
EBK ORGANIC CHEMISTRY
9th Edition
ISBN: 8220100591310
Author: McMurry
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.SE, Problem 68AP
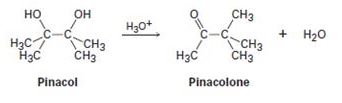
2, 3-Dimethyl-2, 3-butanediol has the common name pinacol. On heating with aqueous acid, pinacol rearranges to pinacolone, 3, 3-dimethyl-2-butanone. Suggest a mechanism for this reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
a. Explain Why electron withdrawing groupe
tend to be meta-Directors. Your answer Should
lyclude all apropriate. Resonance contributing
Structures
6.
Explain why -ll is an ortho -pura
drccton evon though chlorine has a very High
Electronegativity
C.
Ν
H
a.
H3C.
N
H3C
CH3
HCN
Chapter 17 Solutions
EBK ORGANIC CHEMISTRY
Ch. 17.1 - Give IUPAC names for the following compounds:Ch. 17.1 - Prob. 2PCh. 17.2 - The following data for isomeric four-carbon...Ch. 17.2 - Rank the following substances in order of...Ch. 17.2 - Prob. 5PCh. 17.3 - Prob. 6PCh. 17.4 - What reagent would you use to accomplish each of...Ch. 17.4 - Prob. 8PCh. 17.5 - Prob. 9PCh. 17.5 - Prob. 10P
Ch. 17.5 - Use the reaction of a Grignard reagent with a...Ch. 17.6 - How would you carry out the following...Ch. 17.6 - What products(s) would you expect from dehydration...Ch. 17.7 - What alcohols would give the following products on...Ch. 17.7 - What products would you expect from oxidation of...Ch. 17.8 - TMS ethers can be removed by treatment with...Ch. 17.9 - Show the mechanism for the reaction of...Ch. 17.11 - Prob. 18PCh. 17.11 - When the 1HNMR spectrum of an alcohol is run in...Ch. 17.SE - Give IUPAC names for the following compounds:Ch. 17.SE - Draw the structure of the carbonyl compound(s)...Ch. 17.SE - Prob. 22VCCh. 17.SE - Prob. 23VCCh. 17.SE - Name and assign R or S stereochemistry to the...Ch. 17.SE - Evidence for the intermediate carbocations in the...Ch. 17.SE - Acid-catalyzed dehydration of 2,...Ch. 17.SE - Prob. 27MPCh. 17.SE - Treatment of the following epoxide with aqueous...Ch. 17.SE - Prob. 29MPCh. 17.SE - Prob. 30MPCh. 17.SE - Identify the type of substitution mechanism (SN1,...Ch. 17.SE - The conversion of 3 alcohols into alkenes under...Ch. 17.SE - Prob. 33MPCh. 17.SE - The trimethylsilyl (TMS) protecting group is one...Ch. 17.SE - When the alcohol below is treated with POCI3 and...Ch. 17.SE - Phenols generally have lower pKa’s than...Ch. 17.SE - Give IUPAC names for the following compounds:Ch. 17.SE - Draw and name the eight isomeric alcohols with...Ch. 17.SE - Prob. 39APCh. 17.SE - Named bombykol, the sex pheromone secreted by the...Ch. 17.SE - Carvacrol is a naturally occurring substance...Ch. 17.SE - What Grignard reagent and what carbonyl compound...Ch. 17.SE - What carbonyl compounds would you reduce to...Ch. 17.SE - What carbonyl compounds might you start with to...Ch. 17.SE - Prob. 45APCh. 17.SE - What products would you obtain from reaction of...Ch. 17.SE - Prob. 47APCh. 17.SE - How would you prepare the following compounds from...Ch. 17.SE - Prob. 49APCh. 17.SE - What products would you expect to obtain from...Ch. 17.SE - Prob. 51APCh. 17.SE - Propose structures for alcohols that have the...Ch. 17.SE - Propose a structure consistent with the following...Ch. 17.SE - The 1HNMR spectrum shown is that of...Ch. 17.SE - A compound of unknown structure gave the following...Ch. 17.SE - Propose a structure for a compound C15H24O that...Ch. 17.SE - Prob. 57APCh. 17.SE - Prob. 58APCh. 17.SE - Rank the following substituted phenols in order of...Ch. 17.SE - Benzvl chloride can be converted into benzaldehvde...Ch. 17.SE - Prob. 61APCh. 17.SE - Prob. 62APCh. 17.SE - Prob. 63APCh. 17.SE - Prob. 64APCh. 17.SE - Prob. 65APCh. 17.SE - Prob. 66APCh. 17.SE - Dehydration of trans-2-methylcyclopentanol with...Ch. 17.SE - 2, 3-Dimethyl-2, 3-butanediol has the common name...Ch. 17.SE - As a rule, axial alcohols oxidize somewhat faster...Ch. 17.SE - Prob. 70APCh. 17.SE - A problem often encountered in the oxidation of...Ch. 17.SE - Identify the reagents a-f in the Following scheme:Ch. 17.SE - Prob. 73APCh. 17.SE - Prob. 74APCh. 17.SE - Compound A, C8H10O, has the IR and 1H NMR spectra...Ch. 17.SE - Prob. 76APCh. 17.SE - Prob. 77AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- ол 2. восцапан (46:00) Curtius rearrangment 1. NaN3, heat -OHarrow_forwardQuestion 1. Please predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers.arrow_forwardElectrochemistry. Briefly describe the Donnan potential.arrow_forward
- Indicate what the Luther equation is used for?arrow_forwardIndicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.arrow_forwardAt an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forward
- Please predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Hint: In this case you must choose the best answer to demonstrate the stereochemistry of H2 addition. 1.03 2. (CH3)2S BIZ CH₂OH 2. DMS KMnO4, NaOH ΖΗ Pd or Pt (catalyst) HBr 20 1 HBr ROOR (peroxide) HO H-SO HC 12 11 10 BH, THE 2. H2O2, NaOH Brz cold HI 19 18 17 16 MCPBA 15 14 13 A Br H₂O BH3⚫THF Brz EtOH Pd or Ni (catalyst) D₂ (deuterium) 1. Os04 2. H2O2 CH3CO3H (peroxyacid) 1. MCPBA 2. H₂O* H B + H H H "H C H H Darrow_forwardExplain how Beer’s Law can be used to determine the concentration in a selected food sample. Provide examples.arrow_forwardExplain the importance of having a sampling plan with respect to food analysis. Explain the importance of having a sampling plan with respect to food analysis. Provide examples.arrow_forward
- Please predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forwardBriefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forwardElectrochemistry. Briefly describe the Gibbs model and the Gibbs absorption equation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License