers A and B in FIGURE CP18.73 hold the same...Ch. 18 - Prob. 74EAP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- All correct but t1 and t2 from part Aarrow_forwardThree long, straight wires are mounted on the vertices of an equilateral triangle as shown in the figure. The wires carry currents of I₁ = 3.50 A, I2 = 5.50 A, and I3 = 8.50 A. Each side of the triangle has a length of 34.0 cm, and the point (A) is located half way between (11) and (12) along one of the sides. Find the magnitude of the magnetic field at point (A). Solve in Teslas (T). I₁arrow_forwardNumber There are four charges, each with a magnitude of 2.38 μC. Two are positive and two are negative. The charges are fixed to the corners of a 0.132-m square, one to a corner, in such a way that the net force on any charge is directed toward the center of the square. Find the magnitude of the net electrostatic force experienced by any charge. ips que Mi Units estic re harrow_forward
- Two long, straight wires are separated by distance, d = 22.0 cm. The wires carry currents of I1 = 7.50 A and I2 = 5.50 A in opposite directions, as shown in the figure. Find the magnitude of the net magnetic field at point (B). Let r₁ = 12.0 cm, r2 = 7.00 cm, and r3 = 13.0 cm. Solve in T. 12 d A √3arrow_forwardThank you in advance, image with question is attached below.arrow_forwardQuestion is attached, thank you.arrow_forward
- Two very small spheres are initially neutral and separated by a distance of 0.612 m. Suppose that 4.12 × 1013 electrons are removed from one sphere and placed on the other. (a) What is the magnitude of the electrostatic force that acts on each sphere? (b) Is the force attractive or repulsive?arrow_forwardEstimate the diameter of the Moon. During a total solar eclipse, the Moon passes in front of the Sun so that during “totality” their apparent sizes match and the Moon blocks light from the Sun shining on the Earth. a) What do you predict the size of the Moon would be if you were to use a pinhole in an aluminum holder, meter stick, and white paper screen to project light from the full Moon through a pinhole onto a screen that is one meter away from the pinhole? b) Describe in detail how you would use this apparatus and your knowledge of pinhole phenomena to estimate the diameter of the Moon. Assume that the distance between the Earth and the Moon is 250,000 miles.arrow_forwardThe following data was collected for a friction experiment in which an object was observed moving at constant speed over a surface. Graph the Applied Force versus the Normal Force and determine the coefficient of friction. Is this value the coefficient of kinetic friction or the coefficient of static friction? Justify your answer. Trial Normal Force Applied Force 1 4.13 1.44 2 6.41 1.68 3 8.94 2.82 4 11.34 3.94 5 13.82 5.05arrow_forward
- 1. Measurements and Linear Regression 1.1 Introduction The objective of this lab assignment is to represent measurement data in graphical form in order to illustrate experimental data and uncertainty visually. It is often convenient to represent experimental data graphically, not only for reporting results but also to compute or measure several physical parameters. For example, consider two physical quantities represented by x and y that are linearly related according to the algebraic relationship, y=mx+b, (1.1) where m is the slope of the line and b is the y-intercept. In order to assess the linearity between y and x, it is convenient to plot these quantities in a y versus x graph, as shown in Figure 1.1. Datapoints Line of regression Figure 1.1: Best fit line example. Once the data points are plotted, it is necessary to draw a "best fit line" or "regression line" that describes the data. A best fit line is a straight line that is the best approximation of the given set of data, and…arrow_forwardPlease help with Statistical Analysis table. These are trials from a Newton's Laws of Motion lab, please help with standard deviation and margin of error. Thanks!arrow_forwardplease solve and answer the question correctly. thank you!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY