
a) PBr3
Interpretation:
The product obtained when 3-(m-bromophenyl)-2-butanol is treated with PBr3.
Concept introduction:
Alcohols when reacted with PBr3 yield the corresponding alkyl bromides along with H3BO3. When treated with aqueous H2SO4 they undergo dehydration to give
To give:
The products obtained when the alcohol shown is treated with PBr3.
Answer to Problem 22VC
The product obtained when the alcohol is treated with PBr3 is
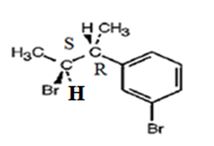
Explanation of Solution
The reaction of the alcohol with PBr3 is a bimolecular nucleophilic substitution (SN2) reaction. It takes place with inversion in configuration at the reaction centre to yield an alkyl bromide.
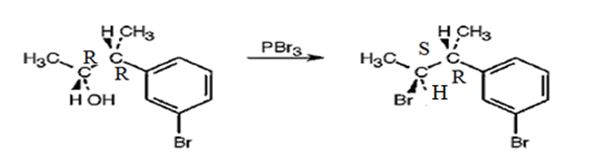
The product obtained when the alcohol is treated with PBr3 is

b) H2SO4
Interpretation:
The product obtained when 3-(m-bromophenyl)-2-butanol is treated with aqueous H2SO4.
Concept introduction:
Alcohols when reacted with PBr3 yield the corresponding alkyl bromides along with H3BO3. When treated with aqueous H2SO4 they undergo dehydration to give alkenes. They react with SOCl2 to yield the corresponding alkyl halide along with SO2 and HCl. Dess-Martin periodinate oxidizes 20 alcohols to ketones. In aromatic electrophilic substitution reactions the alkyl group is an ortho and para directing and activating group. Bromine also is an ortho and para directing but deactivating group.
To give:
The products obtained when the alcohol shown is treated with aqueous H2SO4.
Answer to Problem 22VC
The products obtained when the alcohol is treated with aqueous H2SO4 are two isomeric alkenes (E) and (Z). E isomer is obtained as the major product.
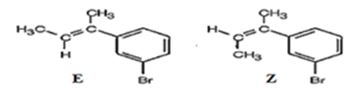
Explanation of Solution
The alcohol gets dehydrated when treated with aqueous H2SO4 yields the E and Z isomers of an alkene. The E isomer is more stable and obtained as the major product as the bulky groups are far away.
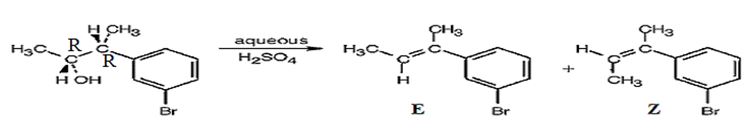
The products obtained when the alcohol is treated with aqueous H2SO4 isomeric alkenes (E) and (Z) are produced. E isomer is obtained as the major product.

c) SOCl2
Interpretation:
The product obtained when 3-(m-bromophenyl)-2-butanol is treated with SOCl2.
Concept introduction:
Alcohols when reacted with PBr3 yield the corresponding alkyl bromides along with H3BO3. When treated with aqueous H2SO4 they undergo dehydration to give alkenes. They react with SOCl2 to yield the corresponding alkyl halide along with SO2 and HCl. Dess-Martin periodinate oxidizes 20 alcohols to ketones. In aromatic electrophilic substitution reactions the alkyl group is an ortho and para directing and activating group. Bromine also is an ortho and para directing but deactivating group.
To give:
The products obtained when the alcohol shown is treated with SOCl2.
Answer to Problem 22VC
The product obtained when the alcohol is treated with SOCl2 is
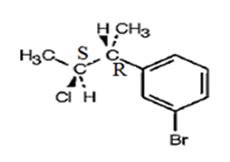
Explanation of Solution
The reaction of the alcohol with SOCl2 is a bimolecular nucleophilic substitution (SN2) reaction. It takes place with inversion in configuration at the reaction centre to yield an alkyl chloride.
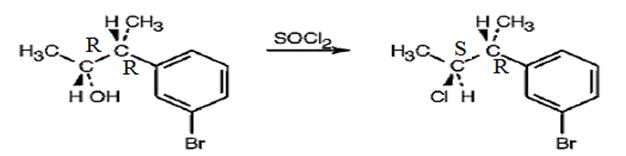
The product obtained when the alcohol is treated with SOCl2 is

d) Dess-Martin periodinate
Interpretation:
The product obtained when 3-(m-bromophenyl)-2-butanol is treated with Dess-Martin periodinate.
Concept introduction:
Alcohols when reacted with PBr3 yield the corresponding alkyl bromides along with H3BO3. When treated with aqueous H2SO4 they undergo dehydration to give alkenes. They react with SOCl2 to yield the corresponding alkyl halide along with SO2 and HCl. Dess-Martin periodinate oxidizes 20 alcohols to ketones. In aromatic electrophilic substitution reactions the alkyl group is an ortho and para directing and activating group. Bromine also is an ortho and para directing but deactivating group.
To give:
The products obtained when the alcohol shown is treated with Dess-Martin periodinate.
Answer to Problem 22VC
The product obtained when the alcohol is treated with Dess-Martin periodinate is
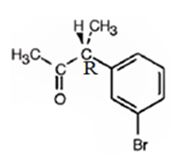
Explanation of Solution
The alcohol is a 20 in nature. Dess-Martin periodinate oxidizes it into a ketone.
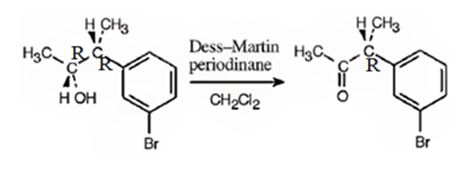
The product obtained when the alcohol is treated with Dess-Martin periodinate is

e) Br2, FeBr3
Interpretation:
The product obtained when 3-(m-bromophenyl)-2-butanol is treated Br2, FeBr3.
Concept introduction:
Alcohols when reacted with PBr3 yield the corresponding alkyl bromides along with H3BO3. When treated with aqueous H2SO4 they undergo dehydration to give alkenes. They react with SOCl2 to yield the corresponding alkyl halide along with SO2 and HCl. Dess-Martin periodinate oxidizes 20 alcohols to ketones. In aromatic electrophilic substitution reactions the alkyl group is an ortho and para directing and activating group. Bromine also is an ortho and para directing but deactivating group.
To give:
The products obtained when the alcohol shown is treated Br2, FeBr3.
Answer to Problem 22VC
The products obtained when the alcohol is treated with Br2, FeBr3 are
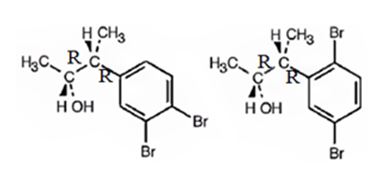
Explanation of Solution
The alcohol undergoes bromination in the benzene ring when treated with Br2, FeBr3. Both bromine and an alkyl group are attached to the ring. Both alkyl and Br are ortho and para directing, the alkyl is activating while the Br is deactivating. Hence during bromination the bromine enters into the ortho and para positions to yield the products.
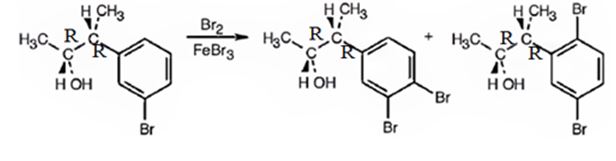
The products obtained when the alcohol is treated with Br2, FeBr3 are

Want to see more full solutions like this?
Chapter 17 Solutions
Study Guide with Student Solutions Manual for McMurry's Organic Chemistry, 9th
- Steps and explanation please. Add how to solve or target similar problems.arrow_forwardWould the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardThis organic molecule is dissolved in an acidic aqueous solution: OH OH A short time later sensitive infrared spectroscopy reveals the presence of a new C = O stretch absorption. That is, there must now be a new molecule present with at least one C = O bond. In the drawing area below, show the detailed mechanism that could convert the molecule above into the new molecule. Videos 849 Explanation Check C Click and drag to start dwing a structure. # 3 MAR 23 Add/Remove steparrow_forward||| 7:47 ull 57% ← Problem 19 of 48 Submit Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the product of this carbocation rearrangement. Include all lone pairs and charges as appropriate. H 1,2-alkyl shift +arrow_forwardWould the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forwardBelow is the SN1 reaction of (S)-3-chlorocyclohexene and hydroxide (OH). Draw the missing curved arrows, lone pairs of electrons, and nonzero formal charges. In the third box, draw the two enantiomeric products that will be produced. 5th attempt Please draw all four bonds at chiral centers. Draw the two enantiomeric products that will be produced. Draw in any hydrogen at chiral centers. 1000 4th attempt Feedback Please draw all four bonds at chiral centers. 8. R5 HO: See Periodic Table See Hint H Cl Br Jid See Periodic Table See Hintarrow_forwardShow that a molecule with configuration π4 has a cylindrically symmetric electron distribution. Hint: Let the π orbitals be equal to xf and yf, where f is a function that depends only on the distance from the internuclear axis.arrow_forward(a) Verify that the lattice energies of the alkali metal iodides are inversely proportional to the distances between the ions in MI (M = alkali metal) by plotting the lattice energies given below against the internuclear distances dMI. Is the correlation good? Would a better fit be obtained by plotting the lattice energies as a function of (1 — d*/d)/d, as theoretically suggested, with d* = 34.5 pm? You must use a standard graphing program to plot the graph. It generates an equation for the line and calculates a correlation coefficient. (b) From the graph obtained in (a), estimate the lattice energy of silver iodide. (c) Compare the results of (b) with the experimental value of 886 kJ/mol. If they do not agree, explain the deviation.arrow_forwardCan I please get help with #3 & 4? Thanks you so much!arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

