
(a)
Interpretation:
The structures of the
Concept introduction:
The Wilkinson’s Catalyst is a common name of coordination compound
Answer to Problem 18.15P
The structures of the transition-metal complexes involved in each of the given mechanistic step are shown below.
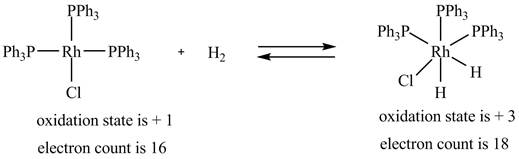
1. The oxidative addition reaction is shown below.
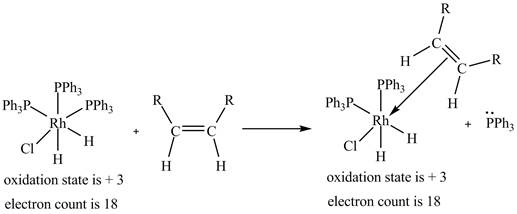
2. The ligand substitution of one
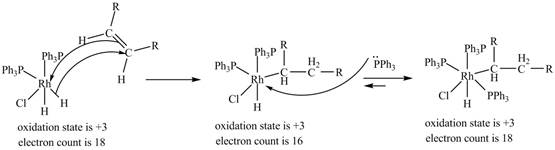
3. 1, 2-insertion of alkene into a
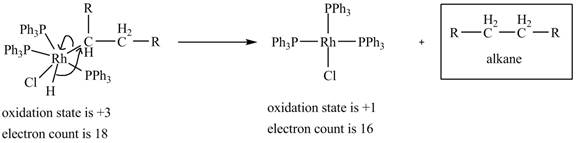
4. Reductive elimination of the
Explanation of Solution
The general formula for the calculation of electron count in a given formula is shown below.
The number of valence electrons present in rhodium is
1. In oxidative addition reaction, the central metal atom gets oxidized with the addition of two ligands and there is an increase in electron count at the central metal atom.
The oxidative addition reaction is shown below.
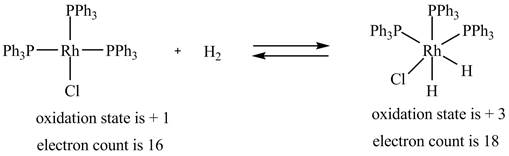
Figure 1
The electron count in
The electron count in
2. The ligand substitution reaction of one
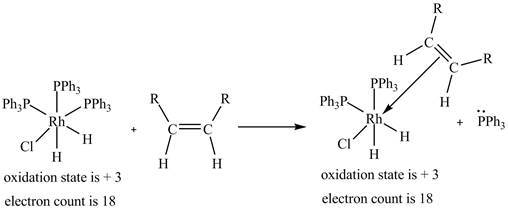
Figure 2
The electron count in
The electron count in
3. 1, 2-insertion of alkene into a
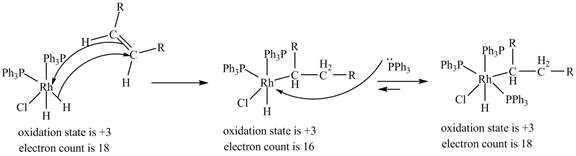
Figure 3
The electron count in
The electron count in
The electron count in
4. Reductive elimination of the alkane product to generate the catalyst is shown below.
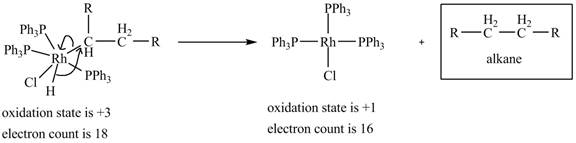
Figure 4
The electron count in
The electron count in
The catalytic hydrogenation of alkene by wilkinson’s catalyst and electron count is shown in Figure 1, 2, 3 and 4.
(b)
Interpretation:
The stereochemistry of the product if
Concept introduction:
The complex that follows
Answer to Problem 18.15P
The reaction between cis-alkene with the deuterium substituted wilkinson’s catalyst is shown below.
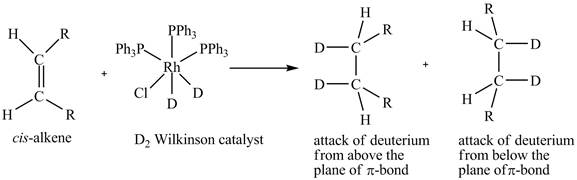
This shows syn-addition of deuterium on alkene.
Explanation of Solution
Reduction of cis-alkene with the deuterium substituted Wilkinson’s catalyst.
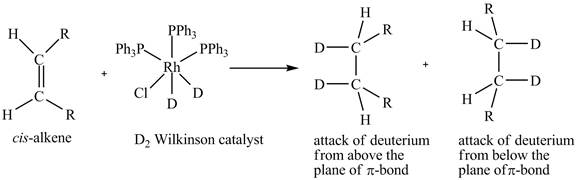
Figure 5
In the reduction of cis-alkene with the deuterium substituted Wilkinson’s catalyst. Hydrogen added on Wilkinson’s catalyst for hydrogenation of alkene is replaced by deuterium. The alkene is present in the plane of the paper, the deuterium can attack the alkene either from below the plane or above the plane.
Therefore, this shows that syn-addition of deuterium also takes place.
The reaction in Figure 5 shows that syn addition of hydrogen takes place even when it is substituted by deuterium.
Want to see more full solutions like this?
Chapter 18 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Construct a molecular orbital energy-level diagram for BeH2. Sketch the MO pictures (schematic representation) for the HOMO and LUMO of BeH2 [Orbital Potential Energies, H (1s): -13.6 eV; Be (2s): -9.3 eV, Be (2p): -6.0 eV]arrow_forwardIndicate the isomers of the A(H2O)6Cl3 complex. State the type of isomerism they exhibit and explain it briefly.arrow_forwardState the formula of the compound potassium μ-dihydroxydicobaltate (III) tetraoxalate.arrow_forward
- Consider the reaction of the cyclopentanone derivative shown below. i) NaOCH2CH3 CH3CH2OH, 25°C ii) CH3!arrow_forwardWhat constitutes a 'reference material', and why does its utilization play a critical role in the chemical analysis of food products? Provide examples.arrow_forwardExplain what calibration is and why it is essential in relation to food analysis. Provide examples.arrow_forward
- The cobalt mu-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forwardThe cobalt mi-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forward3. Arrange the different acids in Exercise B # 2 from the strongest (1) to the weakest acid (10). 1. 2. (strongest) 3. 4. 5. 6. 7. 8. 9. 10 10. (weakest)arrow_forward
- Name Section Score Date EXERCISE B pH, pOH, pка, AND PKD CALCULATIONS 1. Complete the following table. Solution [H+] [OH-] PH РОН Nature of Solution A 2 x 10-8 M B 1 x 10-7 M C D 12.3 6.8 2. The following table contains the names, formulas, ka or pka for some common acids. Fill in the blanks in the table. (17 Points) Acid Name Formula Dissociation reaction Ka pka Phosphoric acid H₂PO₁ H3PO4 H++ H₂PO 7.08 x 10-3 Dihydrogen H₂PO H₂PO H+ HPO 6.31 x 10-6 phosphate Hydrogen HPO₁ 12.4 phosphate Carbonic acid H2CO3 Hydrogen HCO 6.35 10.3 carbonate or bicarbonate Acetic acid CH,COOH 4.76 Lactic acid CH₂CHOH- COOH 1.38 x 10 Ammonium NH 5.63 x 10-10 Phenol CH₂OH 1 x 10-10 Protonated form CH3NH3* 3.16 x 10-11 of methylaminearrow_forwardIndicate whether it is true that Co(III) complexes are very stable.arrow_forwardMnO2 acts as an oxidant in the chlorine synthesis reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





